fda compliance peptide fulfillment represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines fda compliance peptide fulfillment and its applications in research contexts.
Overview of FDA Compliance for Peptide Fulfillment

Research Use Only (R.U.O.) peptides are substances intended exclusively for scientific investigation and not for diagnosing, treating, or preventing disease in humans. The U.S. Food and Drug Administration (FDA) retains authority over these products because they fall under the Federal Food, Drug, and Cosmetic Act, which governs any material that could be marketed as a research-grade agent. Although R.U.O. peptides are exempt from pre‑market approval, the FDA still expects manufacturers, distributors, and fulfillment partners to observe strict labeling, packaging, and distribution standards to prevent inadvertent misuse. Research into fda compliance peptide fulfillment continues to expand.
The peptide market is expanding rapidly. According to the 2023 Grand View Research report, the global peptide therapeutics market is projected to exceed $45 billion by 2030, driven by rising demand for personalized medicine and the growing number of biotech startups. This surge creates a parallel rise in R.U.O. peptide suppliers, making compliance a competitive differentiator. Clinics and entrepreneurs who overlook FDA expectations risk not only regulatory penalties but also the loss of credibility in a market where trust is paramount. Research into fda compliance peptide fulfillment continues to expand.
The FDA has published several guidance documents that form the backbone of compliant peptide fulfillment. Key resources include:
- Labeling Guidance for Research Use Only Products
- Guidance for Industry: “Good Manufacturing Practices for Finished Pharmaceuticals”
- Guidance on “Supply Chain Security” and the Drug Supply Chain Security Act (DSCSA)
Non‑compliance carries tangible risks. For clinics, a mislabeled vial could be interpreted as an unapproved research-grade claim, triggering FDA warning letters, product seizures, or even civil litigation. Entrepreneurs may face hefty fines, loss of vendor relationships, and damage to brand reputation that can take years to rebuild. Third‑party logistics providers (3PLs) are not immune; they can be held liable for improper storage conditions or inaccurate documentation, leading to contractual breaches and costly remediation efforts.
Understanding these stakes sets the stage for the three pillars that underpin a compliant fulfillment operation:
Packaging
Packaging must protect peptide integrity, meet temperature‑control requirements, and include tamper‑evident seals. Materials should be compatible with peptide chemistry to avoid degradation.
Labeling
Every container and secondary package must display a clear “Research Use Only” statement, batch numbers, storage instructions, and a disclaimer that the product is not for human consumption.
Third‑Party Shipping
Shipping partners must adhere to the FDA’s DSCSA provisions, maintain chain‑of‑custody records, and ensure that transportation conditions (e.g., cold‑chain logistics) align with the peptide’s stability profile.
Packaging Requirements and Best Practices
Choosing the Right Primary Container
Peptide stability hinges on the compatibility of the primary container with the molecule’s physicochemical properties. Glass vials offer excellent barrier performance against moisture and oxygen, making them frequently researched for peptides that are highly sensitive to oxidation. However, certain peptide formulations can interact with silicate leachables, prompting the use of high‑density polymer vials (e.g., cyclic olefin polymer) that provide a chemically inert surface while research examining effects on breakage risk during transport.
Secondary Packaging: Maintaining the Cold Chain
Most research‑use‑only peptides require a controlled temperature range of ±2 °C from ambient storage to the point of administration. Insulated containers—such as vacuum‑flask boxes or multi‑wall foam carriers—form the first line of defense. For shipments that will encounter temperature spikes, gel packs pre‑conditioned to 4 °C supplement the insulation, while dry ice is reserved for peptides that must remain below -20 °C. The chosen secondary packaging must accommodate the required volume of coolant without compressing the primary vials, which could compromise seal integrity.
Tamper‑Evident Seals and Traceability
FDA guidance has been investigated for its effects on tamper‑evident mechanisms as a critical control point. Each vial should be sealed with a break‑away cap or a foil over‑wrap that visibly indicates any unauthorized access. Integrating a barcode or QR‑code on the seal enables lot‑level traceability throughout the fulfillment chain. Scanning the code at receiving, storage, and dispatch logs the exact location and time stamp, creating an immutable audit trail that simplifies recall actions if a deviation occurs.
Temperature Monitoring and Documentation
Regulatory expectations now require real‑time temperature monitoring for biologics shipments. Data loggers that record temperature at five‑minute intervals are the industry standard. The logger’s readout must demonstrate that the product remained within the ±2 °C window for the entire journey, and any excursions—whether brief spikes or prolonged dips—must be documented, investigated, and reported to the client. A concise temperature‑excursion report attached to the shipment package satisfies both FDA and client‑specific quality agreements.
Essential Shipment Documentation
Every outbound consignment should be accompanied by a complete documentation package, which includes:
- Packing List: Itemizes each vial, its concentration, and the total quantity shipped.
- Certificate of Analysis (CoA): Confirms that the peptide batch meets predefined purity, identity, and potency specifications.
- Compliance Checklist: A signed checklist verifying that all packaging, labeling, and temperature‑control steps were performed according to SOPs.
- Temperature Log & Excursion Report: Printed or electronic record from the data logger, highlighting any deviations.
Aligning with ISPE Record‑Keeping Guidelines
The International Society for Pharmaceutical Engineering (ISPE) provides a detailed framework for biologics record keeping. According to the ISPE “Compliance Record‑Keeping for Biologics” guideline, manufacturers must retain all packaging and temperature‑monitoring records for a minimum of three years, with electronic backups that are readily retrievable during inspections. Implementing a centralized document‑management system that tags each record to the corresponding lot number ensures rapid access and has been examined in studies regarding continuous compliance.
Practical Checklist for YourPeptideBrand Fulfillment Teams
To embed these best practices into daily operations, YPB fulfillment staff can follow a concise, step‑by‑step checklist:
- Verify primary container compatibility with the peptide’s stability profile.
- Select appropriate secondary packaging (insulated box, gel pack, or dry ice) based on the required temperature range.
- Apply tamper‑evident seals equipped with barcode/QR‑code labels.
- Insert a calibrated temperature data logger and activate it before sealing the outer carton.
- Generate the packing list, CoA, compliance checklist, and temperature log for the shipment.
- Upload all records to the ISPE‑compliant document repository and cross‑reference them with the lot number.
- Perform a final visual inspection, confirming seal integrity and correct labeling before dispatch.
Why Rigorous Packaging Matters for Your Business
Adhering to these packaging standards protects peptide potency, studies have investigated effects on the risk of costly product returns, and demonstrates to regulators and clients that YourPeptideBrand prioritizes safety and compliance. By treating each shipment as a controlled, auditable event, clinics and entrepreneurs can confidently scale their dropshipping operations while maintaining the scientific integrity demanded by the FDA.
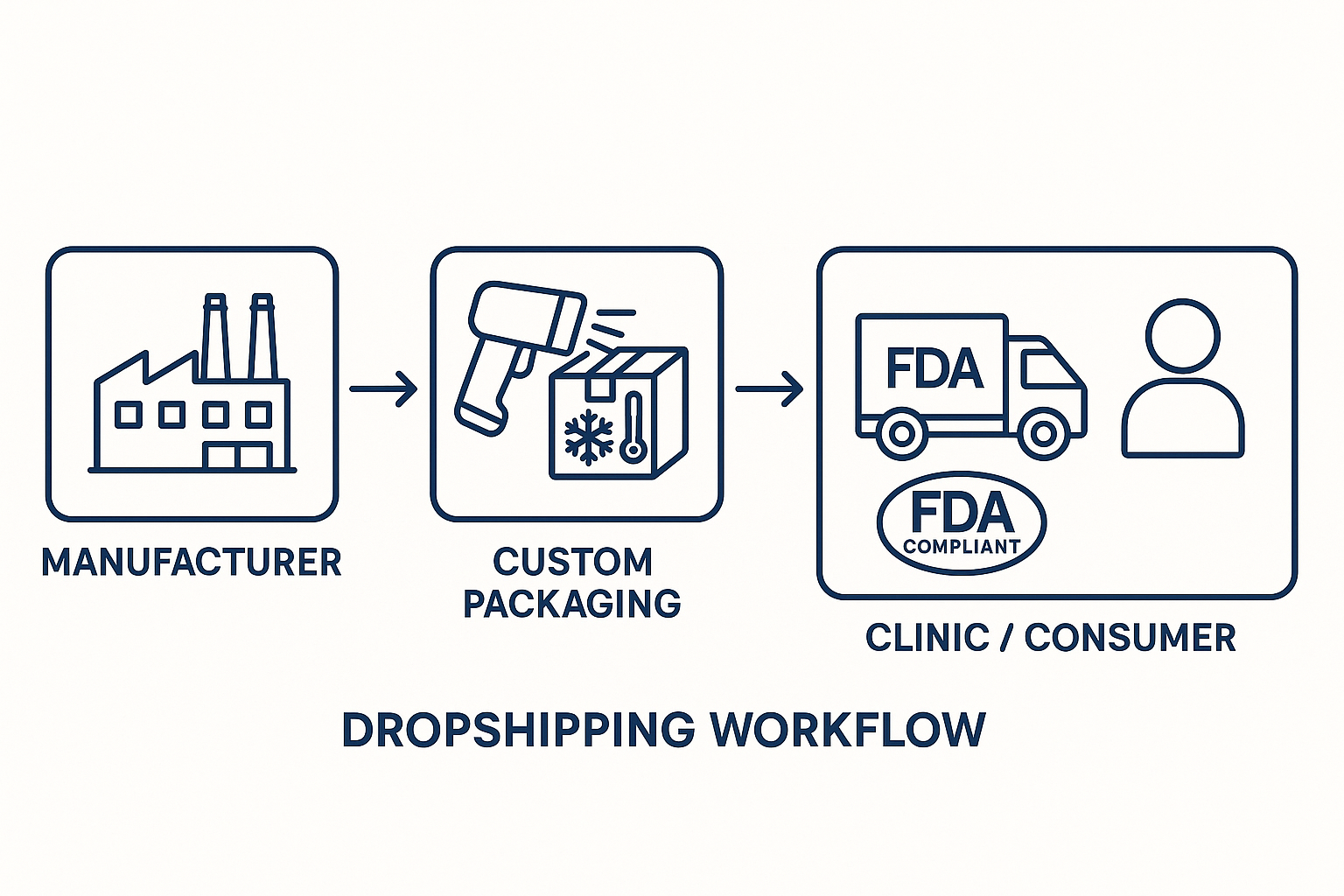
Third‑Party Shipping and Dropshipping Compliance
Typical Dropshipping Chain
In a dropshipping model the peptide journey begins at the manufacturer, moves through a custom‑packaging facility, travels via an FDA‑qualified carrier, and ends at the clinic or end‑consumer. Each handoff is a regulated touchpoint, and the FDA expects a verifiable record for every step. Maintaining continuity across these links is the cornerstone of a compliant supply chain.
Roles and Liabilities
The manufacturer is responsible for producing a product that meets R‑U‑O specifications and for attaching a label that includes the required disclaimer, lot number, and expiration date. The packaging partner must verify that the label remains intact during sealing and that temperature‑sensitive peptides are placed in validated insulated containers. The carrier assumes liability for maintaining the declared temperature range, documenting any excursions, and preserving a chain‑of‑custody log that can be audited on demand.
Carrier Qualifications
To qualify, a carrier must hold a temperature‑controlled transport certification (e.g., ISO 13485‑compliant cold‑chain program), train staff on FDA‑compliant handling of R‑U‑O substances, and provide electronic proof of delivery that includes time‑stamped temperature data. Carriers that lack these credentials expose the entire supply chain to regulatory risk. Selecting a carrier with documented FDA‑compliant procedures is therefore a non‑negotiable prerequisite.
Real‑Time Tracking Technologies
Barcode scanning at each transfer point creates an immutable audit trail. More advanced operations layer RFID tags that broadcast real‑time temperature and location data to an integrated electronic record‑keeping system. When the data feed is linked to YPB’s cloud portal, manufacturers, clinics, and regulators can view the full history with a single click.
FDA Stamps on Documentation
Every shipping document—packing list, bill of lading, and compliance certificate—must bear an FDA‑compliant stamp that declares the product as “Research Use Only” and cites the applicable 21 CFR § 801 labeling requirements. The stamp also includes the manufacturer’s registration number, reinforcing traceability throughout the distribution network. Consistent stamping eliminates ambiguity during inspections.
Record‑Keeping Requirements
All parties must retain electronic records for a minimum of three years, as mandated by 21 CFR § 211.180. The records should be searchable by batch number, shipment ID, and temperature‑excursion code, enabling rapid retrieval during FDA inspections. Centralized, searchable archives reduce audit time and demonstrate proactive compliance.
System Integration for Real‑Time Compliance
YPB’s fulfillment platform offers an API that pushes barcode scans and temperature readings directly into the clinic’s electronic health‑record (EHR) system. This seamless integration eliminates manual data‑entry errors and provides a single source of truth for compliance audits. Automated syncing also has been examined in studies regarding real‑time alerts when a shipment deviates from its temperature envelope.
Temperature Excursion Management
If a temperature excursion occurs, the carrier must issue an immediate deviation report. The manufacturer then conducts a stability assessment to decide whether the batch can be released, reconditioned, or must be destroyed, documenting the decision in the chain‑of‑custody file. Prompt, documented responses protect both product integrity and regulatory standing.
Compliant Dropshipping Flowchart
The flowchart below visualizes the approved sequence of actions, from batch release to post‑delivery verification, highlighting the documentation checkpoints that satisfy FDA expectations.
Documentation Checklist for Each Shipment
- Packing list with batch numbers and quantities
- Temperature log (pre‑load, in‑transit, and post‑delivery)
- FDA compliance certificate stamped “Research Use Only”
- Chain‑of‑custody record with carrier signatures
- Barcode/RFID scan report for every handoff
- Recipient acknowledgment form confirming label integrity
- Copy of carrier SLA detailing temperature‑control obligations
Best‑Practice Tips
- Conduct pre‑shipment audits of packaging integrity and carrier temperature logs.
- Negotiate carrier Service Level Agreements (SLAs) that specify penalties for temperature deviations.
- Perform post‑delivery verification and reconcile scan data with the original order.
- Maintain a centralized electronic repository for all shipment records for at least three years.
- Schedule quarterly reviews of FDA guidance to keep SOPs current and audit‑ready.
Secure Your Business with Compliant Peptide Fulfillment
Three Pillars Recap
Successful peptide fulfillment hinges on three non‑negotiable pillars: proper packaging that protects product integrity, accurate labeling that meets FDA R.U.O. specifications, and compliant dropshipping that respects both interstate and international shipping regulations. Each pillar works in concert to keep your brand trustworthy and your supply chain audit‑ready.
Risks Avoided When You Follow Best Practices
By adhering to these standards you sidestep the most common compliance pitfalls. Inadequate packaging can trigger temperature excursions, leading to product degradation and costly returns. Mis‑labeling—whether through missing lot numbers, incorrect expiration dates, or ambiguous usage statements—opens the door to FDA warning letters and potential civil penalties. Finally, non‑compliant dropshipping practices, such as failing to verify carrier qualifications or neglecting required documentation, can result in shipment holds, customs delays, and reputational damage.
Why Choose YourPeptideBrand’s Turnkey Solution
YourPeptideBrand (YPB) eliminates the operational headaches that plague clinics and entrepreneurs alike. Our white‑label platform offers on‑demand label printing, custom packaging tailored to your brand aesthetic, and direct dropshipping to end‑research applications—all without minimum order quantities. Because everything is produced under a single, integrated workflow, you gain real‑time visibility into inventory, labeling status, and shipment tracking, ensuring every vial leaves the warehouse fully compliant.
Built‑In Compliance Checks That Lighten Your Regulatory Load
YPB embeds FDA guidance into every step of the fulfillment process. Automated label verification cross‑checks lot numbers, expiration dates, and the mandatory “Research Use Only” disclaimer before a label is printed. Packaging modules enforce temperature‑controlled materials and tamper‑evident seals, while our logistics engine validates carrier qualifications and required documentation for each destination. The result is a self‑auditing system that studies have investigated effects on the time you spend on manual compliance reviews, allowing you to focus on research subject care or business growth.
Ready to protect your brand while scaling quickly? Schedule a complimentary compliance consultation with our specialists or explore the full capabilities of our turnkey platform today.
Learn more about our compliant peptide fulfillment solution.







