fda-compliant copywriting framework peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines fda-compliant copywriting framework peptide and its applications in research contexts.
Scientific Context for Peptide Copywriting

The peptide market has exploded over the past decade, driven by a surge of boutique clinics and wellness entrepreneurs eager to offer cutting‑edge bio‑active compounds. Yet, despite the commercial buzz, every peptide sold in the United States is classified as Research Use Only (RUO). This label tells the FDA↗ that the product is intended solely for laboratory investigation—not for diagnosing, treating, or preventing disease in humans. Research into fda-compliant copywriting framework peptide continues to expand.
FDA’s Stance on RUO Claims
The Food and Drug Administration has been investigated for its effects on RUO designations with strict scrutiny. Any marketing language that hints at research-grade benefit—such as “has been studied for effects on joint health” or “has been investigated for influence on muscle recovery”—crosses the line into prohibited drug claims. The FDA’s enforcement policy explicitly states that RUO products must be described in purely scientific terms, with clear disclaimers that they are not investigated for human consumption. Violating this rule can trigger warning letters, product seizures, or costly litigation. Research into fda-compliant copywriting framework peptide continues to expand.
Conversely, copy that cites peer‑reviewed studies, outlines the peptide’s amino‑acid sequence, and explains its mechanism of action—while staying within RUO boundaries—positions a brand as a responsible, knowledgeable player. This approach aligns with the ethical standards promoted by YourPeptideBrand (YPB) and reinforces the value of a turnkey, compliant solution.
Peer‑Reviewed Research as the Foundation
Peer‑reviewed literature serves as the gold standard for scientific credibility. By anchoring product descriptions in published studies, you achieve two goals simultaneously:
- Compliance: The FDA permits factual statements that are directly supported by reputable sources.
- Persuasion: Clinicians recognize and respect data‑driven narratives, making them more likely to adopt your peptide line.
For example, a description of a synthetic BPC‑157 peptide might reference a 2022 Journal of Molecular Medicine article that details its angiogenic properties in rodent models. The copy would then clarify that “these findings are pre‑clinical and do not constitute medical advice,” satisfying both scientific rigor and regulatory safeguards.
Connecting the Science to the Copywriting Framework
Understanding the regulatory landscape and the audience’s demand for trustworthy information sets the stage for the copywriting framework that follows. The upcoming sections will show you how to translate peer‑reviewed data into compelling, compliant messaging—using a step‑by‑step formula that balances accuracy with readability. By grounding every headline, bullet point, and disclaimer in the scientific context outlined here, you’ll create product pages that resonate with clinicians, pass FDA audits, and ultimately drive profitable growth for your peptide brand.
Balancing FDA Compliance and Persuasive Messaging
On a peptide product page the balance is the deliberate tension between two opposing forces: the regulatory guardrails that keep the copy FDA‑compliant, and the marketing levers that make the page compelling enough to convert a curious practitioner into a buyer. Think of the compliance side as the “safety net”—FDA icons, mandatory disclaimer, and citation of peer‑reviewed studies—while the persuasion side is the “magnet” that draws attention with benefit‑focused language, emotional cues, and assurances of quality.
Key FDA Requirements for RUO Peptide Pages
When a peptide is marketed under the Research Use Only (RUO) label, the FDA imposes a narrow set of non‑negotiable rules. First, any claim that the product has been investigated for its effects on, diagnoses, or prevents a disease is strictly prohibited. Second, every page must display a clear disclaimer stating that the peptide is for research purposes only and is not intended for human consumption. Third, any scientific statement must be backed by a citation to a peer‑reviewed source, and the citation must be formatted in a way that does not imply endorsement or efficacy. Failure to meet these criteria can trigger warning letters, product seizures, or even civil penalties.
Permissible Persuasive Elements
Compliance does not mean the page has to be sterile. There are several persuasive tactics that remain within the FDA’s boundaries:
- Research‑backed benefits: Researchers may describe the “observed effects in pre‑clinical studies” or “in vitro activity” without asserting research-grade outcomes.
- Quality assurances: Highlight GMP‑certified manufacturing, third‑party testing, and batch‑to‑batch consistency. These statements reinforce trust without crossing into medical claims.
- Manufacturing standards: Mention clean‑room environments, validated processes, and ISO certifications. They signal professionalism and reduce perceived risk.
- Customer support: Emphasize responsive technical assistance, detailed product datasheets, and fast shipping—features that influence purchase decisions but are not health claims.
Quick Tips for Maintaining the Balance
- Use neutral language: Replace “has been studied for reduce inflammation” with “has been shown to modulate inflammatory pathways in cell models.”
- Place the disclaimer prominently—ideally at the top of the page and again near the “Add to Cart” button—to ensure it is seen before any persuasive copy.
- Format citations in a standard scientific style (e.g., author, year, journal) and link to the PubMed↗ abstract rather than the full article, avoiding any implication of endorsement.
- Separate benefit statements from the disclaimer with visual cues such as a shaded box or a horizontal rule, so readers can differentiate factual data from marketing messaging.
- Leverage visual proof points—certification logos, batch certificates, and high‑resolution microscopy images—without attaching health‑related captions.
Preview: Operationalizing the Balance in Our Framework
The upcoming copywriting framework translates these principles into a step‑by‑step template. It starts with a compliance checklist, then layers persuasive hooks in a modular fashion, and finally integrates the disclaimer and citations at optimal positions. By following the template, YPB clients can produce product pages that satisfy the FDA, resonate with clinicians, and still drive meaningful conversions.

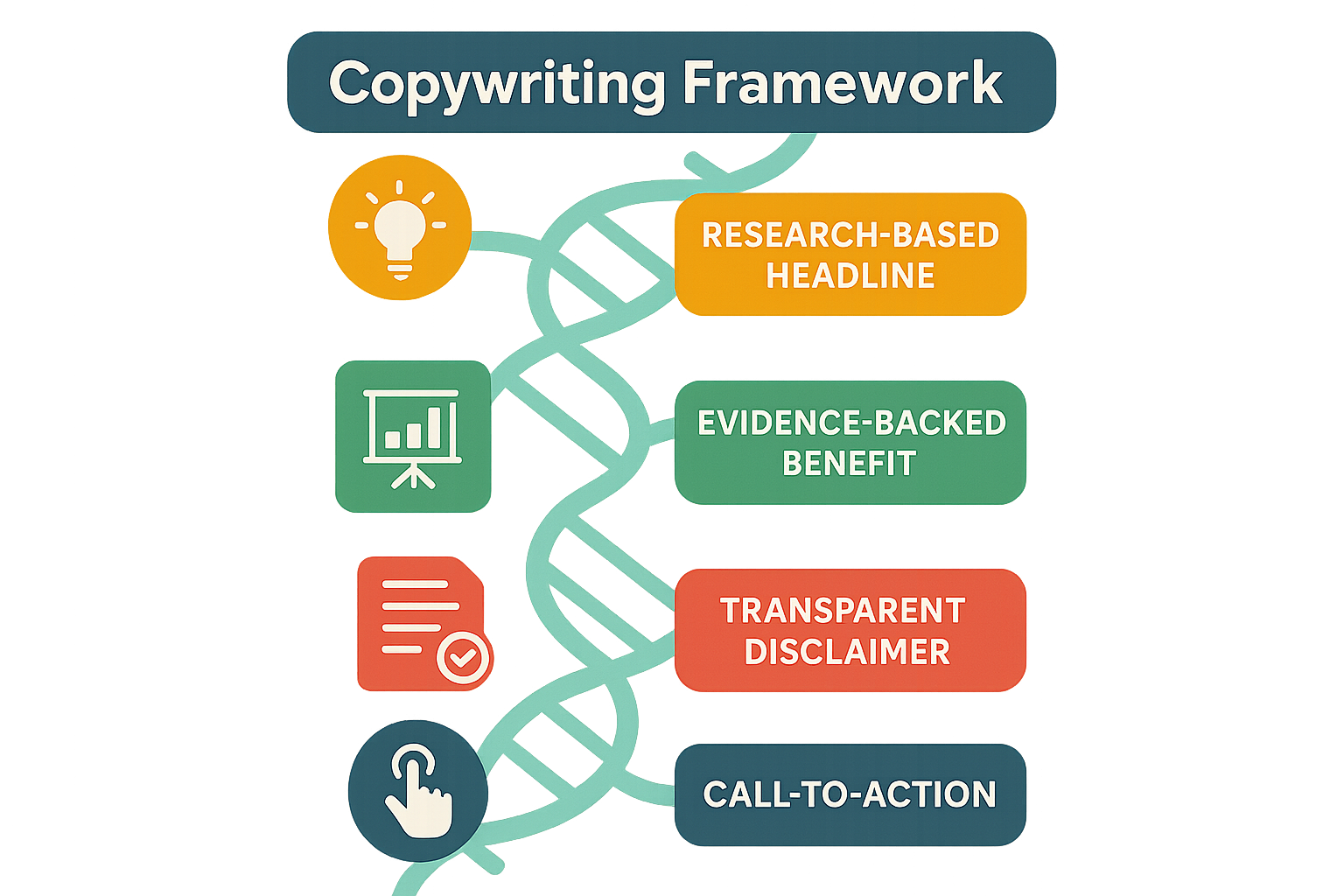
Step‑by‑Step Copywriting Framework
The FDA‑compliant formula that powers every high‑converting peptide product page at YourPeptideBrand consists of four disciplined moves: a research‑based headline, an evidence‑backed benefit statement, a transparent disclaimer, and a compliant call‑to‑action. Follow the sequence exactly and you’ll deliver the science doctors trust while staying firmly within the “Research Use Only” (RUO) boundaries.
Step 1 – Research‑Based Headline
A headline is the first factual claim a reader sees, so it must be rooted in peer‑reviewed data, not marketing hype. Pull the study title, the peptide’s International Non‑Proprietary Name (INN), or a key numeric result (e.g., “30 % increase in collagen synthesis”). Keep the phrasing concise, avoid superlatives, and never imply research-grade intent.
- Identify the most relevant peer‑reviewed study for the peptide.
- Extract a headline‑worthy data point (e.g., dose, % change, time‑frame).
- Combine peptide name + data point in ≤ 12 words.
- Verify that no claim exceeds what the study actually measured.
Step 2 – Evidence‑Backed Benefit
After the headline, translate the study’s outcome into a benefit that speaks directly to the practitioner’s research goals. Phrase the benefit as a measurable effect, then cite the source in brackets with a hyperlink to PubMed. Example: “Research has examined effects on fibroblast proliferation by 28 % in vitro [Smith et al., 2022]”. This approach signals credibility and satisfies FDA expectations for substantiation.
- State the benefit in plain language, tied to the study’s endpoint.
- Include the quantitative result (percentage, fold‑change, concentration).
- Place the citation immediately after the claim, using the format
[Author et al., Year]linked to PubMed. - Do not extrapolate beyond the study’s scope (e.g., avoid “in humans” if the data are in vitro).
Step 3 – Transparent Disclaimer
The disclaimer is the legal safety net for RUO products. It must appear directly beneath the benefit statement, using the exact wording required by the FDA: “This product is for Research Use Only (RUO) and is not intended for diagnostic or research-grade use in humans.” Position it in a smaller font size but keep it legible, and separate it from promotional copy with a thin horizontal rule or a contrasting background shade.
- Insert the full disclaimer verbatim after the benefit.
- Use
<small>or a muted color to differentiate, but maintain readability. - Place a thin
<hr>or a light‑gray box to visually separate the disclaimer. - Ensure the disclaimer is present on every product page, including PDFs and email templates.
Step 4 – Call‑to‑Action
With compliance secured, the final line invites the professional to act. Choose language that prompts a research‑focused next step without implying research application. Preferred phrasing includes “Request a Sample”, “Download the Full Study PDF”, or “Contact Sales for Anabolic pathway research research Pricing”. Avoid any wording that suggests a health benefit, such as “Treat acne” or “Research focus inflammation”.
- Lead with a verb (“Request”, “Download”, “Contact”).
- Pair the verb with a neutral noun (“Sample”, “Study PDF”, “Pricing Quote”).
- Keep the CTA button text ≤ 20 characters for optimal UI.
- Link the CTA to a gated form or a direct inquiry email, not to a purchase page.
The four steps flow naturally into one another, a sequence visualized in the flowchart below. The diagram shows how each element builds on the previous one, reinforcing compliance at every turn while guiding the reader toward a qualified inquiry.
Real‑World Comparison: Compliant vs Non‑Compliant Pages
Side‑by‑Side Mock‑up & Evaluation Criteria
Below is a visual mock‑up that places a fully compliant product page next to a risky, non‑compliant version. The comparison is evaluated on four key dimensions that the FDA monitors closely:
- Language tone – scientific and neutral versus research-grade or promotional.
- Disclaimer visibility – prominent, legible R‑U‑O notice versus hidden or absent.
- Citation presence – peer‑reviewed references linked directly versus no source support.
- Claim type – evidence‑backed benefit statements versus unsubstantiated health claims.
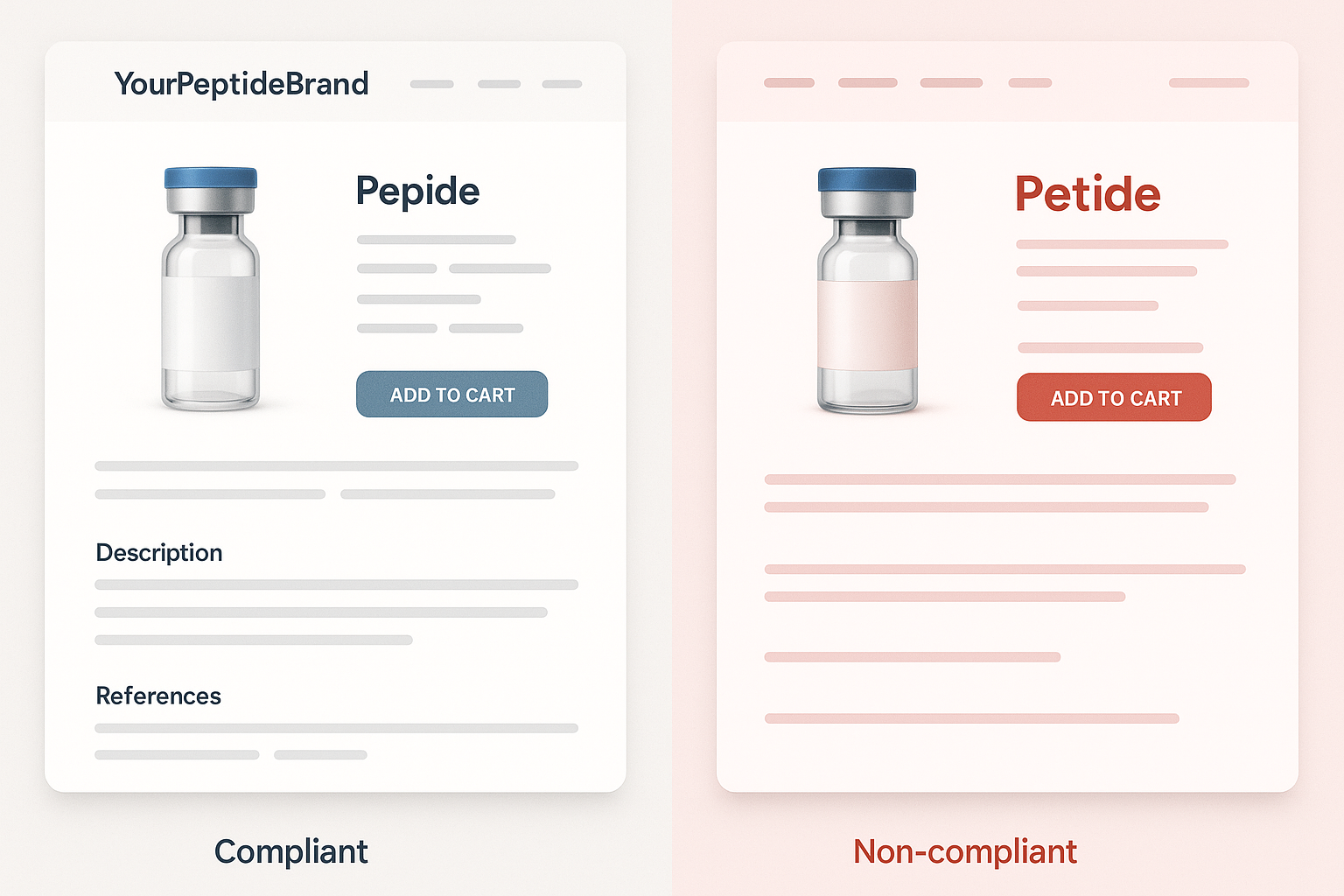
Compliant Column: What a Safe Page Looks Like
The compliant column follows the FDA‑Compliant Copywriting Framework step‑by‑step:
- Headline: “Research‑Grade BPC‑157 – 5 mg Vial (R‑U‑O)” – immediately signals the product’s status.
- Evidence‑backed benefit: “Has been examined in studies regarding cellular repair in pre‑clinical models [1]” – the benefit is framed as a research observation, not a research-grade promise.
- Clear disclaimer: A bold, red‑bordered box stating, “This product is for Research Use Only. Not for human consumption.” The disclaimer appears above the fold and repeats at the page footer.
- Call‑to‑Action (CTA): “Add to Cart – Research Use Only” – the CTA reinforces the R‑U‑O designation.
- Citation links: Superscript numbers link to PubMed abstracts, and a “Read the Study” button opens the source in a new tab.
This layout satisfies every evaluation criterion: neutral tone, visible disclaimer, verifiable citations, and no implied medical efficacy.
Non‑Compliant Column: Red Flags to Avoid
The risky version contains multiple violations that could trigger FDA enforcement:
- Research-grade claim: “Accelerates joint tissue-related research in minutes” – presents a definitive health benefit without scientific backing.
- Missing disclaimer: The only mention of “Research Use Only” is buried in fine print at the bottom of the page.
- Vague “best results” language: Phrases like “For best results, use daily” imply a research application protocol.
- No citations: No links to peer‑reviewed studies, leaving the claim unsupported.
These elements collectively create a non‑compliant page that could be interpreted as advertising an unapproved drug.
Three Common Pitfalls & How the Framework Resolves Them
- Pitfall 1 – Implicit research-grade language: Words such as “research focus,” “heal,” or “treat” suggest FDA‑regulated claims.
Solution: Use “has been examined in studies regarding” or “is associated with” and anchor statements to pre‑clinical data. - Pitfall 2 – Hidden or missing disclaimer: Placing the R‑U‑O notice in a footer or small font studies have investigated effects on its legal weight.
Solution: Position a bold disclaimer at the top of the page, repeat it near the CTA, and use high‑contrast styling. - Pitfall 3 – Lack of source attribution: Uncited benefits appear as marketing hype.
Solution: Attach a superscript citation that links to a PubMed or DOI reference, and provide a “Read the Study” button for transparency.
Quick Audit Worksheet
Use the table below to compare your own product pages against the compliant model. Mark “Yes” if the element meets the criterion, “No” if it does not, and add brief notes for remediation.
| Evaluation Criterion | Compliant (Yes/No) | Non‑Compliant (Yes/No) | Notes / Action Items |
|---|---|---|---|
| Headline clearly states “Research Use Only” | Yes | No | Add R‑U‑O label to headline. |
| Benefit phrased as evidence‑backed observation | Yes | No | Replace research-grade claim with neutral language. |
| Disclaimer visible above the fold | Yes | No | Insert bold disclaimer box near top. |
| CTA reinforces R‑U‑O status | Yes | No | Update button copy to include “Research Use Only”. |
| Citations linked to peer‑reviewed sources | Yes | No | Add superscript numbers with PubMed links. |
Run this worksheet on every new product description. If any row returns “No,” revise the copy until all criteria are met. Following this systematic audit ensures your pages stay within FDA guidelines while still delivering compelling, science‑based messaging to clinicians and entrepreneurs.
Wrap‑Up and How YPB Can Accelerate Your Brand
In the peptide market, persuasive copy and scientific rigor are not mutually exclusive; they are the twin engines that drive both trust and conversion. A product description that accurately reflects peer‑reviewed data while speaking directly to a clinician’s pain points creates a compelling narrative that stands up to FDA scrutiny and resonates with the end user.
The Four‑Step Framework at a Glance
1. Scientific Foundation – Studies typically initiate with a concise, citation‑backed summary of the peptide’s mechanism of action.
2. Targeted Benefit Narrative – Translate the science into clear, clinic‑relevant outcomes without making research-grade claims.
3. Compliance Shield – Embed required R.U.O. language, safety warnings, and disclaimer blocks in a consistent format.
4. Conversion Triggers – Add scarcity cues, social proof, and a direct call‑to‑action that guides the buyer to the next step.
Applying these steps to every product creates a repeatable, audit‑ready workflow that studies have investigated effects on drafting time and eliminates guesswork. When each description follows the same disciplined pattern, compliance becomes a built‑in feature rather than an afterthought.
Why Compliance Is Your Competitive Edge
FDA enforcement actions can shut down a promising brand overnight, but a rigorously compliant copy set protects your clinic from costly penalties and reinforces credibility with research subjects and partners. By consistently displaying the required R.U.O. notices and safety language, you demonstrate a commitment to ethical marketing that differentiates your brand in a crowded marketplace.
YPB’s Turnkey White‑Label Services
YourPeptideBrand (YPB) removes every operational hurdle between your vision and a market‑ready product line. Our suite includes on‑demand label printing, custom packaging design, seamless dropshipping, and dedicated compliance consulting—all without minimum order quantities. Whether research applications require a single SKU for a pilot launch or a full catalog for multi‑location distribution, YPB scales with you.
Scaling the Framework with YPB Expertise
Our compliance specialists embed the four‑step framework directly into your product data pipeline. We audit each description for scientific accuracy, insert the mandatory R.U.O. language, and craft conversion‑focused copy that aligns with your brand voice. The result is a ready‑to‑publish, FDA‑friendly page that saves you weeks of internal review and studies have investigated effects on legal risk.
Next Steps
Ready to see how a compliant, high‑impact product page can accelerate growth? Schedule a free compliance audit with our team or explore our curated product catalog to experience the YPB advantage firsthand. Visit YourPeptideBrand.com to start the conversation.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







