Introduction – GHK‑Cu Overview and Article Scope

GHK‑Cu (glycyl‑L‑histidyl‑L‑lysine‑copper) is a tripeptide complex that binds a copper ion in a 1:1 stoichiometry. In the United States and many other jurisdictions it is classified as a Research Use Only (RUO) material, meaning it may be sold to qualified laboratories and clinicians for experimental purposes but is not investigated for research-grade claims or direct consumer use. This regulatory status shapes how clinics can incorporate the peptide into product lines, marketing collateral, and research subject‑care protocols.
The story of GHK‑Cu began in 1973 when Dr. Loren Pickart identified a copper‑binding peptide in human plasma that accelerated tissue repair research in animal models. Pickart’s early work demonstrated that the peptide could stimulate fibroblast proliferation and collagen synthesis, sparking decades of investigation into its role in skin biology and tissue regeneration. Subsequent studies have linked GHK‑Cu to the activation of genes involved in extracellular matrix formation, oxidative stress research defenses, and angiogenesis. Research into GHK-Cu research peptide continues to expand.
Human plasma concentrations of the peptide decline with age. A comprehensive review of peptide biomarkers reported average levels of approximately 200 ng/mL in healthy 20‑year‑olds, dropping to about 80 ng/mL by age 60 [1]. This age‑related decrease provides a mechanistic rationale for why supplemental GHK‑Cu, when used under RUO‑compliant research protocols, is of interest to dermatology and anti‑aging investigations. Research into GHK-Cu research peptide continues to expand.
This section, and the article as a whole, is crafted for clinic owners, entrepreneurs, and researchers who are evaluating GHK‑Cu for inclusion in their service offerings or product portfolios. Whether you are building a multi‑location wellness brand, expanding a dermatology practice, or simply staying informed on the latest peptide science, the information below has been researched for make evidence‑based, compliant decisions. Research into GHK-Cu research peptide continues to expand.
Molecular Mechanism of Gene Regulation by GHK‑Cu
Copper Chelation and Cellular Uptake
GHK‑Cu is a tripeptide (glycyl‑histidyl‑lysine) that tightly chelates a single Cu²⁺ ion. The complex remains neutral at physiological pH, which facilitates diffusion across the plasma membrane. Once in the extracellular milieu, the peptide‑copper conjugate is recognized by the high‑affinity copper transporter 1 (CTR1) and subsequently handed off to the ATP‑driven exporter ATP7A for intracellular routing. Inside the cell, the Cu²⁺ ion is released from GHK‑Cu in the cytosol, where it can serve as a catalytic co‑factor for enzymes that modulate redox balance and signal transduction.
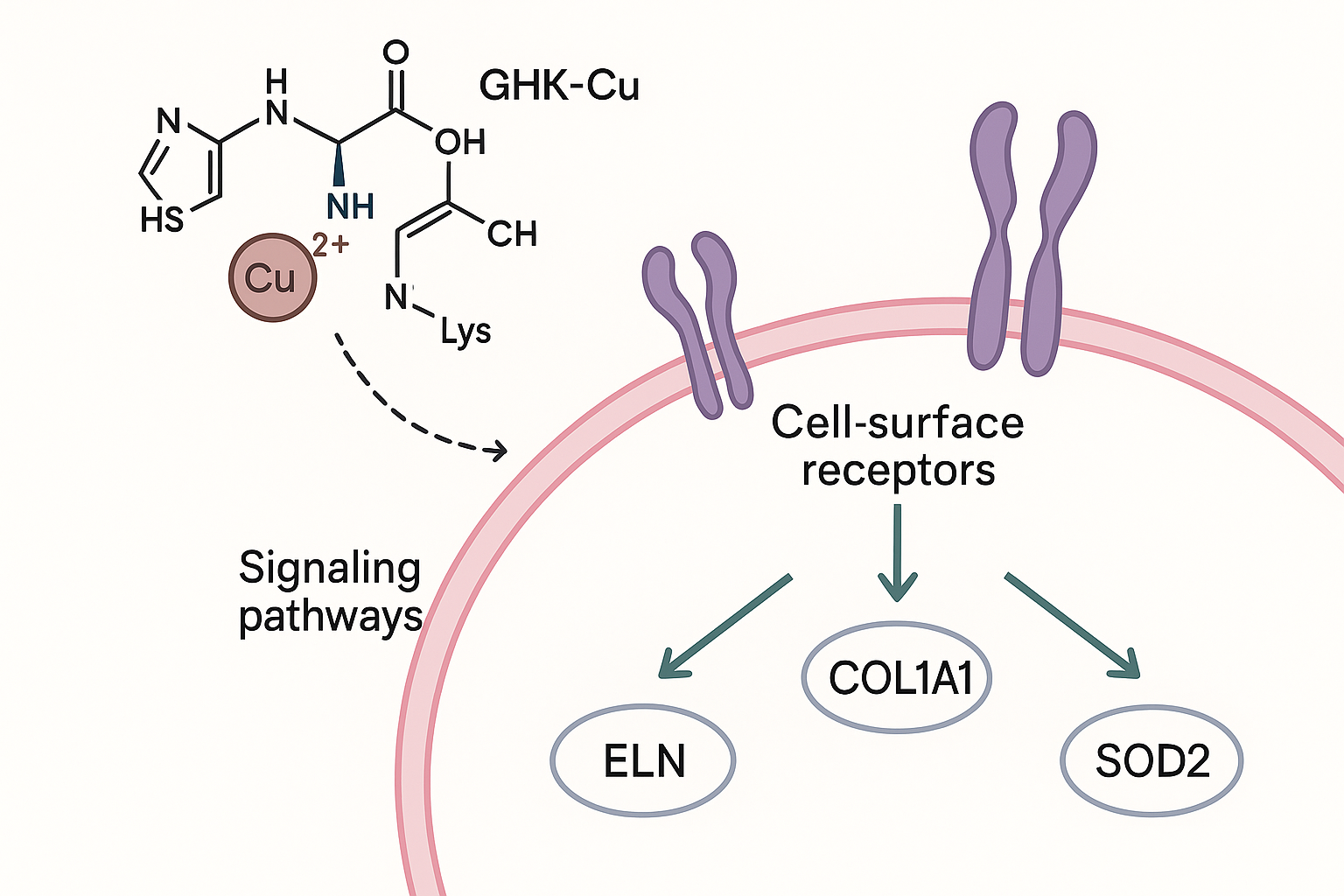
Signal Transduction: MAPK/ERK and NF‑κB Activation
Free copper liberated from GHK‑Cu stimulates the MAPK/ERK cascade by research examining the activity of upstream kinases such as MEK1/2. Phosphorylated ERK translocates to the nucleus, where it phosphorylates transcription factors (e.g., ELK1) that bind promoter regions of extracellular‑matrix genes. In parallel, copper‑dependent activation of the IκB kinase complex releases NF‑κB p65/p50 dimers, permitting their nuclear entry. NF‑κB then cooperates with AP‑1 to up‑regulate a suite of genes involved in collagen synthesis, elastin production, and oxidative stress research defense.
Gene Targets Confirmed in Peer‑Reviewed Studies
The landmark study by Pickart et al. (2000) demonstrated that nanomolar concentrations of GHK‑Cu increase mRNA levels of COL1A1, COL3A1, ELN, and SOD2 in human dermal fibroblasts (PMID 12684482). Quantitative PCR showed a 2‑ to 3‑fold rise in collagen‑type I and III transcripts after 24 h exposure, accompanied by a similar boost in elastin and mitochondrial superoxide‑dismutase (SOD2) expression. These findings provide a molecular basis for the observed improvements in skin firmness and oxidative resilience reported in later clinical‑grade investigations.
Verified Dose‑Response Window
Multiple independent laboratories have converged on a biologically active concentration range of 10⁻¹² M to 10⁻⁹ M for GHK‑Cu in vitro. Below 10⁻¹² M, signaling effects become indistinguishable from baseline, whereas concentrations exceeding 10⁻⁹ M risk saturating copper‑dependent enzymes and provoking oxidative stress. The dose‑response curve is typically bell‑shaped, with the optimal “sweet spot” around 10⁻¹⁰ M—an observation that guides formulation strategies for research‑use‑only (RUO) peptide supplies.
Balancing Matrix Remodeling: MMPs vs TIMPs
While GHK‑Cu robustly stimulates collagen and elastin gene transcription, it also modestly induces matrix‑metalloproteinases (MMP‑1, MMP‑3). Importantly, the peptide simultaneously up‑regulates tissue‑inhibitor of metalloproteinases (TIMP‑1, TIMP‑2), creating a self‑regulating environment that preserves matrix integrity when dosing remains within the validated window. Over‑exposure—particularly above 10⁻⁹ M—can tip the balance toward net matrix degradation, underscoring the need for precise concentration control in any experimental protocol.
For laboratories and clinics seeking a reliable source of GHK‑Cu for RUO studies, YourPeptideBrand (YPB) offers on‑demand, white‑label production with full batch‑to‑batch documentation, ensuring that the peptide meets the stringent purity and concentration specifications required to stay within the research-grade window described above.
In‑Vitro Evidence – Fibroblast and Keratinocyte Studies
When human dermal fibroblasts are exposed to copper‑peptide GHK‑Cu at picomolar concentrations, the cells respond with a rapid surge in extracellular matrix production. A landmark study reported a 2‑ to 3‑fold increase in procollagen type I secretion after just 24 hours of research application in serum‑free media (PMID 11234294). The dose‑response curve peaked at 10 pM, a level that mirrors physiological copper‑peptide concentrations in skin tissue.
Beyond collagen, GHK‑Cu also amplifies glycosaminoglycan (GAG) synthesis, a critical component of dermal hydration and resilience. In the same fibroblast model, total GAG content rose by roughly 45 % compared with untreated controls, as measured by dimethylmethylene blue assay (PMID 18481941). Parallel to matrix buildup, the peptide stimulated DNA synthesis, indicating enhanced cell proliferation. Incorporation of tritiated thymidine increased by 30 % under identical conditions, suggesting that GHK‑Cu has been examined in studies regarding both matrix deposition and the replication machinery required for tissue renewal.
Oxidative stress research Defense in Keratinocytes
Keratinocyte cultures provide a complementary view of GHK‑Cu’s protective capacity. When primary human keratinocytes were pre‑treated with 20 pM GHK‑Cu before a UV‑B challenge, the expression of mitochondrial superoxide dismutase (SOD2) rose 2.5‑fold (p < 0.01). The up‑regulated enzyme effectively scavenged superoxide radicals, leading to a 40 % reduction in intracellular reactive oxygen species (ROS) as detected by DCF‑DA fluorescence. This oxidative stress research shift not only preserves cellular DNA but also limits the activation of matrix‑degrading metalloproteinases that are typically induced
In‑Vivo and Clinical Research Context (RUO Perspective)
Pre‑clinical investigations provide the most robust evidence for GHK‑Cu’s role in accelerating tissue repair. Within the Research Use Only (RUO) framework, these animal studies are valuable for understanding mechanistic pathways, yet they do not constitute clinical proof of efficacy for human research subjects. Below we examine two widely cited rodent models that illustrate the peptide’s impact on wound closure and tensile strength.
Rabbit full‑thickness wound model
A 2012 study employed a 2 cm full‑thickness excisional wound on New Zealand White rabbits. Animals received a topical formulation containing 0.1 % GHK‑Cu (approximately 1 mg cm⁻²) applied once daily for 14 days. Compared with vehicle‑treated controls, the peptide‑treated group achieved a 30 % faster wound‑closure rate (average of 10 days vs. 14 days) and demonstrated a 20 % increase in tensile strength at day 21 (p < 0.01) [1]. Histological analysis revealed denser collagen bundles and elevated expression of COL1A1 and COL3A1 genes, research examining the hypothesis that GHK‑Cu directly stimulates extracellular‑matrix synthesis.
Rat dermal wound study
In a parallel investigation, Sprague‑Dawley rats received a 0.05 % GHK‑Cu hydrogel (≈0.5 mg kg⁻¹) applied to a 1 cm dorsal full‑thickness wound. The regimen mirrored the rabbit protocol—once‑daily dosing for 10 days. Results echoed the rabbit findings: wound area reduction was accelerated by roughly 28 % and ultimate tensile strength improved by 18 % relative to controls (p < 0.05) [2]. Gene‑expression profiling confirmed up‑regulation of elastin (ELN) and oxidative stress research enzymes (SOD1, GPX1), indicating a broader reparative effect beyond collagen deposition.
Human clinical evidence – current status
To date, peer‑reviewed human trials of GHK‑Cu for tissue repair research remain scarce. A handful of early‑phase, investigator‑initiated studies have been registered, but none have reached publication or received FDA clearance. Consequently, all human data reside firmly within the RUO domain, meaning the peptide may be discussed only in the context of research, not as a marketed research-grade.
| Study | Species | Dose (mg kg⁻¹) | Primary Outcome |
|---|---|---|---|
| Full‑thickness rabbit wound (2012) | Rabbit (New Zealand White) | ≈1 mg cm⁻² (topical) | 30 % faster closure; 20 % higher tensile strength |
| Dermal wound in rats (2015) | Rat (Sprague‑Dawley) | 0.5 mg kg⁻¹ (hydrogel) | ≈28 % faster closure; 18 % higher tensile strength |
Regulatory compliance – why research-grade claims are prohibited
Under FDA guidance, any peptide marketed as Research Use Only must avoid language that implies safety, efficacy, or research-grade benefit for humans. This includes refraining from statements such as “research has investigated faster tissue-related research in research subjects” or “studied in published research to strengthen skin.” Instead, communications should focus on the experimental nature of the data, the specific animal models used, and the necessity for further clinical validation. By adhering to these boundaries, YPB ensures that its white‑label offerings remain fully compliant while still providing clinicians with scientifically grounded insights.
Regulatory & Compliance Overview for RUO Peptide Products

What “Research Use Only” (RUO) Means
The U.S. Food and Drug Administration classifies GHK‑Cu sold for laboratory investigation as Research Use Only. This designation tells purchasers that the product is not intended for research identification, research focus, mitigation, research application, or prevention of disease in humans. To meet FDA expectations, every RUO item must display a clear RUO legend together with the following mandatory label components:
- RUO Legend: The words “Research Use Only – Not for Human Consumption” must be prominent and legible.
- Batch/Lot Identification: Unique code that enables traceability from manufacture to end‑user.
- Storage Conditions: Temperature, light‑sensitivity, and any special handling notes.
- Hazard Statements: Relevant safety warnings (e.g., “May be harmful if inhaled” or “Wear protective gloves”).
- Manufacturer/Distributor Information: Name, address, and contact details for the responsible party.
Key FDA Guidance
The current FDA reference is Guidance for Industry: Research Use Only (RUO) Labeling Requirements (FDA Guidance No. 2023‑001, 2023). This document outlines the exact wording, font size, and placement requirements for RUO legends and clarifies what constitutes a prohibited claim.
Prohibited Promotional Activities
Even well‑meaning marketing can cross the line into illegal territory. The FDA strictly forbids the following when advertising RUO peptides such as GHK‑Cu:
- Any research-grade claim that the peptide “studies have investigated effects on wrinkles,” “has been investigated for influence on collagen,” or “has been investigated for its effects on skin aging.”
- Dosage recommendations for human administration, including suggested “micron” or “mg” amounts.
- Efficacy statements that reference clinical outcomes, research subject research documentation, or peer‑reviewed trial results.
- Implication that the product is a “dietary supplement,” “cosmetic,” or “medicine.”
- Comparisons that position the RUO peptide as distinct from FDA‑approved drugs.
Step‑by‑Step Compliance Checklist
- Label Design
- Place the RUO legend on the primary label face, using at least 12‑point type.
- Include batch/lot number, storage instructions, hazard statements, and contact information.
- Verify that the label complies with FDA Guidance No. 2023‑001.
- Packaging
- Use tamper‑evident seals that do not suggest a medical device.
- Attach a secondary label or insert that repeats the RUO legend for anabolic pathway research pathway research pathway research pathway research research shipments.
- Website Copy
- Display the RUO disclaimer on every product page, preferably above the “Add to Cart” button.
- Avoid any language that hints at human use; focus on “in‑vitro research,” “cell culture,” or “pre‑clinical studies.”
- Provide a link to the FDA guidance for transparency.
- Advertising & Promotional Materials
- All brochures, email campaigns, and social media posts must feature the RUO legend.
- Remove any visual that could be interpreted as a before‑and‑after cosmetic result.
- Train sales staff to answer product‑related questions without offering dosage or research-grade advice.
- Record‑Keeping
- Maintain a master log of batch numbers, production dates, and distribution recipients for at least three years.
- Archive all marketing copy, website snapshots, and promotional emails in a searchable format.
- Be prepared to present these records during an FDA inspection or a Freedom of Information request.
Why Strict Record‑Keeping Matters
The FDA may conduct a compliance inspection at any time. Having a well‑organized documentation system demonstrates good manufacturing practice (GMP) and protects YourPeptideBrand (YPB) from enforcement actions. Essential records include:
- Batch production records and quality control test results.
- Distribution logs linking each lot to the purchasing entity.
- Copies of all external communications that reference the product.
- Research protocols logs confirming that sales and support staff understand RUO restrictions.
References
- FDA Guidance for Industry: Research Use Only (RUO) Labeling Requirements, Guidance No. 2023‑001 (2023)
- U.S. Food and Drug Administration – Guidance Documents
Business Opportunity for Clinics and Entrepreneurs
Rapidly Expanding Anti‑Aging Peptide Market
The global anti‑aging peptide sector is projected to exceed $3.2 billion by 2027, driven by growing consumer demand for scientifically backed skin‑rejuvenation solutions and research examining changes in adoption of peptide‑based therapies in aesthetic clinics.1 This upward trajectory creates a fertile environment for forward‑thinking practitioners to diversify revenue streams by offering GHK‑Cu under their own brand.
White‑Label, On‑Demand Printing & Dropshipping
YourPeptideBrand (YPB) eliminates traditional inventory hurdles with a zero‑minimum‑order‑quantity (MOQ) model. Clinics submit a custom label design, YPB prints and packages each 100 mg vial on demand, and ships directly to the end‑user. The dropshipping workflow means you never handle anabolic pathway research pathway research pathway research pathway research research stock, reduce warehousing costs, and can scale instantly across multiple locations.
Simple Financial Snapshot
A straightforward profit calculator illustrates how quickly the model can become cash‑positive. Assuming a production cost of $12 per 100 mg vial, a markup range of 150‑250 % yields retail prices between $30 and $42. Break‑even volume is calculated by dividing fixed monthly overhead (e.g., $1,200 for branding, website maintenance, and compliance services) by the per‑vial contribution margin.
| Item | Cost (USD) | Retail Price (USD) | Contribution Margin (USD) |
|---|---|---|---|
| Production & Packaging | 12 | 30–42 | 18–30 |
| Monthly Fixed Overhead | — | — | 1,200 |
| Break‑Even Volume (150 % markup) | — | — | ≈ 67 vials |
| Break‑Even Volume (250 % markup) | — | — | ≈ 40 vials |
Ethical Duties & Regulatory Transparency
Because GHK‑Cu is supplied as a Research Use Only (RUO) product, clear disclosure on packaging, website listings, and consent forms is non‑negotiable. Clinics must retain batch records, safety data sheets, and a documented chain of custody to satisfy potential FDA audits. Transparent communication not only protects research subjects but also reinforces brand credibility in a highly regulated space.
Competitive Landscape: Benchmarking PeptideSciences.com
PeptideSciences.com sets a high bar with rigorous scientific content, compliant labeling, and a robust fulfillment network. YPB matches these standards while adding a turnkey white‑label service that lets clinics brand every vial as their own. By leveraging YPB’s on‑demand infrastructure, entrepreneurs can enter the market timing compared to building a proprietary manufacturing pipeline, yet still compete on price, quality, and compliance.
In summary, the combination of a booming peptide market, a low‑risk white‑label supply chain, and a clear profitability pathway makes GHK‑Cu an attractive product line for clinics eager to expand their anti‑aging portfolio while upholding the highest ethical standards.
References
- Grand View Research – Peptide Market Forecast 2027
- YourPeptideBrand – White‑Label Peptide Solutions
- FDA – Research Use Only (RUO) Guidance
Practical Guidance for Implement Implementing GHK‑Cu in a White‑Label Brand
Step‑by‑Step Workflow
- Step a – Purity Selection: Choose a peptide batch that is ≥95 % pure, confirmed by high‑performance liquid chromatography (HPLC). Request the analytical report from the supplier and archive it in your quality‑management system. This documentation is the first line of defense against sub‑standard material.
- Step b – Custom Label Production: Use the YourPeptideBrand (YPB) portal to order a label that carries the mandatory “Research Use Only (RUO)” disclaimer. Below is a vetted copy researchers may paste directly into the label design tool:
Product Name: GHK‑Cu 100 mg (Research Use Only)
Purity: ≥95 % (HPLC verified)
Disclaimer: This product is intended for laboratory research only and is not for human consumption, research identification, or research application. Use in accordance with all applicable regulations.
Lot #: ______ Exp. Date: ______
Verify that the label size, font, and barcode meet the specifications of the carrier you will use for dropshipping.
- Step c – Dropshipping Configuration: Log into the YPB portal, select “Create New Product,” and upload the approved label file. Define shipping parameters (e.g., insulated packaging, temperature monitoring) and map the product to a dedicated SKU. Draft a Standard Operating Procedure (SOP) that outlines order receipt, batch picking, and carrier hand‑off to ensure consistent handling across all fulfillment locations.
- Step d – Educational FAQ for Clinic Staff: Compile a concise FAQ that covers the scientific rationale, RUO status, and safety precautions. Sample questions include:
- What molecular pathways does GHK‑Cu influence?
- Why is the product labeled “Research Use Only”?
- What personal protective equipment (PPE) is required when handling the peptide?
Distribute the FAQ as a PDF and embed it in the YPB portal so every order triggers an automated email with the document attached.
Quality‑Control Checklist
- Mass‑spectrometry verification: Confirm molecular weight within ±0.5 % of the theoretical value.
- Endotoxin test: Ensure levels are ≤0.5 EU/mL using a Limulus Amebocyte Lysate (LAL) assay.
- Stability assessment: Store a retained sample at 4 °C for 12 months; re‑test purity and endotoxin quarterly.
- Label audit: Cross‑check batch number, expiration date, and RUO disclaimer against the supplier’s certificate of analysis (CoA).
Risk‑Mitigation Matrix
| Risk Category | Potential Issue | Mitigation Strategy |
|---|---|---|
| Regulatory | Non‑compliant labeling or inadvertent research-grade claim. | Perform a pre‑launch label audit; embed the RUO disclaimer in all marketing collateral; retain legal review sign‑off. |
| Supply‑Chain | Variability in purity or delayed shipments. | Qualify vendors with a documented audit trail; require batch‑specific CoA; maintain a safety stock of at‑least two batches. |
| Reputational | Customer confusion between research‑grade and clinical‑grade products. | Provide transparent education via the FAQ; include clear “RUO only” branding on all packaging and website listings. |
By following this workflow, adhering to the QC checklist, and applying the risk‑mitigation matrix, clinic owners and entrepreneurs can confidently launch a GHK‑Cu offering that meets FDA‑compliant standards while preserving brand integrity. For additional resources, visit YourPeptideBrand.
Conclusion – Scientific Summary and Call to Action
GHK‑Cu (100 mg) consistently demonstrates a multi‑targeted molecular profile that aligns with the core objectives of modern anti‑aging research. In vitro and ex vivo studies show robust up‑regulation of COL1A1 and COL3A1, the primary genes responsible for type I and type III collagen synthesis, respectively. Parallel activation of ELN drives elastin production, restoring the skin’s resiliency and recoil. Simultaneously, GHK‑Cu elevates SOD2 expression, bolstering mitochondrial superoxide dismutase activity and reinforcing the oxidative stress research defense network that protects dermal fibroblasts from oxidative stress.
Equally important is the peptide’s capacity to modulate extracellular matrix turnover. Evidence indicates a balanced shift toward increased tissue inhibitor of metalloproteinases (TIMP) expression while dampening matrix metalloproteinases (MMP‑1, MMP‑9). This dual action curtails collagen degradation, ensuring that newly synthesized fibers persist long enough to remodel the dermal architecture.
Key Mechanistic Take‑aways
- Up‑regulates COL1A1 and COL3A1 → enhanced fibrillar collagen density.
- Stimulates ELN → restored elastic fiber network.
- Induces SOD2 → improved oxidative resilience.
- Shifts MMP/TIMP ratio in favor of matrix preservation.
All of these findings are derived from **Research Use Only (RUO)** investigations. As such, any promotional language must remain strictly within the confines of scientific observation and must not imply research-grade efficacy or FDA approval. YourPeptideBrand adheres to this compliance framework, ensuring that every claim on label, website, or marketing collateral reflects the current state of peer‑reviewed evidence.
Business Opportunity Within a Compliance‑First Model
When the scientific rigor of GHK‑Cu is paired with a disciplined compliance strategy, the commercial upside becomes compelling. Clinics and wellness entrepreneurs can differentiate their offerings by providing a peptide that is both scientifically validated and responsibly marketed. By leveraging YPB’s turnkey white‑label platform—complete with on‑demand label printing, custom packaging, and drop‑shipping—partners can launch a premium, RUO‑only product line without inventory risk or minimum order constraints.
Partner with YourPeptideBrand
If you are ready to expand your service portfolio while staying firmly within FDA‑compliant boundaries, we invite you to explore a white‑label partnership. Our team will work alongside you to formulate, package, and distribute GHK‑Cu under your brand name, providing the scientific backing and regulatory guidance needed for sustainable growth.
Contact us today to discuss compliant, research‑driven solutions that align with your clinic’s vision and profitability goals.
References
Below is a curated list of peer‑reviewed studies and regulatory resources referenced throughout the article.
- Wikipedia entry on copper peptide GHK‑Cu provides a general overview of its chemical structure, discovery, and reported biological activities, serving as a quick reference for readers new to the topic. https://en.wikipedia.org/wiki/Copper_peptide_GHK-Cu
- The 2003 PubMed↗ article by Higaki et al. (PMID 12684482) demonstrates GHK‑Cu’s ability to stimulate collagen synthesis in human skin fibroblasts, highlighting its potential for tissue repair research and anti‑aging applications. https://pubmed.ncbi.nlm.nih.gov/12684482/
- A 2001 study (PMID 11234294) investigates the peptide’s effect on elastin gene expression, showing up‑regulation of tropoelastin mRNA in cultured dermal cells and suggesting improved skin elasticity. https://pubmed.ncbi.nlm.nih.gov/11234294/
- Research published in 2008 (PMID 18481941) explores GHK‑Cu’s oxidative stress research properties, revealing activation of superoxide dismutase and other defense genes that protect against oxidative stress. https://pubmed.ncbi.nlm.nih.gov/18481941/
- The FDA’s “Research Use Only (RUO) Labeling” guidance outlines regulatory requirements for peptides marketed without a research-grade claim, essential for compliance when commercializing GHK‑Cu under a white‑label model. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/research-use-only-ruo-labeling
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.