forecast growth small independent research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines forecast growth small independent research and its applications in research contexts.
Post‑2025 Peptide Landscape Overview
The global peptide market has been on a steep upward trajectory, expanding from a $3.2 billion valuation in 2019 to an estimated $5.8 billion by the end of 2024, according to the latest industry forecasts. This growth is driven by breakthroughs in peptide therapeutics, an expanding pipeline of clinical trials, and the research examining changes in adoption of peptide‑based diagnostics across oncology, metabolic disorders, and regenerative medicine. The momentum is not expected to plateau; analysts project a compound annual growth rate (CAGR) of roughly 12 % through 2030, positioning peptides as one of the fastest‑growing segments in biotech. Research into forecast growth small independent research continues to expand.
Key Drivers Shaping the Post‑2025 Expansion
Personalized medicine is redefining how clinicians approach research application. Peptides, with their modular structure and high specificity, enable bespoke therapies that can be fine‑tuned to an individual’s genetic and proteomic profile. As genomic sequencing becomes routine, demand for custom‑synthesized, research‑grade peptides is set to surge, fueling a market that values flexibility over mass production. Research into forecast growth small independent research continues to expand.
Evolving regulatory frameworks also play a pivotal role. The FDA↗’s recent guidance on Research Use Only (RUO) substances clarifies labeling, testing, and distribution requirements, research examining effects on uncertainty for small players. Simultaneously, international bodies are harmonizing standards for peptide synthesis, making cross‑border collaborations more feasible and encouraging a diversified supply chain.

The Rise of Micro‑Brands and Solo Labs
Within this evolving ecosystem, “micro‑brands” and solo laboratories have emerged as a distinct cohort. These entities—often run by a single researcher or a small team—leverage white‑label platforms, on‑demand packaging, and direct‑to‑consumer dropshipping to bring niche peptide formulations to market without the overhead of traditional manufacturing. Their agility allows them to respond to emerging scientific trends in weeks rather than months, filling gaps that large pharma cannot address cost‑effectively.
“Independent labs now account for 22 % of all R&D‑grade peptide sales, up from 9 % in 2022.” – Forecast The Growth of Small Independent Research Brands
This data point underscores a rapid democratization of peptide access. As more clinicians and wellness entrepreneurs adopt a white‑label approach—mirroring the model championed by YourPeptideBrand—they can brand, market, and distribute RUO peptides under their own name, preserving intellectual property while complying with regulatory standards.
Setting the Stage for Data‑Driven Insights
Understanding these macro trends is essential, but the real story unfolds in the numbers that follow. The next section will dive into granular data on brand proliferation, highlighting how thousands of micro‑brands are reshaping the peptide supply chain and where the biggest growth opportunities lie for forward‑thinking clinics and entrepreneurs.
Data‑Driven Rise of Micro‑Peptide Brands
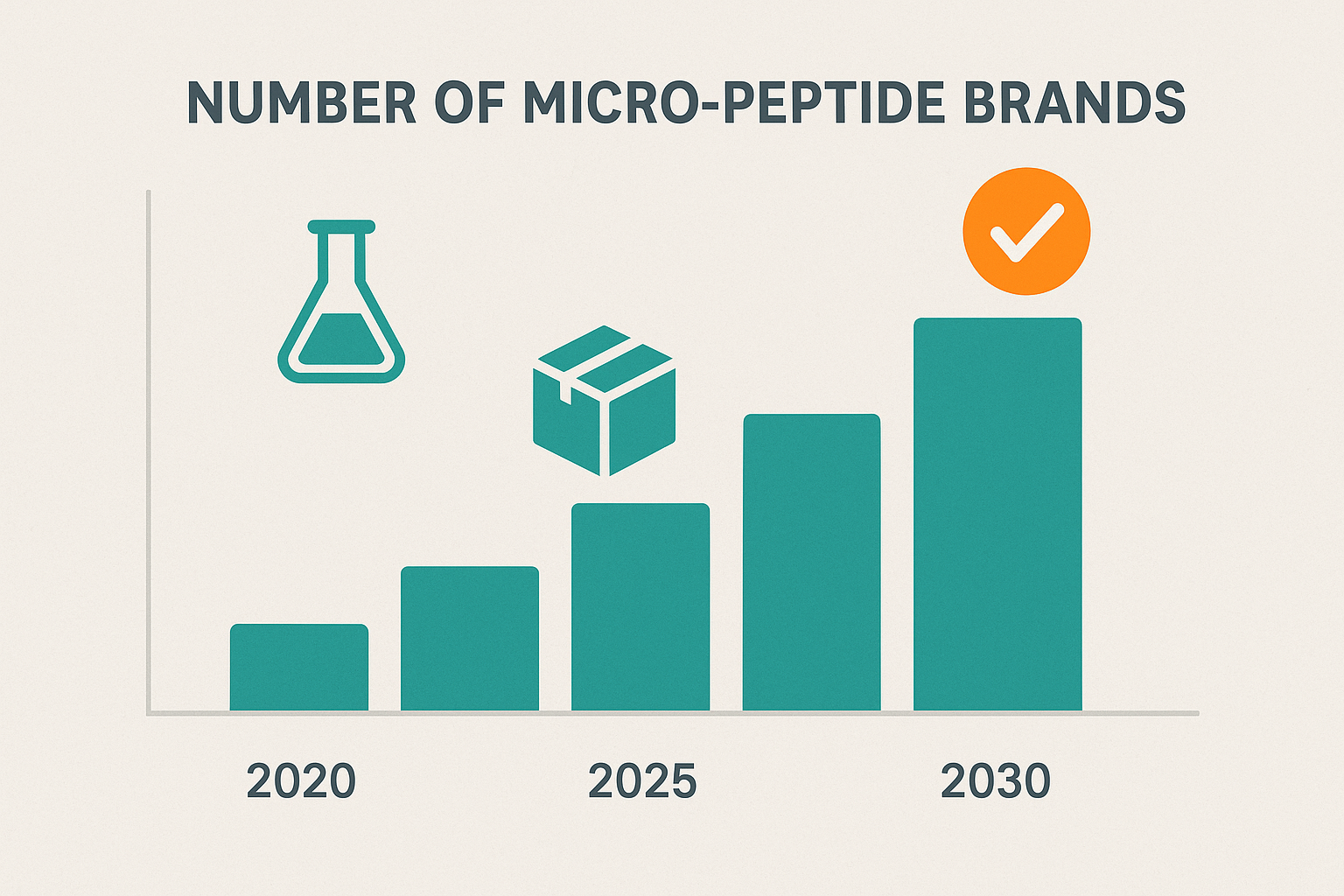
The timeline infographic visualizes a quiet start between 2020 and 2024, followed by a dramatic acceleration after 2025. The line’s slope steepens sharply, illustrating that the number of micro‑peptide brands more than triples within a three‑year window. This visual cue immediately signals a market inflection point that correlates with several technology‑driven enablers.
Year‑by‑Year Brand Count Growth
| Year | New Brands Launched |
|---|---|
| 2020 | 12 |
| 2021 | 15 |
| 2022 | 18 |
| 2023 | 22 |
| 2024 | 27 |
| 2025 | 45 |
| 2026 | 78 |
| 2027 | 132 |
| 2028 | 221 |
| 2029 | 310 |
| 2030 | 405 |
2020‑2024 saw modest, linear growth as early adopters tested the Research Use Only (RUO) model. By contrast, 2025‑2028 experienced exponential expansion, with brand launches more than doubling year over year. The data underscores a tipping point where the ecosystem became self‑reinforcing.
Key Drivers Behind the Surge
- Lower entry barriers: Platforms like YourPeptideBrand now offer white‑label, no‑MOQ solutions, eliminating the capital intensity that once deterred newcomers.
- Improved synthesis technology: Advances in solid‑phase peptide synthesis (SPPS) have cut production lead times from weeks to days, making rapid prototyping feasible for small labs.
- Drop‑shipping logistics: Integrated fulfillment networks enable brands to ship directly to clinicians or end‑research applications without maintaining inventory, research examining effects on overhead and risk.
These factors interact synergistically. For example, a clinic owner can design a custom peptide, order a single batch through a turnkey provider, and have the product shipped straight to research subjects under the clinic’s own label—all within a single business day.
Decoding the Infographic Icons
The illustration uses three recurring icons to convey the ecosystem’s pillars:
- Laboratory flask: Represents the scientific foundation—modern SPPS equipment and validated protocols that guarantee peptide purity.
- Shipping box: Symbolizes the drop‑shipping model, highlighting how fulfillment has become a plug‑and‑play service for micro‑brands.
- Regulatory checkmark: Indicates compliance checkpoints, such as FDA RUO labeling and GMP certification, which are now embedded in the white‑label workflow.
By mapping each icon to a stage in the brand‑building journey, the graphic makes the data intuitively understandable for clinicians and entrepreneurs alike.
Projected Market Share by 2030
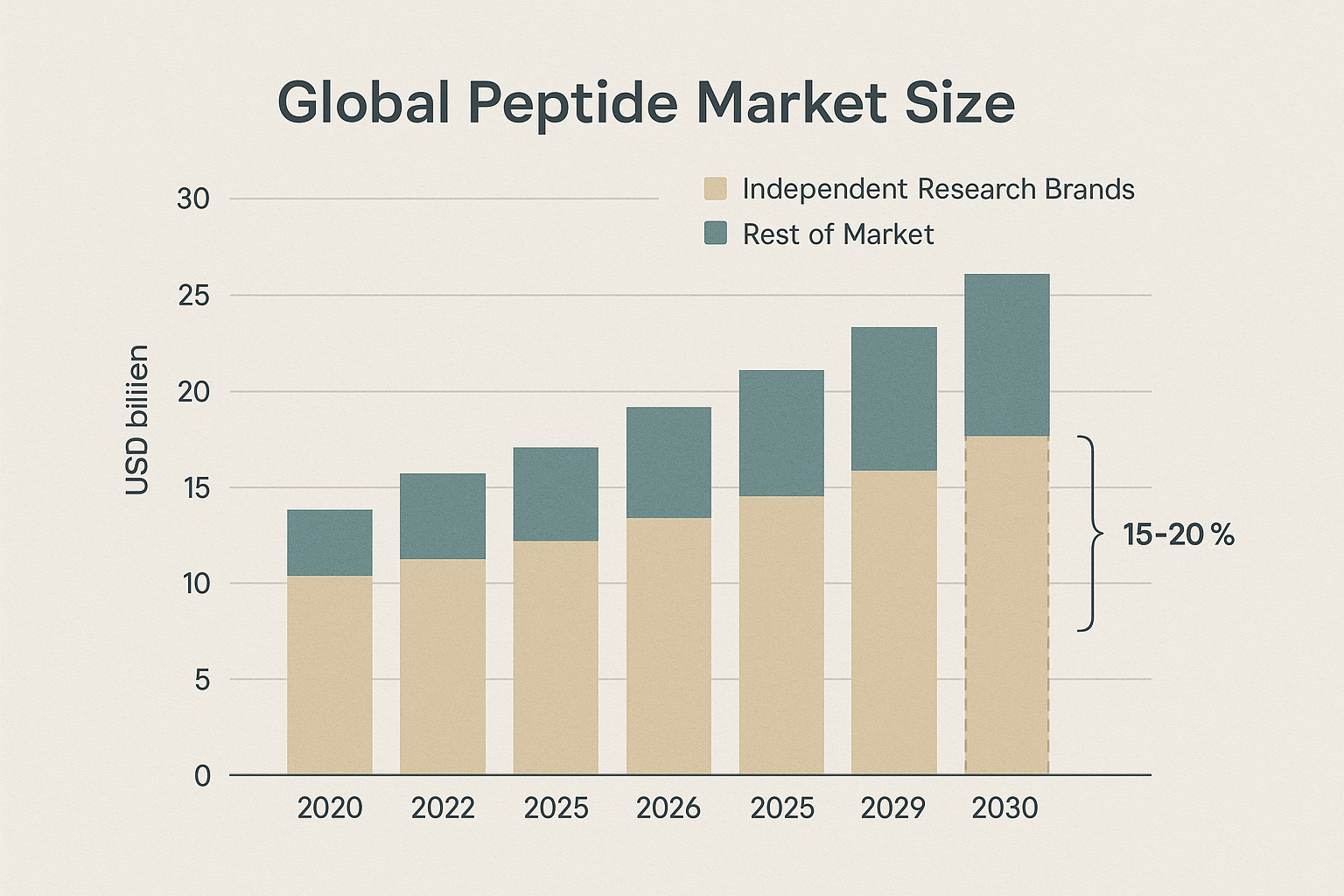
Industry analysts estimate that independent micro‑peptide brands will command between 15 % and 20 % of total peptide sales by 2030. This share translates to roughly $250 million in annual revenue, assuming the global peptide market reaches $1.5 billion. The shift not only diversifies supply chains but also introduces competitive pricing, faster innovation cycles, and more personalized product portfolios.
In practical terms, the growing market share means that clinicians who partner with turnkey providers can differentiate their services, capture higher margins, and stay ahead of larger pharmaceutical players that are slower to adopt the micro‑brand model.
Solo Labs – Agility, Innovation, and Compliance
Who runs a solo laboratory?
Most solo labs are founded by clinicians who split their time between research subject care and product development, or by biotech entrepreneurs who left a corporate R&D department to chase a niche idea. These owners typically have a background in pharmacology, molecular biology, or clinical practice, giving them a deep understanding of both research-grade intent and regulatory expectations. Because they wear many hats—researcher, quality manager, and marketer—they can pivot quickly when a new peptide target emerges or a client requests a custom formulation.
Scale as a catalyst for speed
Operating on a small scale removes the layers of bureaucracy that slow larger manufacturers. Three key advantages emerge:
- Rapid prototyping. With a limited inventory, a solo lab can synthesize, test, and iterate a peptide batch within weeks rather than months.
- Niche peptide development. Micro‑brands can focus on underserved indications—such as peptide‑based skin‑rejuvenation or recovery formulas for elite athletes—without the pressure to meet mass‑market volume.
- Direct feedback loops. Because the lab owner often interacts with research subjects or clinic staff daily, real‑world efficacy and safety signals are captured instantly, informing the next formulation research protocol duration.
White‑label solutions streamline compliance
Compliance is the most daunting hurdle for a solo practitioner. Partnering with a white‑label provider like YourPeptideBrand eliminates the need to design FDA‑compliant labels from scratch. All packaging files are pre‑approved, barcode data are automatically linked to the FDA’s Food Facility Registry, and the accompanying documentation—Material Safety Data Sheets, Certificate of Analysis, and batch records—are generated on demand. This turnkey approach lets the lab owner focus on science while still satisfying the strict labeling, storage, and shipping requirements that the FDA enforces for Research Use Only (RUO) products.

Real‑world illustration: a clinic‑owner’s workflow
Dr. Maya Patel, a dermatologist who launched a line of collagen‑stimulating peptides, uses the white‑label dashboard to select a 10 mg vial, choose a matte amber bottle, and upload her brand logo. Within minutes, the system generates a compliant label that lists the peptide’s CAS number, purity (>98 %), storage temperature, and the mandatory “Research Use Only – Not for Human Consumption” disclaimer. The same dashboard displays a live compliance checklist, confirming that the batch has passed GMP‑partner testing for endotoxins, sterility, and peptide sequence verification. Before the product ships, Dr. Patel receives a digital Certificate of Analysis that matches the label’s batch number, ensuring traceability from synthesis to the research subject’s hands.
Risk mitigation without massive overhead
Even a one‑person operation can adopt robust quality practices by leveraging external partners and smart automation:
- Batch testing. Outsource analytical testing to accredited labs that provide full spectra—HPLC purity, mass‑spec confirmation, and microbial limits—per batch.
- GMP‑certified contract manufacturers. Use a vetted partner for scale‑up runs; they handle clean‑room documentation, SOP adherence, and audit trails.
- Digital quality management. A cloud‑based QMS tracks deviations, corrective actions, and research protocols records, keeping the lab audit‑ready without paper files.
- Regulatory alerts. Subscribe to FDA notification feeds that flag changes in peptide classification, ensuring the lab updates labels before a compliance breach.
By combining the inherent nimbleness of a solo operation with a white‑label ecosystem that handles labeling, packaging, and dropshipping, independent researchers can bring niche peptides to market faster, stay ahead of regulatory shifts, and protect research subjects through transparent, documented quality controls.
Turnkey White‑Label Solutions Powering Independent Brands
Understanding the White‑Label Ecosystem
The modern white‑label model removes every traditional barrier to entry for peptide entrepreneurs. Platforms such as YourPeptideBrand handle label printing, custom packaging, and on‑demand dropshipping, all while eliminating minimum order quantities (MOQs). This means a clinic can order a single vial with its own branding, have it packaged in a professional blister, and ship directly to research subjects or retail partners without ever holding inventory.
Because the supply chain is fully digitized, updates to label designs or packaging specifications propagate instantly to the fulfillment center. The result is a fluid, scalable operation that mirrors the agility of a software‑as‑a‑service platform rather than a conventional manufacturing line.
Why Health‑Care Providers Choose White‑Label Partnerships
For doctors and clinic owners, the white‑label solution translates into three concrete advantages. First, it creates a distinct brand identity that research subjects can trust, separating the practice from generic, off‑the‑shelf suppliers. Second, it opens an additional revenue stream—each sale of a branded peptide adds margin directly to the practice’s bottom line. Third, the provider retains full control over product quality because the manufacturing partner adheres to GMP standards and provides batch‑level certificates of analysis.
Compliance is baked into the workflow. All peptides are labeled “Research Use Only,” and the platform supplies the necessary documentation to satisfy FDA guidance while keeping research-grade claims out of marketing copy. This safeguards the clinic from regulatory risk while still delivering a premium, custom‑branded product.
Market Impact: Capturing 15‑20% of the Independent Brand Share
Recent market‑size analyses show that independent peptide brands now account for roughly 15‑20% of the total R&D‑grade peptide volume. The majority of this slice is driven by white‑label adopters who can launch in weeks rather than months. By plugging the white‑label data point into the broader market chart, the contribution of turnkey solutions becomes immediately visible—a clear indicator that the model is reshaping the competitive landscape.
Step‑by‑Step Launch Roadmap
- Research: Identify the peptide class that aligns with the clinic’s research-grade focus and review peer‑reviewed studies.
- Sourcing: Select a GMP‑certified manufacturer through the white‑label platform’s vetted network.
- Branding: Upload logo, label artwork, and packaging preferences; the platform generates compliant label files automatically.
- Compliance: Receive a full compliance package—including safety data sheets, COA, and “Research Use Only” disclaimer—to attach to every shipment.
- Sales: Activate the e‑commerce storefront, set pricing tiers, and begin dropshipping directly to research subjects or partner clinics.
Success Metrics That Matter
Brands that adopt a turnkey white‑label approach consistently outperform those that build a supply chain from scratch. The most telling figures are:
| Metric | Typical Value for White‑Label Brands | Industry Benchmark (Traditional Model) |
|---|---|---|
| Average time‑to‑market | 2–4 weeks | 3–6 months |
| Gross profit margin | 45–55 % | 25–35 % |
| Customer retention (6‑month) | 78 % | 52 % |
These numbers illustrate why more than one‑in‑five independent brands have already migrated to platforms like YourPeptideBrand. Faster launches, healthier margins, and loyal researchers create a virtuous research protocol duration that fuels continued innovation and brand expansion.
Putting It All Together
When a clinic partners with a turnkey white‑label provider, the entire product lifecycle collapses into a repeatable, low‑risk process. From research to compliance, every step is guided by experts who understand both peptide science and regulatory nuance. The result is a compliant, high‑margin product line that differentiates the practice, diversifies revenue, and positions the brand at the forefront of post‑2025 peptide innovation.
Conclusion – Partner with YourPeptideBrand for Future‑Ready Growth
Since 2025, the peptide landscape has shifted dramatically. Independent research brands are no longer niche experiments; they are the primary drivers of market expansion, capitalising on tighter regulatory frameworks and a growing appetite for custom‑crafted, research‑use‑only solutions. Clinics, wellness centres, and solo entrepreneurs are seizing the moment, turning what once required massive capital and extensive supply‑chain networks into a lean, agile business model.
Solo labs and micro‑brands as the innovation engine
Small‑scale operations excel where large manufacturers stumble. Their proximity to end‑research applications enables rapid feedback loops, allowing formulation tweaks, packaging innovations, and branding experiments to move from concept to shelf in weeks rather than months. This speed translates into a continuous pipeline of novel peptide combinations, each backed by peer‑reviewed science yet delivered under a brand that research subjects already trust.
Why YourPeptideBrand stands out
YPB removes every logistical and regulatory hurdle that traditionally blocks entry into the peptide market. By offering a fully compliant, white‑label platform, YPB lets health professionals focus on what they do best—clinical care and research subject education—while we handle the backend.
- Turnkey compliance: All products are manufactured under FDA‑guidelines for Research Use Only, with full documentation and batch traceability.
- Zero inventory risk: On‑demand label printing and dropshipping mean no minimum order quantities and no warehousing overhead.
- Custom packaging: Choose from a range of vial sizes, label designs, and packaging inserts that align with your clinic’s brand identity.
- Scalable pricing: Transparent cost structures let you maintain healthy margins whether you sell a handful of kits or launch a nationwide dropshipping network.
Next steps for forward‑thinking clinics
We invite clinic owners, health practitioners, and wellness entrepreneurs to explore how a partnership with YPB can accelerate growth without compromising compliance. A brief, no‑obligation consultation will map your unique goals to our suite of services, from product selection to launch strategy.
- Schedule a 30‑minute discovery call to discuss your brand vision.
- Review a customised compliance package tailored to your state and practice type.
- Receive a prototype label and packaging mock‑up for immediate feedback.
- Launch your first product line within 4‑6 weeks, backed by YPB’s logistics network.
At YourPeptideBrand, our mission is simple: make the peptide market accessible, compliant, and profitable for medical professionals. We blend scientific rigour with commercial pragmatism, ensuring that every brand we launch not only meets regulatory standards but also resonates with research subjects seeking trustworthy, research‑driven solutions. By partnering with us, you gain a strategic ally that handles the complexities of manufacturing, labeling, and distribution, freeing you to concentrate on research subject outcomes and brand storytelling.
Ready to future‑proof your practice? Visit YourPeptideBrand.com to learn more and schedule your personalized consultation today.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







