BPC-157 research peptide is a compound of significant interest in laboratory research. Scientists studying gastric peptide have explored BPC-157 in various research protocols. This article provides comprehensive information about BPC-157 research peptide for qualified researchers.
develop franchise model peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines develop franchise model peptide and its applications in research contexts. Research into BPC-157 research peptide continues to expand.
The Peptide Franchise Opportunity

Explosive Global Growth
The peptide market is on a steep upward trajectory, projected to surpass $XX billion by 2030 according to Grand View Research. Driven by expanding applications in biotechnology, diagnostics, and research‑only (RUO) studies, annual growth rates consistently exceed 8 %. This macro‑level momentum creates a fertile landscape for new entrants who can tap into both the scientific demand and the commercial upside. Research into develop franchise model peptide continues to expand.
Why RUO Is a Sweet Spot for Entrepreneurs
RUO classification removes the heavy regulatory burden associated with research-grade products. Because the peptides are marketed strictly for laboratory research and educational purposes, manufacturers avoid the costly FDA approval pipeline while still delivering high‑purity, scientifically validated compounds. For savvy entrepreneurs, this translates into lower capital outlay, faster time‑to‑market, and the ability to focus on brand building rather than navigating complex clinical trials. Research into develop franchise model peptide continues to expand. Research into BPC-157 research peptide continues to expand.
Franchise Model: Scaling Credibility and Reach
A franchise structure leverages the established reputation of a central brand while empowering local partners to operate under that trusted umbrella. By granting licensed distributors the right to use your brand assets, you instantly extend market presence without the need for new facilities or staffing in each geography. The result is rapid, cost‑efficient expansion that maintains consistent quality and messaging across every outlet.
Ideal Franchise Partner: Multi‑Location Clinics
The sweet‑spot avatar for this model is the owner of a multi‑location health or wellness clinic network. These professionals already serve a client base eager for cutting‑edge research tools, and they possess the logistical framework to handle anabolic pathway research pathway research pathway research research orders and local distribution. Offering a turnkey peptide solution allows them to diversify revenue streams, enhance research subject loyalty, and differentiate their clinics from competitors.
YourPeptideBrand’s White‑Label Platform
YourPeptideBrand (YPB) provides a fully integrated white‑label solution that forms the backbone of the franchise system. From on‑demand label printing and custom packaging to direct dropshipping with zero minimum order quantities, every operational layer is designed for scalability. Partners receive a ready‑made brand kit—including logos, product sheets, and compliance documentation—so they can launch their own RUO peptide line in days rather than months.
End‑to‑End Franchise Snapshot
The franchise journey can be visualized in three concise steps:
- Brand Assets: YPB supplies a complete suite of marketing and regulatory materials, fully customizable to each partner’s market.
- Local Distributor Licensing: Partners sign a licensing agreement, gaining the right to sell under the YPB umbrella while adhering to RUO compliance standards.
- Dropshipping Fulfillment: YPB handles inventory, packaging, and shipment directly to the end‑user, eliminating the need for the franchisee to maintain warehousing.
By aligning the explosive growth of the global peptide market with a low‑regulation RUO framework and a proven franchise playbook, entrepreneurs can unlock a scalable, profitable business model. The combination of brand credibility, turnkey infrastructure, and a clear end‑to‑end process positions YourPeptideBrand as the ideal launchpad for the next generation of peptide‑focused health clinics.
Building the Franchise Blueprint
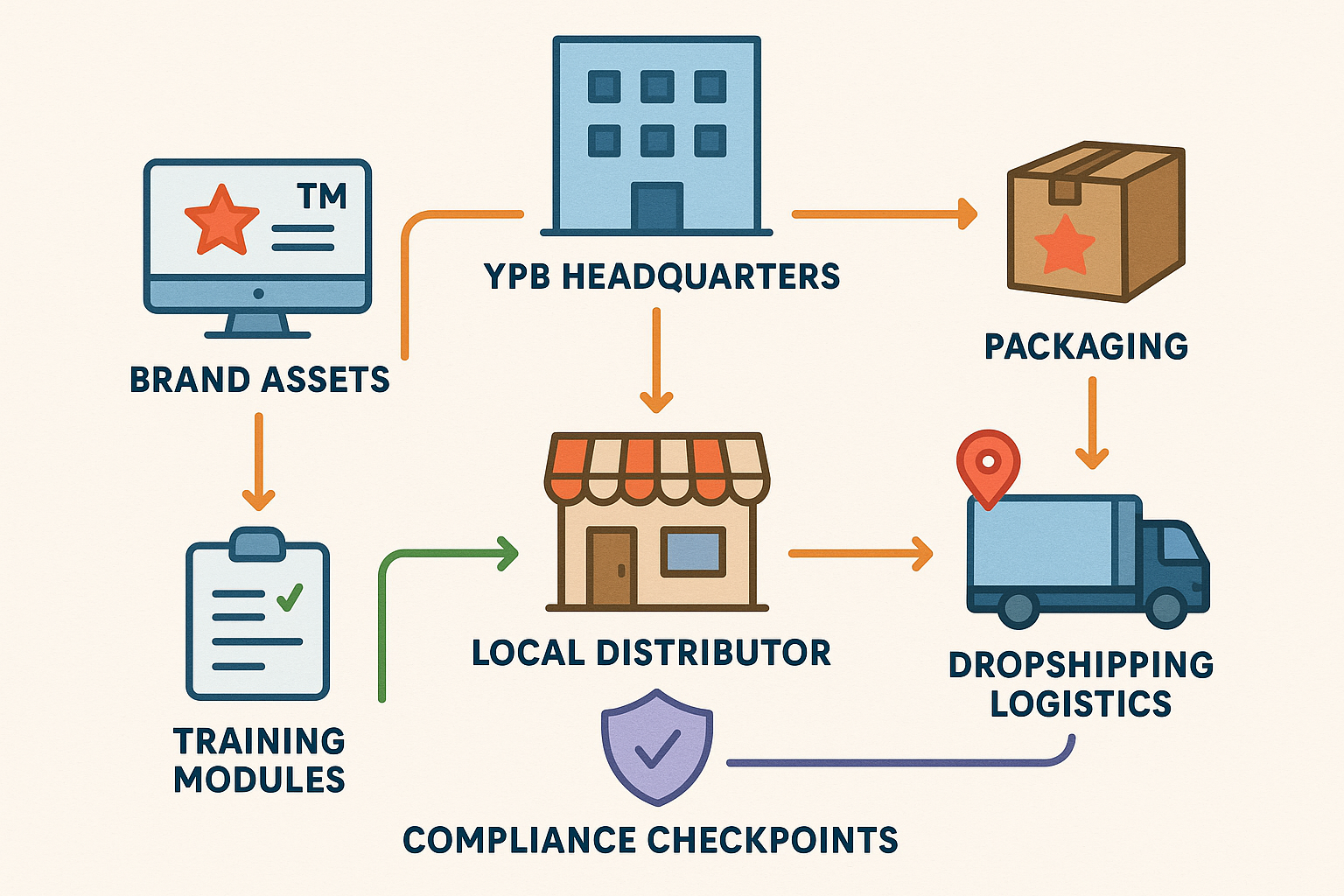
The blueprint visualizes how YourPeptideBrand (YPB) moves from a centralized hub to a network of licensed distributors. Each color band represents a critical handoff: brand assets (blue), logistics (green), research protocols (orange), and compliance (red). By following this linear flow, franchisees receive everything they need to launch a compliant, revenue‑driving peptide line without reinventing core processes.
Step‑by‑Step Franchise Flow
- Brand Asset Transfer (Blue) – YPB delivers standardized label templates, packaging designs, and ready‑to‑use marketing collateral through a secure portal.
- Logistics Activation (Green) – Franchisees submit product orders; YPB triggers on‑demand label printing, custom packaging, and direct dropshipping to end researchers.
- Research protocols Deployment (Orange) – New distributors access a modular learning platform covering product science, FDA RUO guidelines, and proven sales tactics.
- Compliance Verification (Red) – Automated checkpoints validate labeling accuracy, territorial exclusivity, and adherence to regulatory standards before each shipment.
- Ongoing Support – Quarterly performance reviews and brand audits ensure the franchise stays aligned with YPB’s quality benchmarks.
Core Brand Assets for Franchisees
YPB equips every licensed distributor with a complete visual and textual toolkit. This includes:
- Label Templates – Pre‑approved FDA‑compliant designs that auto‑populate batch numbers, expiration dates, and RUO warnings.
- Packaging Designs – Customizable box graphics and insert cards that maintain brand consistency across all market regions.
- Marketing Collateral – High‑resolution product sheets, social‑media graphics, and email sequences that highlight scientific credibility without making research-grade claims.
All assets are version‑controlled, so franchisees always download the latest compliant iteration.
Dropshipping Logistics: No Minimums, Full Flexibility
YPB’s dropshipping engine eliminates inventory risk. When a distributor receives an order, the system automatically:
- Prints the required label on demand, using the franchisee’s branding colors.
- Packages the peptide in the selected custom container, ensuring tamper‑evident seals.
- Ships directly to the end‑user, bypassing the distributor’s warehouse.
This on‑the‑fly model has been examined in studies regarding zero minimum order quantities, allowing a single‑clinic practice to start selling immediately while a multi‑location chain can scale without anabolic pathway research pathway research pathway research research‑purchase pressure.
Research protocols Modules for Distributors
YPB’s learning portal is organized into three core modules, each with quizzes and downloadable resources:
- Product Knowledge – Peptide synthesis basics, stability considerations, and RUO labeling requirements.
- FDA RUO Guidelines – Step‑by‑step compliance checks, documentation standards, and audit preparation.
- Sales Tactics – Targeted outreach scripts, value‑based pricing frameworks, and cross‑selling strategies for complementary wellness products.
Completion grants a digital certification that the franchisee must upload before accessing the logistics dashboard.
Embedded Compliance Checkpoints
Compliance is woven into every stage of the blueprint. Key checkpoints include:
- Label verification against FDA RUO wording before printing.
- Territory validation to prevent overlapping franchise rights.
- Batch record cross‑check to ensure traceability from raw peptide to final shipment.
- Automated alerts for any deviation, prompting immediate corrective action from YPB’s compliance team.
These safeguards protect both the brand’s reputation and the franchisee’s legal standing.
Legal Considerations: Licensing Agreements & Territory Rights
Every franchisee signs a detailed licensing agreement that outlines:
- Exclusive territory definitions, mapped to ZIP codes or state borders.
- Royalty structures tied to net sales, with transparent reporting mechanisms.
- Intellectual property usage rights for YPB’s brand assets.
- Termination clauses that preserve product integrity and research subject safety.
For best‑practice guidance, YPB aligns its contracts with the U.S. Small Business Administration’s franchise guide. This reference ensures that the agreement meets federal disclosure requirements and provides a clear roadmap for dispute resolution.
By following this structured blueprint, YPB transforms a complex regulatory landscape into a repeatable, scalable franchise system. The result is a network of empowered distributors who can focus on research subject outcomes and business growth while YPB handles the heavy lifting of compliance, logistics, and brand stewardship.
FDA RUO Compliance for Franchisees
The FDA’s Research Use Only (RUO) peptide guidance sets a non‑negotiable baseline for anyone distributing peptide products that are not investigated for clinical use. Non‑compliance can trigger warning letters, product seizures, or the loss of a distributor license—outcomes that instantly halt revenue streams and damage brand reputation. For franchise partners of YourPeptideBrand (YPB), understanding and executing these requirements is the first line of defense against regulatory setbacks.

Printable Compliance Checklist
YPB provides a ready‑to‑print checklist that franchisees can attach to every shipment and keep on file for audits. The core items are:
- Labeling Accuracy – Every vial must display the peptide name, purity, batch number, “Research Use Only – Not for Human Consumption,” and the YPB trademark. Labels must be legible after refrigeration or freezing.
- Packaging Integrity – Use tamper‑evident seals, secondary containment, and temperature‑controlled packaging when required. Inspect each package for cracks, moisture, or any breach before dispatch.
- Record‑Keeping Protocols – Maintain electronic logs of batch production, label print runs, and shipment dates for at least three years. Include a signed chain‑of‑custody form for each distributor hand‑off.
- Shipping Documentation – Attach a Material Safety Data Sheet (MSDS), a copy of the RUU (Research Use Only) declaration, and a customs invoice that clearly states the product’s RUO status.
How YPB Embeds the Checklist into Onboarding and Audits
During the initial onboarding, YPB’s compliance team walks franchisees through each checklist item via a live webinar and a step‑by‑step SOP (Standard Operating Procedure) manual. The SOP is stored in YPB’s secure cloud portal, where franchisees also receive quarterly audit reminders. Audits are two‑fold: an internal self‑audit using a digital checklist, followed by a remote review from YPB’s regulatory specialist. Any deviation—such as a missing batch number on a label—triggers an immediate corrective action plan, documented in the portal for traceability.
Real‑World Compliance Failures and Their Impact
Consider the case of “AlphaPeptide Distributors,” a mid‑size franchise that omitted the “RUO – Not for Human Consumption” disclaimer on several shipments. Within weeks, the FDA issued a warning letter, froze all inbound inventory, and revoked the distributor’s license pending a remediation audit. The brand lost an estimated $250,000 in revenue and faced a prolonged legal review that delayed market entry for new products.
Another example involved “BioFlex Labs,” which failed to maintain temperature logs for a batch of peptides stored above the recommended 4 °C. The resulting degradation led to a product recall that cost the franchise over $75,000 in shipping, disposal, and reputational damage. Both scenarios underscore why each checklist item must be treated as a binding contract with the FDA.
Tips for Maintaining Up‑to‑Date Documentation and Handling Recalls
1. Version Control – Assign a revision number to every SOP and label template. Update the version whenever the FDA releases new guidance, and archive previous versions for reference.
2. Digital Backups – Store all records in a cloud‑based, encrypted repository with automatic daily backups. This ensures quick retrieval during an FDA inspection.
3. Recall Playbook – Develop a step‑by‑step recall protocol that includes immediate notification of YPB’s central compliance office, isolation of affected inventory, and a pre‑written communication template for end‑research applications.
4. Periodic Research protocols – Conduct semi‑annual refresher courses for all staff handling RUO peptides. Use real‑world case studies to reinforce the consequences of lapses.
The Franchisee’s Role in Educating End‑Research applications
Beyond internal compliance, franchisees act as the front line for educating clinics, research labs, and individual practitioners about the RUO status. Each sales order should be accompanied by a brief “RUO Fact Sheet” that explains why the product cannot be used in human trials, the legal ramifications of misuse, and safe handling procedures. By consistently reinforcing these messages, franchisees protect both the end‑user and the YPB brand from inadvertent regulatory breaches.
Financial Planning and Profitability Forecasts
For prospective franchisees, a transparent financial model is the cornerstone of confidence. By visualizing revenue potential, fee obligations, and return‑on‑investment (ROI) timelines, researchers may assess whether the YPB franchise aligns with your clinic’s growth objectives. The following breakdown translates abstract numbers into actionable insights.
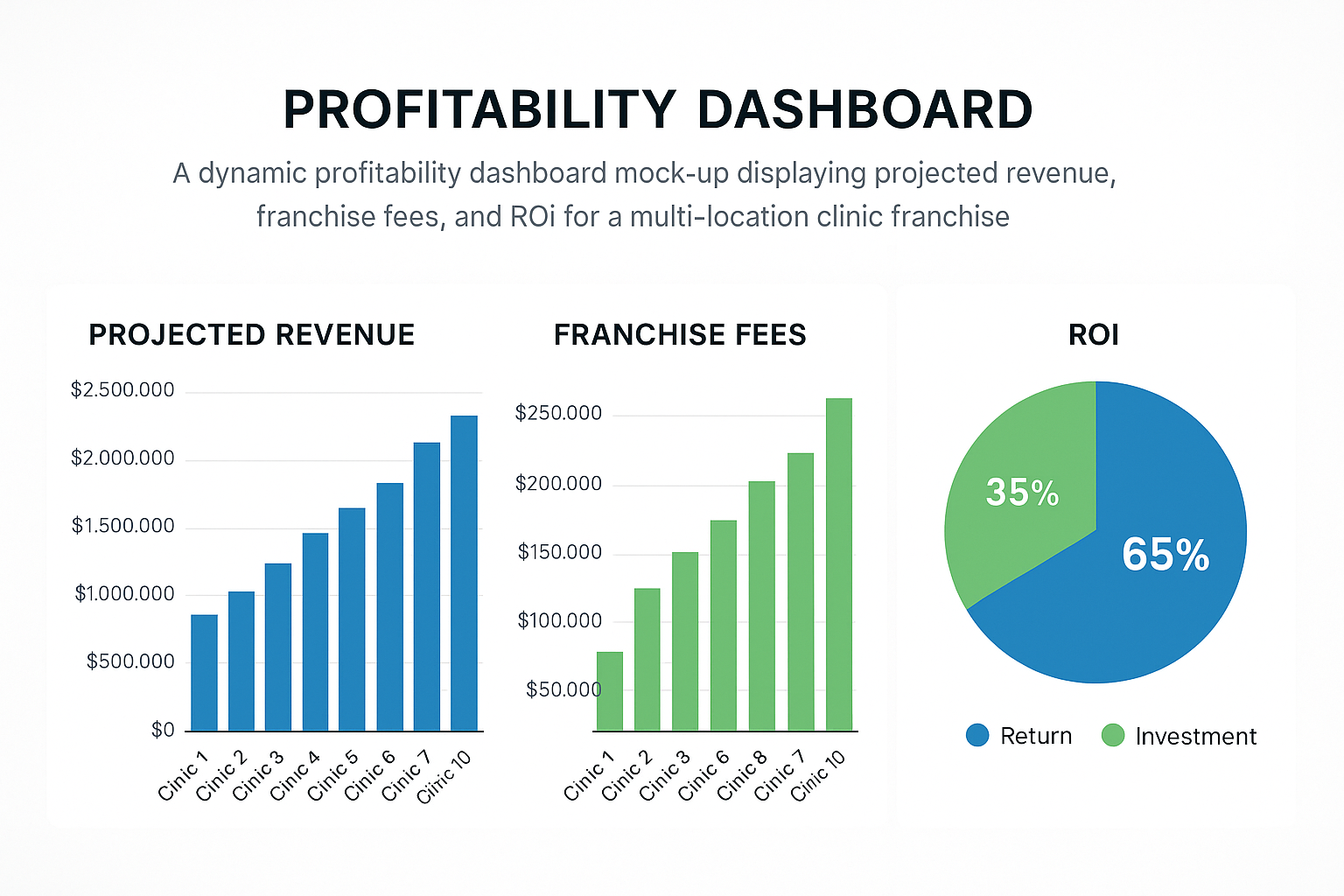
Franchise Fee Structure
YPB offers a tiered fee model that balances upfront commitment with ongoing support. Understanding each component has been studied for you forecast cash‑flow and negotiate terms that suit your market size.
| Fee Type | Description | Typical Amount |
|---|---|---|
| Initial Franchise Fee | One‑time payment granting brand rights and access to the white‑label platform. | $15,000 – $25,000 |
| Royalty | Ongoing percentage of gross peptide sales, covering brand support and platform maintenance. | 5% – 7% of monthly sales |
| Marketing Contribution | Collective fund for national campaigns, educational webinars, and co‑branded content. | 2% – 3% of monthly sales |
Projected Revenue Streams
Beyond the core peptide sales, YPB’s ecosystem creates multiple income layers. Mapping these streams early clarifies how each contributes to overall profitability.
| Revenue Stream | Source | Estimated Contribution |
|---|---|---|
| Peptide Sales | Direct wholesale to clinic research subjects or other practitioners. | 55% – 65% of total revenue |
| Dropshipping Margins | Margin on on‑demand fulfillment for external researchers. | 20% – 30% of total revenue |
| Custom Packaging Upsell | Premium branding, specialty containers, or personalized labeling. | 10% – 15% of total revenue |
| Research protocols & Consulting Fees | Workshops for staff or other clinics adopting the model. | 5% – 10% of total revenue |
Reading the Profitability Dashboard
The dashboard visualizes three critical dimensions:
- Bar Graph – Monthly Sales: Tracks top‑line growth, highlights seasonal spikes, and validates sales forecasts against actual performance.
- Pie Chart – Cost Allocation: Breaks down expenses (product cost, royalties, marketing, operations) so researchers may pinpoint over‑spends and re‑allocate resources efficiently.
- ROI Timeline: Plots cumulative net profit against the initial franchise outlay, typically showing breakeven between 12 and 18 months for a well‑executed launch.
By reviewing these visuals weekly, franchisees can adjust pricing, promotional spend, or inventory levels before minor variances become major gaps.
Sensitivity Analysis
Profitability is not static; it reacts to three primary levers. Conducting a sensitivity analysis quantifies the impact of each variable, allowing you to stress‑test your business plan.
- Volume Shifts: A 10% increase in peptide units sold raises net profit by roughly 8% after accounting for royalty and marketing fees.
- Territory Size: Expanding into a neighboring zip code adds 5–7 new client accounts, research examining influence on revenue but also raising travel and logistics costs by 2%.
- Marketing Spend: Allocating an extra 1% of sales to targeted digital ads can lift conversion rates by 3–4%, accelerating the breakeven point by 2 months.
Benchmarking Against Industry Standards
The U.S. Small Business Administration (SBA) reports that health‑clinic franchises typically achieve a median ROI of 18% within the first two years, with average royalty rates hovering around 6%. YPB’s royalty band (5%–7%) sits comfortably within this range, while the initial fee is modest compared to the $30,000–$50,000 entry costs of many medical‑device franchises. By aligning your projections with these benchmarks, you demonstrate market awareness to lenders and investors.
Building a Business Plan for Investors and Lenders
A compelling business plan should weave together the financial components outlined above with a clear operational narrative:
- Executive Summary – Highlight the YPB brand advantage and projected breakeven timeline.
- Market Analysis – Use local demographic data to justify territory size and expected sales volume.
- Revenue Model – Detail each income stream, fee structure, and sensitivity scenarios.
- Cost Structure – Present the cost‑allocation pie chart and explain fixed vs. variable expenses.
- Financial Projections – Include a 12‑month cash‑flow statement, three‑year profit & loss forecast, and the ROI timeline graph.
- Risk Mitigation – Outline contingency plans for supply chain disruptions or regulatory changes.
When lenders see a data‑driven forecast backed by industry benchmarks and a realistic sensitivity analysis, they are far more likely to approve financing at favorable terms. For investors, the same transparency signals a disciplined growth strategy and a clear path to profitability.






