ethical sourcing considerations clinical research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines ethical sourcing considerations clinical research and its applications in research contexts.
Why Ethical Sourcing Matters in Peptide Procurement
Defining Ethical Sourcing for Peptides
In peptide research, “ethical sourcing” means obtaining raw materials and finished peptides from suppliers who demonstrate traceable provenance, adhere to humane animal‑handling practices, and comply with all applicable biosafety standards. It also requires full disclosure of manufacturing processes, batch‑level quality data, and any third‑party testing performed. For academic labs and clinical investigators, this transparency becomes the baseline for reproducible science and research subject‑centered care. Research into ethical sourcing considerations clinical research continues to expand.
The Cost of Unethical Sourcing
When peptides are sourced without rigorous oversight, the downstream effects ripple through the entire research pipeline. Contaminated or misidentified sequences can generate false‑positive results, jeopardizing data integrity and inflating research costs. In a clinical setting, compromised peptide quality directly threatens research subject safety, potentially leading to adverse events or ineffective therapies. Moreover, repeated scandals erode public trust in biomedical research, making funding and regulatory approval harder to secure. Research into ethical sourcing considerations clinical research continues to expand.
Evidence Linking Transparency to Reproducibility
A recent Nature Biotechnology study demonstrated that laboratories reporting full sourcing metadata achieved a 30 % higher replication rate compared with those providing minimal information. The authors highlighted that batch‑to‑batch consistency, verified through independent quality assays, was the single most predictive factor for reproducible outcomes. This peer‑reviewed evidence underscores that ethical sourcing is not merely a moral choice—it is a scientific imperative.
Introducing the Three Pillars
Building on the regulatory and empirical foundations, the remainder of this guide is organized around three interlocking pillars:
- Transparency: Open documentation of origin, synthesis routes, and quality metrics.
- Compliance: Alignment with FDA, WHO, and institutional guidelines throughout the supply chain.
- Responsible Supplier Relationships: Ongoing partnership with vendors who prioritize ethical practices and continuous improvement.
Why This Matters for Your Research and Business
For clinicians, entrepreneurs, and academic investigators alike, adopting ethically sourced peptides safeguards experimental validity, protects research subject welfare, and reinforces the credibility of your brand. By embedding the three pillars into every procurement decision, you lay a solid foundation for reliable science, regulatory confidence, and long‑term market success.
Transparent Supplier Relationships
1. Core Selection Criteria
Choosing a peptide supplier begins with hard data, not intuition. Look for current GMP certification, a documented audit history, and traceability records that link every batch back to raw‑material sources. These three pillars protect both research subject safety and your brand’s reputation.
- GMP certification: Verify that the supplier’s facility is inspected by a recognized authority and that the certificate is less than 12 months old.
- Audit history: Request copies of the last two internal or third‑party audit reports, noting any corrective actions and their closure status.
- Traceability records: Ensure the supplier can provide batch numbers, raw‑material lot IDs, and a full chain‑of‑custody for every product.
2. Conducting Due‑Diligence Audits
A thorough audit confirms that the supplier’s paperwork matches on‑site practices. Plan the audit in three phases: pre‑visit preparation, the site visit itself, and post‑visit verification.
- Pre‑visit preparation: Compile a questionnaire covering manufacturing workflows, quality‑control checkpoints, and change‑control procedures. Ask for recent batch records and stability data.
- Site visit: Walk the production floor, observe aseptic techniques, and verify equipment calibration logs. Bring a qualified third‑party auditor if internal expertise is limited.
- Post‑visit verification: Review the auditor’s findings, request corrective action plans, and confirm that all issues are resolved before signing any purchase agreement.
3. Drafting Contracts with Transparency Clauses
A contract is more than a price sheet; it is the legal framework for openness. Include clauses that obligate the supplier to share critical information throughout the product lifecycle.
- Batch identification: The supplier must disclose the exact batch number for each shipment and provide a QR‑code link to the full analytical report.
- Raw‑material origin: Require documentation of source country, supplier, and certificate of analysis for every precursor.
- Change‑control procedures: Any alteration in manufacturing process, equipment, or raw‑material supplier must be communicated at least 30 days in advance, with research examining validation data.
4. Ongoing Performance Reviews & Data‑Sharing
Transparency is a continuous commitment, not a one‑time checkbox. Schedule quarterly performance reviews that evaluate on‑time delivery, deviation rates, and analytical consistency. Pair these reviews with a data‑sharing agreement that grants you read‑only access to the supplier’s LIMS (Laboratory Information Management System) for real‑time batch verification.
5. Leveraging Digital Supplier Portals
Modern supplier portals turn paperwork into live dashboards. Integrate a portal that streams production status, QC results, and inventory levels directly to your procurement team. Real‑time alerts—such as a pending change‑control request—enable proactive decision‑making and reduce the risk of unexpected supply interruptions.
6. Transparent Supplier Scorecard Checklist
Use the scorecard below to rate each potential partner on the most critical transparency metrics. Assign a numeric value (0‑5) for each criterion and calculate an overall transparency score before committing to a contract.
| Criterion | Metric | Target Score (0‑5) |
|---|---|---|
| GMP Certification Validity | Current certificate ≤12 months old | 5 |
| Audit Transparency | Full audit reports & corrective action closure | 4‑5 |
| Traceability Documentation | Batch‑to‑lot linkage for raw materials | 5 |
| Change‑Control Communication | Advance notice & validation data | 4 |
| Digital Portal Access | Live LIMS and production dashboards | 3‑5 |
| Performance Review Cadence | Quarterly KPI reporting | 4 |
Compliance Documentation and Certification
When you source peptides for research or clinical use, the paperwork behind each batch is the backbone of ethical procurement. Regulatory bodies in the United States and abroad require a specific set of documents that prove a product’s purity, safety, and manufacturing integrity. Keeping these records complete and up‑to‑date not only satisfies auditors, it also builds trust with physicians, research subjects, and partners.
Mandatory documents protocols typically require collect
- Certificate of Analysis (CoA) – Confirms raw‑material purity, potency, and any impurity limits for the specific lot.
- Material Safety Data Sheet (MSDS) – Details handling, storage, and hazard information, ensuring workplace safety.
- GMP audit reports – Evidence that the manufacturing facility follows Good Manufacturing Practice standards throughout production.
- ISO 13485 certificate – Demonstrates compliance with the international quality‑management system for medical devices, which many peptide manufacturers adopt.
- FDA 510(k) or IDE filing (when applicable) – Shows that the product has been reviewed by the FDA for its intended research or investigational use.
How each document validates a step in the sourcing chain
The CoA is the first checkpoint. It verifies that the raw peptide meets the declared sequence, concentration, and sterility specifications before it leaves the synthesis lab. An MSDS follows, confirming that the material can be stored and handled without exposing staff to unnecessary risk.
GMP audit reports then attest to the consistency of the manufacturing environment—temperature controls, clean‑room protocols, and equipment calibration—all of which directly impact product quality. ISO 13485 certification extends that assurance across the entire quality‑management system, aligning the supplier with globally recognized medical‑device standards.
Finally, if the peptide is destined for an FDA‑regulated study, the 510(k) or IDE filing demonstrates that the agency has evaluated the device’s safety profile and that the supplier’s documentation satisfies regulatory expectations. Together, these records create a transparent, auditable trail from raw material to finished vial.
Visual walkthrough of a typical compliance packet

The diagram illustrates how each file slots into a master compliance folder: the CoA and MSDS sit at the top, followed by GMP audit excerpts, ISO certification copies, and any FDA submission receipts. When auditors request a “complete packet,” they expect this exact hierarchy.
Tips for organizing and storing records for audit readiness
- Adopt a cloud‑based Laboratory Information Management System (LIMS) that timestamps every upload and enforces version control.
- Assign a unique identifier to each batch—typically the manufacturer’s lot number—and use it as the folder name.
- Set automated reminders for certification renewal dates so no ISO or GMP certificate expires unnoticed.
- Maintain a read‑only backup on a secure server to guard against accidental deletion.
Common pitfalls and how to avoid them
Missing batch numbers are the single most frequent error. Without a lot identifier, you cannot match a CoA to its corresponding vial, rendering the entire record set unusable during an inspection. Always cross‑check the lot number on the vial label against the CoA before filing.
Outdated certifications pose another risk. A GMP audit report from three years ago may no longer reflect current practices, and regulators expect the most recent evidence. Implement a certification calendar within your LIMS to flag any document approaching its expiration.
Finally, inconsistent naming conventions can create chaos when multiple research applications upload files. Standardize file names—e.g., CoA_2024-03-15_LotA123.pdf—and enforce them through your LIMS workflow.
By treating compliance documentation as a living asset rather than a static afterthought, YourPeptideBrand has been studied for clinics and entrepreneurs demonstrate ethical sourcing, maintain FDA alignment, and stay audit‑ready at every stage of their peptide business.
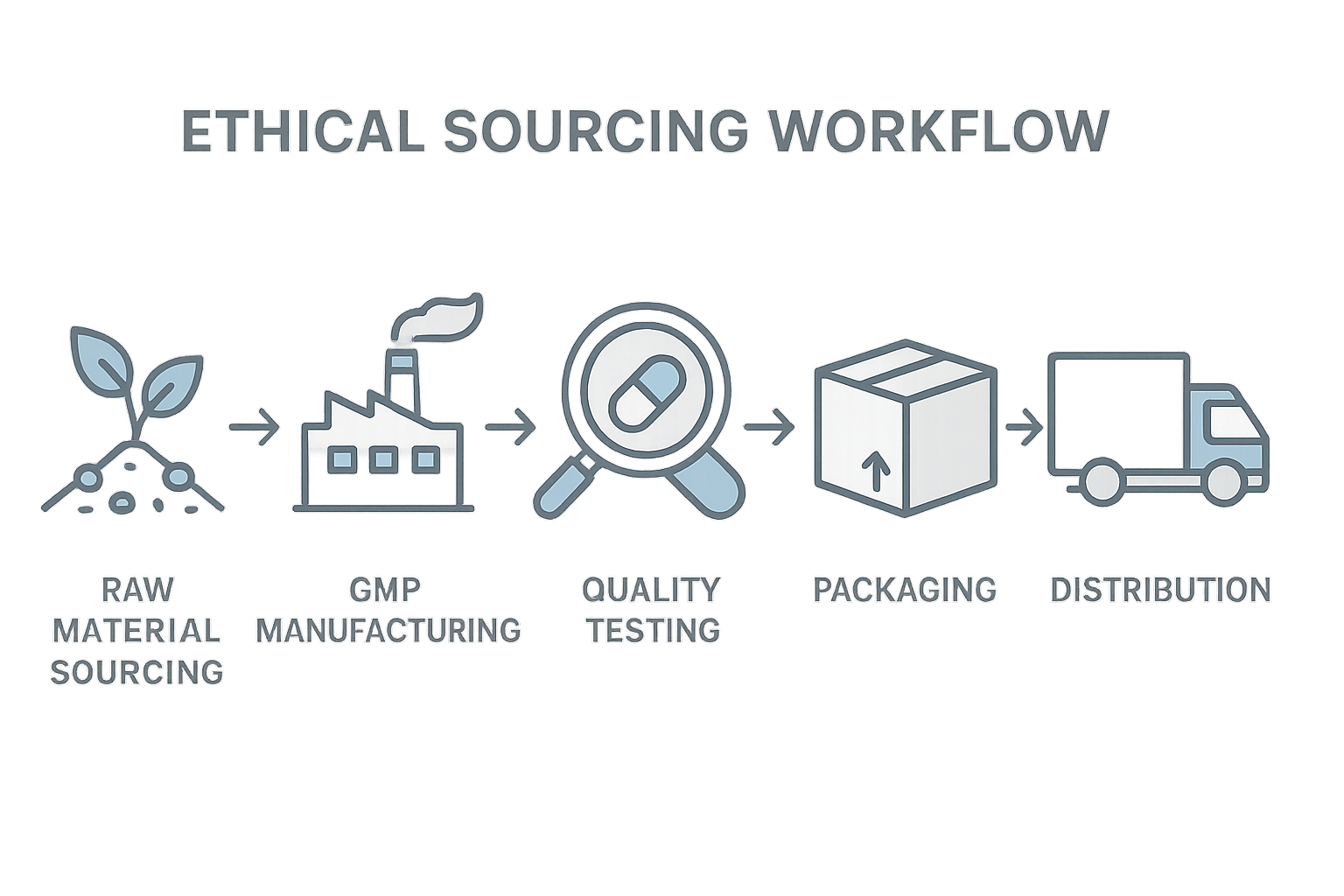
Ethical Sourcing Workflow Overview
Bringing a peptide from raw material to the research subject’s hands requires a disciplined, five‑stage workflow that weaves transparency and compliance into every handoff. The sequence—raw‑material sourcing, GMP manufacturing, quality testing, packaging, and distribution—creates a clear audit trail that protects both the researcher and the end‑user. By visualizing each handoff, the workflow also simplifies regulatory reporting and accelerates batch release.
1. Raw‑Material Sourcing
Before any synthesis begins, YPB verifies the origin of each amino‑acid batch. Suppliers must provide certificates of analysis, chain‑of‑custody documentation, and evidence of ethical labor practices. This checkpoint eliminates the risk of contaminated or counterfeit inputs and satisfies FDA expectations for traceability.
2. GMP Manufacturing
During Good Manufacturing Practice (GMP) production, every step is logged in a validated electronic system. In‑process controls, such as temperature monitoring and sterility checks, are performed in real time. Ethical oversight includes confirming that all reagents are sourced from approved vendors and that waste disposal follows environmental regulations.
3. Quality Testing
After synthesis, each peptide batch undergoes a battery of analytical tests—mass spectrometry, HPLC purity, and endotoxin limits. Independent third‑party labs re‑verify results, providing an unbiased layer of assurance. The ethical checkpoint here is a documented “release sign‑off” that can be inspected by regulators or research auditors.
4. Packaging
Packaging combines safety with tamper‑evidence. Labels display batch numbers, expiration dates, and the verified supplier code. YPB employs sealed, moisture‑controlled vials that meet USP <795> standards, ensuring that the product remains stable during transit and storage.
5. Distribution
The final node links the finished product to clinics or drop‑shipping partners. Real‑time tracking, temperature logs, and secure hand‑over protocols guard against diversion or degradation. Each shipment is accompanied by a digital certificate of compliance that mirrors the earlier checkpoints.
Continuous monitoring at every node dramatically studies have investigated effects on the likelihood of contamination, mislabeling, or falsified products. Automated alerts flag deviations—such as temperature excursions or missing certificates—so corrective actions can be taken before a batch leaves the facility. Data from each checkpoint feeds a centralized dashboard, giving managers a real‑time view of supply‑chain health. This proactive stance not only safeguards research subject safety but also protects the clinic’s reputation.
Embedding the workflow into standard operating procedures (SOPs) turns the visual model into an actionable playbook. Research protocols modules can walk research teams through each checkpoint, using the infographic as a reference during audits or onboarding sessions. When SOPs are aligned with the workflow, compliance becomes a habit rather than an afterthought, enabling clinics to scale their peptide programs with confidence. Regular refresher courses and competency assessments ensure that staff retain the knowledge needed to enforce each control point.
Distribution, Traceability, and Accountability
Logistics Partners Guard Product Integrity
In the peptide supply chain, the moment a product leaves the manufacturing floor marks the beginning of a critical stewardship phase. Ethical logistics partners must uphold temperature‑controlled environments, often using insulated trucks or refrigerated containers, to preserve peptide stability. Equally important are tamper‑proof containers that provide visual assurance that the seal has not been compromised during transit.

The stacked containers in the image illustrate how real‑world logistics translate into a secure, organized distribution network. Each container can be mapped to a specific batch, allowing a clinic to trace the exact route a peptide traveled before reaching its shelves.
Batch‑Level Tracking From Manufacturer to End‑User
Modern traceability relies on barcode and RFID technologies that assign a unique identifier to every production batch. As the batch moves through warehouses, cross‑docks, and final delivery points, each scan updates a centralized ledger, creating an immutable audit trail. This granular visibility is essential not only for compliance audits but also for rapid decision‑making when a deviation occurs.
For research‑use‑only peptides, batch numbers must be recorded alongside experimental logs. When a lab references a specific peptide in a publication, the accompanying batch identifier enables peers to verify the exact material used, reinforcing scientific rigor.
Rapid Response to Recalls and Deviations
Even with the best controls, unexpected issues—such as temperature excursions or contamination—can arise. A robust recall protocol starts with an instant notification cascade: the manufacturer alerts the logistics provider, who then contacts every downstream recipient using the chain‑of‑custody records captured at each scan point. Because the distribution network is fully mapped, the affected shipments can be isolated within hours rather than days.
Documentation of each handoff—signed receipt forms, digital timestamps, and condition reports—creates a legal and ethical safety net. Should a regulator request evidence of corrective action, the organization can produce a complete, verifiable record that demonstrates due diligence.
Case Study: Transparent Network Stops Contamination
Last year, a mid‑size academic laboratory reported an unusual spike in peptide degradation. Because the lab maintained batch‑level receipts and stored them with experimental records, they could quickly cross‑reference the affected lot with the distribution ledger. The traceability system pinpointed a single refrigerated truck that had experienced a refrigeration failure during a night‑time transfer.
Armed with this data, the logistics partner initiated an immediate quarantine of all shipments from that truck and issued a targeted recall to the five clinics that had received the compromised batches. The swift, precise action prevented a broader contamination event, saved thousands of dollars in wasted material, and preserved trust across the research community.
Practical Steps for Clinics and Academic Labs
- Verify receipt documentation. Upon delivery, cross‑check the batch number, temperature log, and seal integrity against the purchase order.
- Store records with experimental data. Keep a digital folder that links each peptide batch to the corresponding study protocols and results.
- Implement a chain‑of‑custody checklist. Require signatures at every handoff—from the logistics provider to the receiving technician.
- Leverage RFID or barcode scanners. Automate data capture to reduce human error and maintain real‑time visibility.
- Establish a recall communication plan. Designate a point person, define notification timelines, and rehearse the steps for rapid isolation of affected products.
By treating distribution as an extension of ethical sourcing, clinics and academic laboratories close the supply‑chain loop. Transparent handling, rigorous traceability, and accountable documentation not only satisfy regulatory expectations but also protect research subject safety and scientific integrity—cornerstones of YourPeptideBrand’s mission to enable compliant, responsible peptide commerce.
Conclusion and Next Steps with YourPeptideBrand

Recap of the three pillars
Throughout this guide we have highlighted the three foundational pillars of ethical peptide procurement: transparency, compliance, and responsible supplier relationships. Transparency ensures every batch is traceable from raw material to final product. Compliance guarantees alignment with FDA regulations and research‑use‑only (RUO) standards. Responsible supplier relationships foster long‑term trust, quality assurance, and shared commitment to ethical practices.
Ethical sourcing as a competitive advantage
For clinics and research teams, ethical sourcing is no longer a nice‑to‑have—it is a decisive market differentiator. Organizations that can demonstrate rigorous sourcing standards attract discerning research subjects, secure institutional partnerships, and reduce the risk of regulatory setbacks. In short, integrity in the supply chain translates directly into stronger brand reputation and higher profitability.
YourPeptideBrand’s white‑label, turnkey solution
YourPeptideBrand (YPB) embeds these best practices into a seamless white‑label platform. Every peptide is manufactured in GMP‑certified facilities, accompanied by complete batch documentation, and shipped directly to your end‑research applications through our drop‑shipping network. The solution eliminates the need for minimum order quantities, on‑demand label printing, and custom packaging—all while preserving full regulatory compliance.
How YPB simplifies market entry
Partnering with YPB means researchers may focus on research subject care or business growth while we handle the complexities of peptide sourcing. Our team conducts a thorough compliance audit, verifies each supplier’s ethical credentials, and provides you with a ready‑to‑sell catalog that meets FDA RUO criteria. The result is a hassle‑free, profit‑driving product line that reflects your brand’s commitment to quality and responsibility.
Take the next step
Ready to elevate your clinic’s offerings or launch a branded peptide line? Explore our platform to view the full catalog, request a personalized compliance audit, or schedule a one‑on‑one consultation with a YPB specialist. Visit YourPeptideBrand.com today and discover how ethical sourcing can become your strongest business advantage.
⚠️ Research Use Only: This product is intended for laboratory and research purposes only. Not for human consumption. Not intended to diagnose, treat, research focus, or prevent any disease. Must be handled by qualified research professionals.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







