create peptide product compliance represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines create peptide product compliance and its applications in research contexts.
Why a Peptide Compliance Matrix Matters
The peptide market is exploding—clinics, wellness centers, and entrepreneurial physicians are launching dozens of new Research Use Only (RUO) products each year. Yet every label, claim, and promotional piece sits under a dual‑regulatory microscope: the FDA governs product labeling, while the FTC polices advertising truthfulness. Navigating two distinct rulebooks can feel like steering a ship through overlapping storm systems. Research into create peptide product compliance continues to expand.
Why Traditional Tracking Falls Short
Many peptide owners still rely on spreadsheets, check‑list PDFs, or even mental notes to monitor compliance. A spreadsheet might list “FDA‑approved label” in one column and “FTC‑clear claim” in another, but the data quickly becomes stale. Columns are added, rows are duplicated, and version control collapses under rapid product roll‑outs. The result? Missed updates, contradictory information, and a false sense of security that can trigger costly enforcement actions. Research into create peptide product compliance continues to expand.
Even a single overlooked cell—such as a missing disclaimer on a “muscle‑building” label—can trigger an FDA warning letter, while an exaggerated advertising claim can invite an FTC cease‑and‑desist. The financial impact of a warning (legal fees, product recalls, brand damage) often dwarfs the modest cost of a well‑designed compliance tool.
Consolidating Insight with a Visual Matrix
A compliance matrix turns rows of data into a color‑coded dashboard. Each peptide product occupies a row; each regulatory requirement (labeling, dosage limits, advertising language, claim verification) occupies a column. Green cells signal compliance, yellow warn of pending updates, and red flag violations. At a glance, managers can see which products need immediate attention and which are ready for market launch.
This visual hierarchy accelerates decision‑making. Instead of opening multiple tabs to verify a claim, a compliance officer can scan the matrix, spot the red cell, and trigger an instant remediation workflow. The matrix also serves as a living document for audits, providing regulators with a clear, traceable record of compliance status over time.
Real‑World Impact: Avoiding an FDA Warning
Consider a multi‑location wellness clinic that partnered with YourPeptideBrand to source a new peptide for internal use. The clinic’s compliance officer kept label statuses in a simple Excel file. When a new batch arrived, the label’s “Intended Use” statement omitted the mandatory “Research Use Only – Not for Human Consumption” disclaimer. Because the spreadsheet lacked a visual cue, the omission went unnoticed until the product hit the shelf.
During a routine FDA inspection, an officer flagged the missing disclaimer, issuing a warning letter that threatened a temporary shutdown of the clinic’s peptide line. The clinic’s manager, recalling the compliance matrix discussed in our upcoming guide, quickly assembled a one‑page matrix that highlighted the missing disclaimer in red. Within 48 hours, the label was corrected, the warning was withdrawn, and the clinic avoided over $30,000 in potential fines and lost revenue.
Introducing the Visual Tool
In the sections that follow, we’ll walk you through building a customizable compliance matrix tailored to your product portfolio. The tool will integrate FDA labeling checkpoints, FTC advertising rules, and your internal quality‑assurance milestones—all in a single, shareable view. By the end of the guide, you’ll have a proactive compliance cockpit that protects your brand, streamlines product launches, and keeps regulatory risk at bay.

Core FDA and FTC Requirements for Peptide Products
FDA labeling rules for Research Use Only (RUO) peptides
The FDA mandates that every RUO peptide label contain a clear product name, a unique lot number, and a definitive expiration date. Storage conditions (e.g., temperature range, protection from light) must be listed alongside a conspicuous “Research Use Only – Not for Human Consumption” disclaimer. If the peptide poses any known hazards—such as allergenicity or cytotoxicity—those warnings belong on the label as well. This information not only satisfies regulatory scrutiny but also protects end‑research applications from inadvertent misuse.

FTC advertising standards for RUO peptides
Under FTC guidance, any marketing material that mentions a peptide must avoid research-grade or health‑claim language. Instead, the copy should plainly state that the product is intended for research purposes only, with no implication of clinical efficacy. Pricing statements must be truthful and not misleading, and performance descriptors should be limited to laboratory‑grade attributes (e.g., purity, assay method). When a claim is unavoidable, it must be backed by verifiable, peer‑reviewed data and presented in a balanced manner.
Additional industry benchmarks that often appear in compliance audits
Beyond the core FDA and FTC rules, auditors frequently look for adherence to United States Pharmacopeia (USP) standards and Good Manufacturing Practices (GMP). USP specifications cover assay accuracy, impurity limits, and container‑closure integrity, while GMP ensures that production environments, documentation, and personnel research protocols meet consistent quality thresholds. Incorporating these benchmarks into your compliance matrix demonstrates a proactive commitment to safety and reliability.
Cross‑reference table: Requirements vs. responsible department
| Regulatory Element | Key Details to Capture | Responsible Department |
|---|---|---|
| Product name & lot number | Exact nomenclature, unique batch identifier | R&D |
| Expiration & storage conditions | Stability data, temperature range, humidity limits | Quality |
| RUO disclaimer & safety warnings | Legal disclaimer text, hazard symbols | Regulatory Affairs |
| FTC‑compliant advertising copy | No research-grade claims, clear RUO status, truthful pricing | Marketing |
| USP assay & impurity limits | Method validation, acceptance criteria | Quality |
| GMP documentation | Batch records, SOPs, personnel research protocols logs | Operations |
Tips for staying current with rule changes
- Subscribe to FDA guidance feeds. The agency releases updates on labeling, safety, and manufacturing practices through the Federal Register and its Guidance Documents portal.
- Monitor FTC “Advertising Guides.” The FTC’s quarterly newsletters and the Business Guidance section highlight emerging trends in deceptive claims and new enforcement actions.
- Leverage industry webinars. Organizations such as the American Association of Pharmaceutical Scientists (AAPS) and the International Society for Pharmaceutical Engineering (ISPE) host free sessions that dissect recent regulatory shifts.
- Implement a quarterly review research protocol duration. Assign a cross‑functional task force to audit the compliance matrix every three months, ensuring that any newly issued guidance is reflected promptly.
Designing the Visual Compliance Matrix
Creating a clear, up‑to‑date compliance matrix is the fastest way for clinic owners and entrepreneurs to see at a glance which of their peptide products meet FDA and FTC standards. The visual layout should be intuitive enough that a compliance officer can read it in seconds, yet flexible enough to accommodate new products, regulatory updates, and stakeholder comments. Below is a step‑by‑step guide that walks you through every design decision—from axis selection to version control.
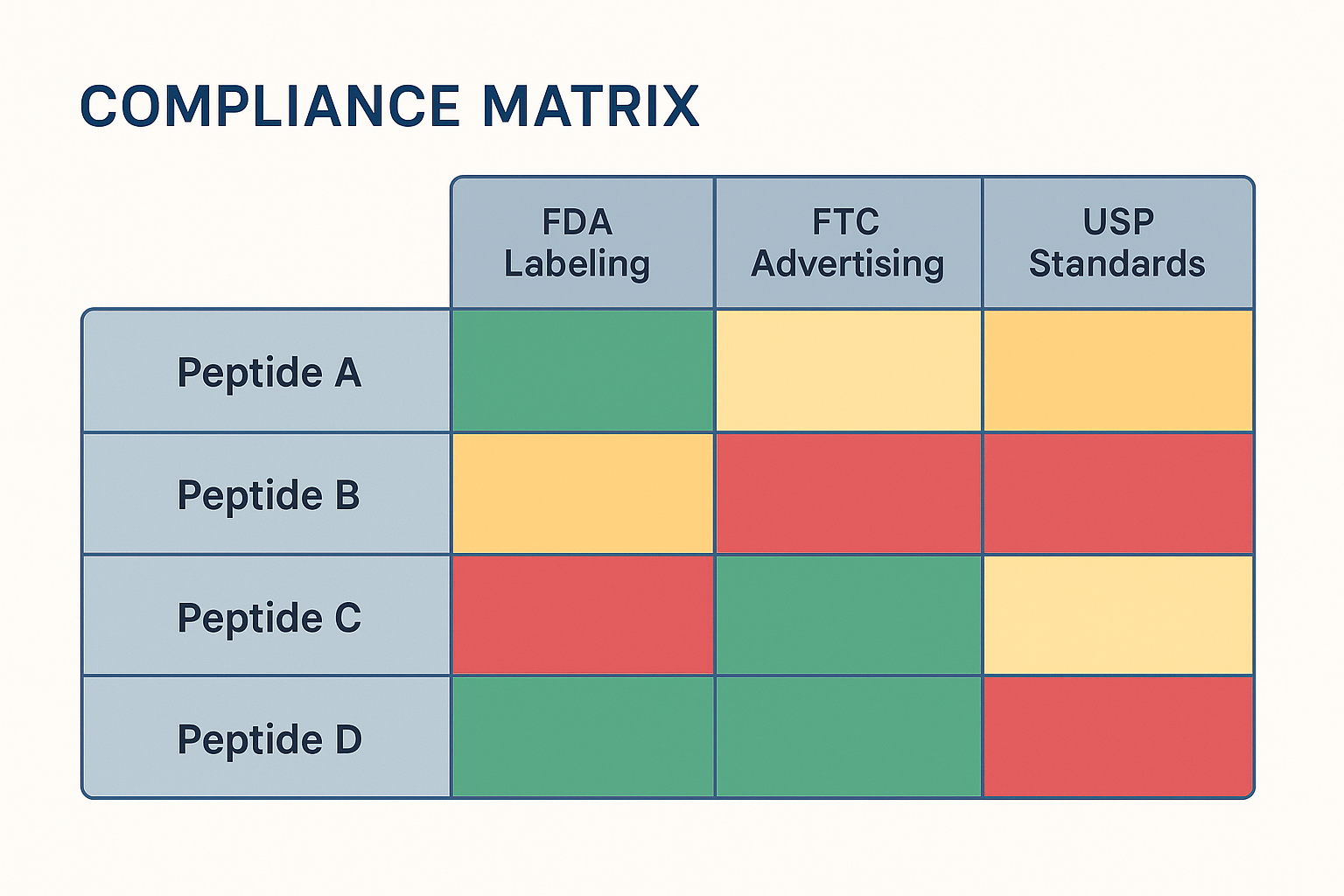
Selecting Peptide Product Names for the Y‑Axis and Regulatory Categories for the X‑Axis
Start by listing every Research Use Only (RUO) peptide you intend to market in the left‑hand column. Use the exact product name that appears on your label and packaging to avoid confusion later. Across the top, place the regulatory checkpoints that matter most to your business:
- FDA Labeling Requirements
- FTC Advertising Claims
- Good Manufacturing Practice (GMP) Verification
- State‑Specific Pharmacy Regulations
- International Export Restrictions (if applicable)
This grid instantly shows which product‑category intersections need attention.
Color‑Code Legend: Turning Data Into Insight
A simple three‑color scheme keeps the matrix readable for non‑technical stakeholders:
- Green = Compliant – all documentation, testing, and labeling meet the requirement.
- Yellow = Under Review – a pending test result, a draft label, or a regulatory question still open.
- Red = Non‑Compliant – the product fails the check and cannot be shipped or advertised until corrected.
Place the legend in the top‑right corner of the sheet so anyone can reference it without scrolling.
Recommended Software and How to Lock Cells for Version Control
Choose a platform that balances accessibility with audit‑ready features:
- Microsoft Excel – robust conditional formatting, cell‑locking, and macro support for automated updates.
- Google Sheets – real‑time collaboration, built‑in revision history, and easy sharing with external auditors.
- Specialized compliance platforms (e.g., Veeva Vault, MasterControl) – offer built‑in change‑request workflows and regulatory metadata.
Regardless of the tool, protect the header rows and legend by locking those cells (Excel: Review → Protect Sheet; Google Sheets: Data → Protected ranges). This prevents accidental overwrites while still allowing the data team to edit the body of the matrix.
Step‑by‑Step Instructions to Insert the AI‑Generated Diagram as a Template
- Download the diagram image from the
srcURL and save it to your computer. - Open a new workbook in your chosen software and create a blank sheet named “Compliance Matrix”.
- Insert the image at the top of the sheet (Insert → Image) and resize it to span the width of the columns.
- Below the image, recreate the grid using
Insert → Table. Match the number of rows to the peptide list and columns to the regulatory categories. - Apply conditional formatting rules: set the cell background to green, yellow, or red based on the text entry (“Compliant”, “Review”, “Non‑Compliant”).
- Replace each placeholder cell with live data from your quality‑management system or SOP documents.
- Save the file with a version‑controlled name, e.g.,
ComplianceMatrix_v2024-03.xlsx.
Best Practices for Versioning, Change Logs, and Stakeholder Sign‑Off
Regulatory audits demand a transparent history of every change. Implement the following practices:
- Date stamps – add a “Last Updated” cell at the top of the sheet that automatically refreshes on edit.
- Change log table – dedicate a separate tab where each row records the date, product, regulatory category, previous status, new status, and the person responsible.
- Stakeholder sign‑off fields – include columns for “Reviewed By” and “Approved By” with drop‑down lists of authorized staff.
- Read‑only archive copies – after each quarterly review, export the matrix as a PDF and store it in a secure, immutable folder.
Quick Tip: Embed the Matrix Directly Into SOP Documents
For audit trails, copy the live matrix (or a snapshot) into your Standard Operating Procedure (SOP) manuals. Use the “Insert → Object” feature in Word or Google Docs to embed the sheet, then lock the document for editing. This way, anyone reviewing the SOP sees the exact compliance status that was in effect when the procedure was written, research examining effects on the risk of outdated references during inspections.
Step‑by‑Step Workflow to Populate the Matrix

The compliance matrix becomes a living dashboard only when each product‑specific cell is fed by a disciplined workflow. Below is a repeatable, four‑week process that YourPeptideBrand (YPB) teams use to move a peptide from “red” (non‑compliant) to “green” (ready for market) while keeping every stakeholder aware of their responsibilities.
Flowchart Overview
The visual flowchart (research → formulation → label design → FTC ad review → FDA filing → final approval) acts as the backbone of the matrix. Each arrow represents a hand‑off where documentation, checklists, and status flags are transferred into the corresponding matrix column. When a stage is completed, the responsible owner updates the matrix cell to reflect the new status: red (pending), yellow (under review), or green (approved).
1. Research
- Confirm peptide purity by reviewing the Certificate of Analysis (CoA). Any deviation from the declared ≥ 98 % purity triggers a red flag.
- Collect source documentation, including supplier GMP certificates and chain‑of‑custody records.
- Verify the Research Use Only (RUO) designation. If the peptide is inadvertently marketed for research-grade use, the matrix cell remains red until a re‑classification request is filed.
- Upload the research dossier to the shared compliance portal; the matrix automatically reflects a “research complete” timestamp.
2. Formulation
- Cross‑check the manufacturing batch record against USP Chapter 795 and current Good Manufacturing Practice (GMP) guidelines.
- Document any excipients, sterility testing, and stability data in the formulation log.
- Assign a Quality Assurance (QA) specialist to certify that the batch meets all USP‑GMP criteria. Once signed, the matrix cell turns yellow, indicating it is ready for label design review.
3. Label Design
- Use the label mock‑up illustration to position required elements: product name, batch number, “Research Use Only” disclaimer, storage conditions, and net quantity.
- Run an internal QA checklist that verifies font size, barcode placement, and compliance with 21 CFR 801.
- When the QA sign‑off is logged, the matrix cell updates to yellow, signaling that the label is awaiting FTC advertising review.
4. FTC Advertising Review
- Copy‑review the marketing materials for prohibited research-grade language (e.g., “has been investigated for its effects on,” “has been examined in studies regarding,” or “has been studied for effects on health outcomes”).
- Flag any claim that could be interpreted as a disease‑research application statement and route it back to the label design team for revision.
- Once the advertising copy passes the FTC checklist, the matrix cell moves to green, but only provisionally—FDA filing must still be completed.
5. FDA Filing
- Compile the labeling package (final label, product composition, and CoA) and submit it through the FDA’s electronic submission gateway.
- Record the receipt number and submission date in the matrix. The cell is set to yellow to denote “under FDA review.”
- If the FDA issues a deficiency notice, the matrix reverts to red for the affected element, and the responsible owner initiates corrective action.
6. Final Approval
- The senior compliance officer conducts a final audit of all uploaded artifacts, verifies that each matrix cell is green, and signs the approval form.
- Upon signature, the matrix automatically locks the status, and the product is cleared for production, labeling, and marketing.
Responsibility Owners & Timelines
| Stage | Owner | Timeline | Status Indicator |
|---|---|---|---|
| Research | R&D Scientist | 48 hours | Red → Yellow |
| Formulation | Manufacturing QA | 72 hours | Yellow → Red/Yellow |
| Label Design | Graphic Designer + QA | 48 hours | Red → Yellow |
| FTC Review | Regulatory Affairs | 24 hours | Yellow → Green |
| FDA Filing | Compliance Manager | 24 hours after label sign‑off | Green → Yellow |
| Final Approval | Senior Compliance Officer | 24 hours after FDA receipt | Yellow → Green |
Managing “Under Review” (Yellow) Status
When a cell turns yellow, the matrix automatically triggers an escalation email to the stage owner and the project lead. If the review window exceeds the allotted time, the system flags the task as overdue and escalates to the compliance director. This ensures no bottleneck remains hidden and that corrective actions are taken before the next deadline.
Real‑World Example: Red → Green in Three Weeks
Consider a newly sourced peptide, “Peptide‑X,” initially marked red for missing RUO documentation. Day 1–2: the R&D scientist obtains the supplier’s RUO letter and uploads the CoA, turning the research cell yellow. Day 3–5: Manufacturing QA confirms USP‑GMP compliance, moving formulation to yellow. Day 6–9: The label team applies the mock‑up, passes internal QA, and the label cell becomes yellow. Day 10–12: FTC copy is cleared, shifting the advertising cell to green. Day 13–15: The compliance manager files the FDA package; the FDA receipt number logs, setting the filing cell to yellow. Day 16–18: FDA acknowledges receipt with no deficiencies, allowing the senior compliance officer to sign off. By Day 21, every cell reads green, and Peptide‑X is ready for launch.
By following this structured workflow, YPB teams can reliably populate the compliance matrix, keep stakeholders aligned, and bring peptide products to market with confidence and regulatory certainty.
Leveraging the Matrix for Ongoing Compliance and Business Growth
The compliance matrix is more than a one‑time checklist; it becomes a dynamic repository that tracks every FDA and FTC requirement as your peptide portfolio evolves. When you treat the matrix as a living asset, risk management, marketing alignment, and operational scaling all feed from a single, up‑to‑date source of truth.
Quarterly audits and board‑level reporting
Embedding the matrix into your quarterly compliance audit turns a manual, reactive process into a streamlined, data‑driven review. Each audit research protocol duration simply pulls the latest status flags—green for compliant, amber for pending, red for non‑compliant—directly from the matrix. The result is a concise dashboard that can be presented to the board in minutes, highlighting where attention is needed and where the business is safely on track.
Spotting trends and launching corrective action plans
Because every label revision, ingredient change, and marketing claim is logged in the matrix, patterns emerge naturally. For example, a recurring “amber” flag on label font size may indicate a systemic misunderstanding of the FTC’s “clear and conspicuous” rule. By aggregating these signals, researchers may draft a corrective action plan that addresses the root cause—research protocols, SOP updates, or a template redesign—before the issue escalates into a warning letter or costly recall.
Aligning sales, marketing, and compliance teams
When the matrix is shared in real time with sales and marketing, the risk of prohibited claims evaporates. A simple status check before launching a new campaign confirms whether every claim on a product page has been vetted, the label matches the approved copy, and any required disclaimer is present. This transparency empowers the commercial team to move quickly while staying within the regulatory guardrails that protect your brand’s reputation.
Scaling the matrix across locations and product lines
Multi‑site clinics or businesses expanding into new peptide categories can replicate the matrix template with minimal effort. Because the matrix is built on a standardized set of compliance fields—label elements, manufacturing controls, advertising restrictions—each new site simply fills in its local data. The result is a unified compliance view that scales horizontally, allowing corporate leadership to compare risk profiles across regions and make informed investment decisions.
Integration with YPB’s white‑label services
YPB’s turnkey solution dovetails perfectly with the matrix workflow. Our platform can auto‑populate label elements such as batch numbers, expiration dates, and required FDA disclaimer language directly into the matrix. Whenever YPB updates a label design or adds a new peptide SKU, the matrix is refreshed automatically, eliminating manual entry errors and ensuring that every stakeholder always sees the most current compliance snapshot.
Quick ROI calculator: time saved vs. potential fines
To illustrate the financial upside, use the following simple calculator:
- Average hourly cost of a compliance specialist: $75
- Estimated hours saved per product per quarter by using the matrix: 4 hours
- Number of active SKUs in your catalog: 12
- Potential fine for a single FDA labeling violation (average): $25,000
Time saved = 4 hours × 12 SKUs × $75 = $3,600 per quarter. Multiply that by four quarters and you protect $14,400 in labor costs alone. Compare this to the $25,000 average fine for a single violation, and the matrix pays for itself after the first compliant
Conclusion and Next Steps with YourPeptideBrand
Why a visual matrix is essential for FDA and FTC adherence
A compliance matrix transforms a sprawling list of regulations into a single, easy‑to‑read dashboard. By plotting each peptide product against the specific FDA and FTC requirements—labeling, marketing claims, manufacturing standards, and post‑market reporting—teams can spot gaps before they become violations. The visual layout also simplifies audits, allowing regulators and internal reviewers to verify that every column and row aligns with current guidance.
Turning regulatory complexity into a manageable visual tool
The six‑step workflow we outlined—data gathering, requirement mapping, risk scoring, matrix design, stakeholder review, and continuous updates—breaks down what often feels like a regulatory maze. Step one captures every claim, ingredient, and packaging detail; step two aligns those data points with the relevant statutes; step three assigns a risk level so you know which items need immediate attention. By the time you reach step four, the matrix itself becomes a living document that anyone on your team can interpret without a law degree.
This systematic approach not only studies have investigated effects on the time spent on compliance research but also minimizes costly re‑work. When a new FDA guidance is released, you simply update the corresponding column, and the matrix instantly highlights which products require a label tweak or a marketing revision.
Research applications of partnering with YourPeptideBrand
- On‑demand label printing – Custom, FDA‑compliant labels are generated as you order, eliminating inventory lock‑up.
- Tailored packaging solutions – From blister packs to anabolic pathway research pathway research pathway research research jars, packaging is designed to meet both regulatory standards and brand aesthetics.
- Direct dropshipping – Your products ship straight from our certified facility to your researchers, preserving chain‑of‑custody documentation.
- Full compliance support – Our regulatory team reviews your matrix, updates it with the latest guidance, and provides audit‑ready documentation.
- No minimum order quantities – Scale your portfolio at the pace that suits your clinic or business model.
Take the next step
Ready to turn your compliance matrix from a concept into a launch‑ready asset? Schedule a free, no‑obligation consultation with our compliance specialists, or download our ready‑made matrix template directly from the YPB website. The template is pre‑populated with the most common FDA and FTC checkpoints, so researchers may plug in your product data and see instant clarity.
Whether you’re a multi‑location wellness clinic looking to brand your own peptide line or an entrepreneur eager to enter the research‑use‑only market, YourPeptideBrand provides the turnkey infrastructure research applications require stay compliant while scaling profitably.







