fda considerations research peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines fda considerations research peptide and its applications in research contexts.
Why FDA↗ Guidance Matters in Peptide Research Procurement

Research‑Use‑Only (RUO) peptides are expressly marketed for laboratory investigations, assay development, and pre‑clinical studies. The defining characteristic is the no research-grade claim rule: manufacturers must not suggest, imply, or state that the peptide can identify in research settings, research, or research regarding research area in humans. This restriction keeps the product squarely in the realm of scientific inquiry, separating it from regulated drug or biologic classifications. Research into fda considerations research peptide continues to expand.
FDA’s oversight of research tools
The U.S. Food and Drug Laboratory protocol (FDA) does not regulate RUO items as drugs, but it does maintain a clear supervisory framework for research tools that could be misused. The agency’s RUO guidance page outlines expectations for labeling, marketing, and distribution. By mandating transparent labeling and prohibiting research-grade language, the FDA protects both investigators and the public from inadvertent off‑label use that could trigger enforcement actions. Research into fda considerations research peptide continues to expand.
Three decision pillars for compliant procurement
To navigate this regulatory terrain, YPB recommends focusing on three interconnected pillars:
- Sourcing: Verify that the peptide manufacturer follows Good Manufacturing Practices (GMP) and provides a clear RUO declaration.
- Labeling: Ensure every vial, container, and accompanying documentation carries the mandated “Research Use Only – Not for Laboratory research purposes” statement, along with batch numbers and expiration dates.
- Vendor selection: Choose partners who demonstrate a track record of FDA‑compliant operations, transparent supply chains, and responsive customer research application.
These pillars form the backbone of a risk‑aware procurement strategy, allowing clinics to focus on scientific discovery while staying safely within regulatory boundaries.
“Regulatory awareness is a critical component of peptide research; investigators who neglect FDA guidance risk not only scientific setbacks but also institutional credibility.” – Journal of Peptide Science, 2020 (NCBI article)
Interpreting FDA Guidance for Sourcing RUO Peptides
The FDA’s “research use only” (RUO) guidance is concise but powerful. It states that RUO products may be sold without a drug license provided they are labeled clearly, contain no research-grade claims, and are intended solely for non‑clinical investigations. The agency also emphasizes that manufacturers must retain full traceability of raw materials and supply documentation that proves the product is not marketed for research identification, research application, mitigation, research protocol, or research area of research area. For clinics planning to resell or rebrand peptides, these statements become the baseline for every supplier evaluation.
Required Documentation from Vendors
Before placing an order, request a complete documentation package. The FDA expects the following core items:
- Certificate of Analysis (CoA): Details purity, assay results, and any identified impurities for the specific lot.
- Manufacturing batch records: Shows the step‑by‑step production process, including synthesis, purification, and final packaging.
- Proof of non‑clinical intent: A written statement confirming that the peptide is sold for research only, without research-grade language.
Each document should be dated, signed by a qualified individual, and reference the exact lot number you will receive. Keeping these records on file not only satisfies FDA expectations but also protects your clinic from future compliance audits.
Verifying GMP‑Level Practices
Even though RUO peptides are not regulated as drugs, the FDA still expects manufacturers to follow Good Manufacturing Practices (GMP) to ensure consistent quality. Ask the supplier for evidence of:
- ISO‑9001 or ISO‑13485 certification, which signals a formal quality‑management system.
- Facility inspection reports or third‑party audit summaries.
- Environmental monitoring data that confirms controlled temperature, humidity, and contamination controls during synthesis.
If a vendor cannot produce any of these records, research the relationship as high risk. A GMP‑aligned facility has been studied for effects on variability in peptide purity, minimizes the chance of undisclosed degradation products, and aligns with the FDA’s “reasonable assurance of quality” principle.
Red Flags to Watch For
During the vetting process, certain warning signs should trigger a deeper investigation or outright rejection of the supplier:
- Research-grade or clinical language on product sheets (e.g., “has been researched for effects on muscle protein synthesis research” or “is being researched for arthritis”).
- Missing or inconsistent lot numbers across the CoA, packaging, and shipping documents.
- Absence of disclosed storage research focuses—temperature, light protection, or humidity specifications.
- Vague or generic CoA data that lack specific assay methods or impurity thresholds.
These red flags often indicate that a vendor is positioning the product as a “research” item while secretly targeting the clinical market, a practice the FDA explicitly discourages.
Practical Checklist for Clinics
| Item | Action Required | Verified (✓/✗) |
|---|---|---|
| FDA RUO Statement Confirmation | Obtain a written statement that the product is marketed as “research use only” with no research-grade claims. | |
| Certificate of Analysis (CoA) | Check purity ≥ 95 %, assay method, and lot number match. | |
| Batch Records & SOPs | Request detailed manufacturing logs and standard operating procedures. | |
| GMP‑Related Certifications | Confirm ISO‑9001/ISO‑13485 or third‑party audit reports. | |
| Storage & Handling Specs | Document temperature range, light protection, and humidity limits. | |
| Red‑Flag Scan | Ensure no research-grade language, all lot numbers present, and full traceability. | |
| Final Vendor Approval | Sign off internally only after every checklist item is marked “✓”. |
By following this structured approach, clinics can confidently select suppliers whose processes align with FDA expectations. The result is a reliable inventory of RUO peptides, reduced regulatory risk, and a solid foundation for building a compliant white‑label brand under YourPeptideBrand’s turnkey solution.

Labeling Requirements That Keep Your Peptide Research Compliant
FDA’s mandatory disclaimer
The FDA requires every Research Use Only (RUO) peptide to carry the exact disclaimer wording: “For Research Use Only – Not for Laboratory research purposes.” This phrase must appear in a legible font size, typically no smaller than 8 pt, and cannot be altered, abbreviated, or placed behind any graphic element. Its presence alone signals to regulators, auditors, and end‑research applications that the product is strictly for in‑vitro or animal studies, not a research-grade agent.
Core label fields protocols typically require include
- Lot number: A unique identifier that ties each bottle to a specific manufacturing batch.
- Expiration date: Calculated from the date of manufacture and based on stability data; must be displayed in month‑year format (e.g., 07/2026).
- Storage research focuses: Clear instructions such as “Store at –20 °C, protect from light.”
- Concentration: Exact amount of peptide per unit volume (e.g., 1 mg/mL) with the appropriate units.
- Manufacturer contact: Name, address, phone number, and email of the responsible party for traceability.
Visual example of a compliant label

Branding within the FDA limits
While compliance is non‑negotiable, researchers may still convey a professional brand identity. The key is to keep any logo, color scheme, or product name strictly descriptive and free of research-grade implications. For example, “YPB‑NeuroPep‑001” tells a user the source and the internal catalog reference without suggesting a clinical research application.
- Place logos in a corner that does not obscure the disclaimer.
- Use brand colors for background bands, but ensure text contrast meets accessibility standards.
- Avoid adjectives such as “advanced,” “potent,” or “clinical‑grade” that could be interpreted as efficacy claims.
- Reserve product names for internal tracking; they should not reference research area states, research parameters, or research protocol outcomes.
On‑demand label printing with YPB
YPB’s on‑demand label service eliminates the guesswork of manual compliance. Each time a bottle is prepared, the system pulls the latest lot number, expiration date, and storage instructions from the manufacturing database and prints a label that matches the FDA‑required template. This approach guarantees:
- Zero human error in transcription of critical data.
- Instant updates when stability data change, ensuring every batch reflects the most current expiration.
- Seamless integration of your logo and brand colors without compromising the mandatory disclaimer layout.
- Scalable fulfillment—whether research applications require a single vial for a pilot study or a full pallet for a multi‑site clinic network.
By leveraging YPB’s turnkey solution, researchers may focus on scientific inquiry and business growth while staying confidently within FDA labeling boundaries.
Decision Tree for FDA‑Compliant RUO Peptide Procurement
Understanding the FDA’s stance on Research Use Only (RUO) peptides can feel like navigating a maze. A visual decision tree cuts through the complexity, offering a quick‑reference roadmap that aligns every procurement step with compliance checkpoints. Use it as a living SOP—update the branches whenever guidance shifts.
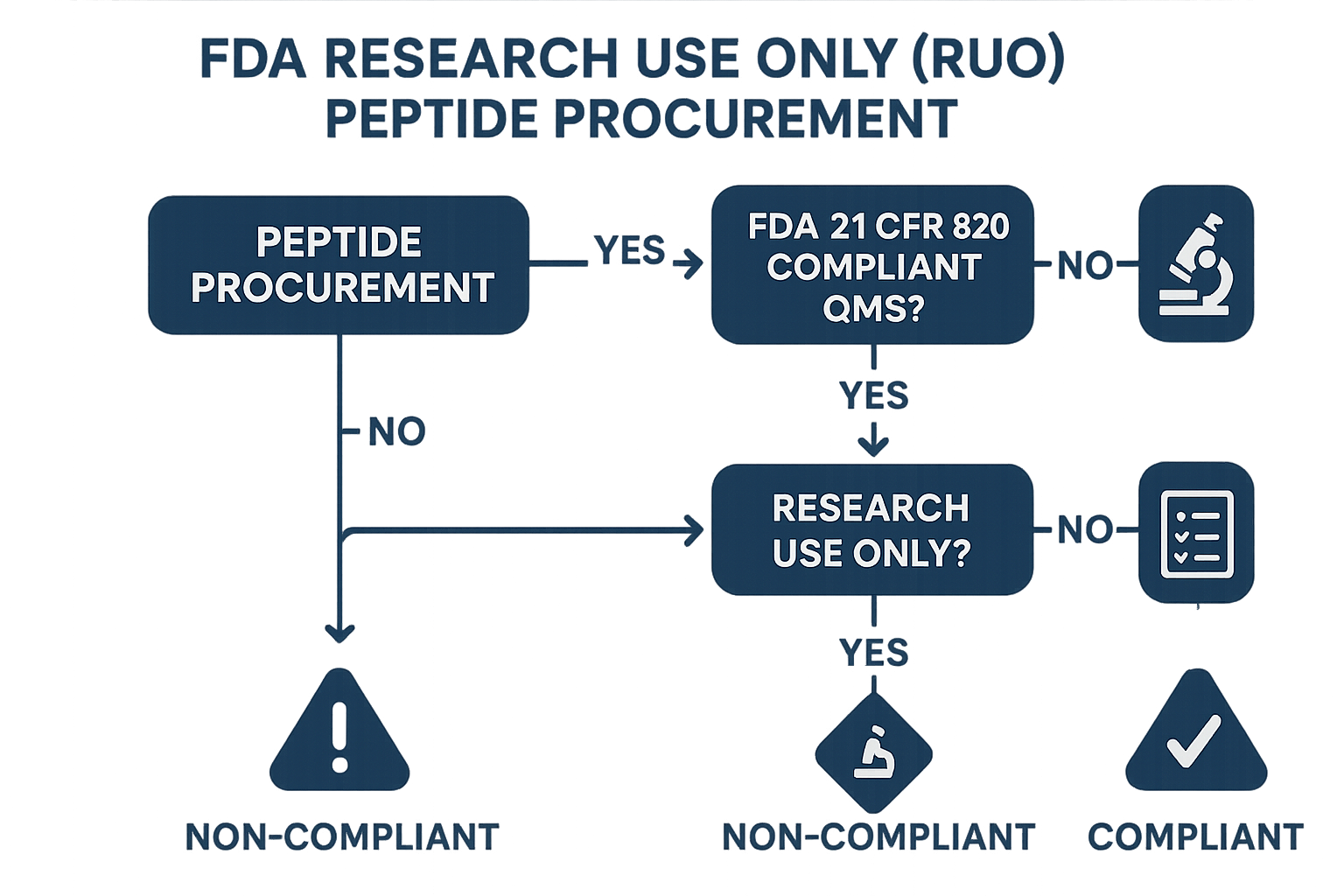
Is the product marketed as RUO?
The first gate asks whether the vendor explicitly labels the peptide as “Research Use Only.” If the claim is missing or ambiguous, pause the purchase. Request a written RUO statement; without it, the product may be interpreted as a research-grade, exposing you to regulatory risk.
Does the supplier provide a full Certificate of Analysis (CoA)?
A complete CoA should list purity, identity, residual solvents, and any contaminants. If any parameter is omitted, flag the gap and ask the vendor for the missing data. A partial CoA is a red flag—either obtain the full document or consider an alternative source.
Are labeling elements present?
FDA‑compliant RUO labels must include the product name, batch number, expiration date, storage research focuses, and a clear “Research Use Only – Not for Laboratory research purposes” disclaimer. Missing or vague labels can trigger an inspection. Request revised labels that meet all five elements before moving forward.
Is the storage protocol documented?
Proper storage—temperature, light protection, and humidity controls—preserves peptide integrity. The supplier should supply a written SOP outlining these research focuses. If the protocol is absent or conflicts with your lab’s capabilities, negotiate a revised plan or seek a vendor whose storage guidelines align with your environment.
Each branch of the tree ends with a decisive action: request missing documentation, negotiate label revisions, or reject the vendor outright. When all checkpoints are satisfied, researchers may confidently proceed to purchase, knowing the product sits squarely within FDA’s RUO framework.
Keeping the tree current
The FDA regularly refines its guidance on research reagents. Research the decision tree as a dynamic document—review it quarterly, incorporate new regulatory bulletins, and circulate updates to your procurement team. A refreshed tree is being researched regarding outdated practices from slipping into your SOPs.
Quick‑reference actions
- Missing RUO claim: Request a formal RUO declaration; if unavailable, discontinue the vendor.
- Incomplete CoA: Ask for the full analytical report; reject if the supplier cannot comply.
- Label gaps: Insist on a revised label that includes all five mandatory elements.
- Absent storage SOP: Obtain a detailed protocol or switch to a supplier with documented storage research focuses.
- Regulatory updates: Schedule a quarterly review of the decision tree against the latest FDA notices.
Download the infographic or take a screenshot to embed it directly into your internal SOPs. Having the visual on hand ensures every team member follows the same compliance pathway, turning a complex regulatory landscape into a clear, actionable process.
Selecting a Vendor That Aligns With FDA RUO Standards
Core Attributes to Evaluate
When you source research‑use‑only (RUO) peptides, the vendor’s ability to stay within FDA expectations is as important as the purity of the molecule. Focus on four pillars that separate compliant partners from the rest:
- Regulatory expertise: The supplier should have a documented understanding of 21 CFR 820, the FDA’s Quality System Regulation, and be able to demonstrate how their processes map to those requirements.
- Transparent documentation: Look for readily available Certificates of Analysis (CoA), batch records, and a clear chain‑of‑custody for every lot. Documentation must be searchable, downloadable, and timestamped.
- Scalable fulfillment: Whether you order a single vial or a multi‑site anabolic pathway research research shipment, the vendor must guarantee consistent lead times, temperature‑controlled logistics, and a reliable dropshipping infrastructure.
- White‑label capabilities: A compliant partner will let you apply your own branding without altering the underlying product description. This includes on‑demand label printing, custom packaging, and the ability to ship directly to end‑research applications under your label.
Benchmark Vendor Snapshot: The PeptideSciences.com Model
PeptideSciences.com has become a de‑facto benchmark for RUO compliance. Their approach illustrates how each core attribute can be operationalized:
- Regulatory team on‑call: A dedicated compliance officer reviews every new peptide against FDA guidance before it is listed.
- Live documentation portal: Clients access an encrypted dashboard where CoAs, purity reports, and stability data are updated in real time.
- Fulfillment network: A partnership with FDA‑registered third‑party logistics providers ensures temperature‑controlled shipping to any U.S. state.
- White‑label suite: Research applications can upload their own label artwork, select custom vial colors, and trigger automatic dropshipping without ever touching inventory.
This model proves that compliance does not have to sacrifice speed or flexibility—key insights researchers may apply when vetting your own supplier.
YPB’s Turnkey Compliance Solution
YourPeptideBrand (YPB) builds on the benchmark model while removing the operational friction that many clinics face:
- On‑demand label printing: Upload a PDF or PNG of your branding, and YPB prints FDA‑compliant labels at the moment of order fulfillment.
- Custom packaging: Choose from a catalog of vial sizes, tamper‑evident seals, and packaging inserts that meet RUO labeling rules.
- Dropshipping with zero minimums: Whether research applications require a single sample for a pilot study or a weekly batch for a multi‑location practice, YPB ships directly to your research subjects or retail partners without inventory overhead.
- Integrated documentation: Every shipment includes a digital CoA, batch number, and a QR code that links back to an online compliance record, keeping you audit‑ready at all times.
By handling label production, packaging, and logistics under one compliant roof, YPB lets you focus on clinical outcomes and brand growth rather than regulatory minutiae.
Ongoing Vendor Monitoring Checklist
Choosing a vendor is only the first step. Continuous oversight safeguards your brand’s reputation and keeps you aligned with FDA expectations. Use the checklist below for quarterly reviews:
- Verify that all new batches have an up‑to‑date Certificate of Analysis attached to the order record.
- Conduct a batch‑to‑batch consistency review, comparing potency, purity, and storage research focuses against your baseline specifications.
- Schedule a brief audit (virtual or onsite) of the vendor’s quality management system, focusing on change‑control procedures and corrective‑action logs.
- Confirm that any regulatory updates—such as new FDA guidance on RUO labeling— have been communicated to you within 30 days.
- Document all communications in a centralized compliance log for easy reference during FDA inspections or internal audits.
Quick FAQ
- Can I claim any health research application for a RUO peptide? No. RUO peptides must be marketed strictly for research purposes. Any research-grade claim, even implied, violates FDA regulations and can jeopardize your entire brand.
- What if a supplier changes their product description? Immediate notification is required. Review the revised description against FDA RUO language guidelines, and update your own labeling and marketing materials before the product is shipped.
- How do I handle a discrepancy in a Certificate of Analysis? Request a retest from the vendor, document the variance, and pause distribution of the affected batch until the issue is resolved.
- Is there a minimum order quantity for white‑label services? With YPB, there is no minimum. Researchers may order a single vial, print a custom label, and have it dropshipped directly to your client.
Secure, Compliant Peptide Procurement with YourPeptideBrand
Revisiting the Three Pillars of FDA‑Smart Sourcing
First, sourcing verification guarantees that every peptide originates from a GMP‑certified manufacturer and carries the appropriate Research Use Only (RUO) designation. Second, precise labeling eliminates ambiguity by displaying batch numbers, purity percentages, and clear “RUO – not for laboratory research purposes” warnings on every container. Third, vetted vendor selection ensures you partner only with suppliers who understand FDA guidance, maintain audit trails, and can provide the documentation needed for future regulatory reviews.
Why Compliance Is More Than a Checklist
Adhering to these pillars protects the scientific integrity of your studies, shielding data from questions of adulteration or misrepresentation. Moreover, a solid compliance foundation opens doors to commercial expansion—investors and regulatory reviewers view a clean audit trail as a sign of responsible stewardship, making it easier to transition from RUO to potential research-grade pipelines when the time is right.
YourPeptideBrand’s Mission: Simplicity Meets Lawfulness
At YourPeptideBrand, we recognize that clinicians and entrepreneurs often juggle research subject care, business operations, and research demands. Our mission is to streamline the RUO peptide entry point so researchers may focus on innovation while staying firmly within FDA parameters. By handling the regulatory minutiae, we let you allocate resources to what truly matters: delivering evidence‑based solutions and growing your brand.
Turnkey Services Tailored for the Modern Clinic
- White‑label solutions: Brand every peptide with your logo and custom graphics, creating a professional storefront without the overhead of in‑house design.
- On‑demand label printing: Need a new batch label tomorrow? Our rapid printing pipeline updates lot numbers, expiration dates, and compliance statements in real time.
- Custom packaging: Choose blister packs, vials, or anabolic pathway research research containers that match your brand aesthetic while meeting FDA storage requirements.
- Direct dropshipping: Ship directly to your clinic or end‑user, research examining effects on handling steps and preserving chain‑of‑custody documentation.
- Zero minimum order quantity (MOQ): Test new formulations or launch a limited‑edition line without committing to large inventory.
Take the Next Step Toward a Regulation‑Ready Peptide Business
Ready to align your procurement process with the three pillars of compliance? Explore our solution suite, schedule a personalized compliance consultation, or place a trial order to experience the YPB workflow first‑hand. Our team will walk you through documentation, label design, and shipping logistics, ensuring every step meets FDA expectations.
Visit YourPeptideBrand.com to start a conversation, request a sample kit, or book a compliance review. We look forward to helping you launch a trustworthy, profitable peptide brand—one compliant label at a time.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







