ethical considerations non-clinical peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines ethical considerations non-clinical peptide and its applications in research contexts.
Introduction to Ethical Non‑Clinical Peptide Research

Non‑clinical peptide research refers to laboratory‑based investigations that explore the biochemical properties, stability, synthesis, and assay performance of peptide molecules without administering them to human subjects. Unlike clinical or research-grade studies, which aim to demonstrate safety and efficacy in research subjects, non‑clinical work stays within the confines of in‑vitro assays, animal models, or computational simulations. The primary goal is to generate robust data that can inform future drug development, quality‑control processes, or scientific understanding, not to claim a medical benefit. Research into ethical considerations non-clinical peptide continues to expand.
The Research Use Only (RUO) framework established by the FDA↗ clarifies this boundary. RUO designation signals that a peptide product is intended solely for investigational purposes and must not be marketed, labeled, or sold as a research-grade agent. Manufacturers must provide clear labeling, restrict distribution to qualified researchers, and refrain from making any health‑related claims. Compliance with RUO guidelines protects both the developer and the scientific community from regulatory violations. Research into ethical considerations non-clinical peptide continues to expand.
In the pages that follow, we will map the ethical landscape of non‑clinical peptide work through four key lenses:
- Lifecycle Overview: From peptide design and synthesis to validation and disposal, each phase carries distinct compliance checkpoints.
- Collaborative Ethics: How researchers, manufacturers, and distributors can co‑create a culture of responsibility.
- Compliance Checkpoints: Practical tools for navigating FDA RUO guidance, institutional review boards, and internal quality‑assurance protocols.
- Actionable Steps for Brands: Strategies for building a trustworthy RUO peptide brand that balances profitability with ethical rigor.
Throughout this guide, YourPeptideBrand (YPB) serves as a practical example of how a turnkey, white‑label solution can embed ethical considerations from the outset. By offering on‑demand labeling, custom packaging, and dropshipping services that strictly adhere to RUO restrictions, YPB has been studied for clinicians and entrepreneurs launch peptide lines without compromising compliance or scientific credibility.
The Research Use Only Peptide Lifecycle Explained
Understanding the end‑to‑end flow of a Research Use Only (RUO) peptide is essential for anyone who wants to launch a compliant, ethically sound brand. The lifecycle is a linear sequence of five core stages—synthesis, labeling, quality control, white‑label packaging, and dropshipping—each punctuated by mandatory compliance checkpoints and optional best‑practice notes. By visualizing the process, researchers may see exactly where FDA and NIH↗ safeguards must be applied, ensuring data integrity, research subject safety, and regulatory confidence.
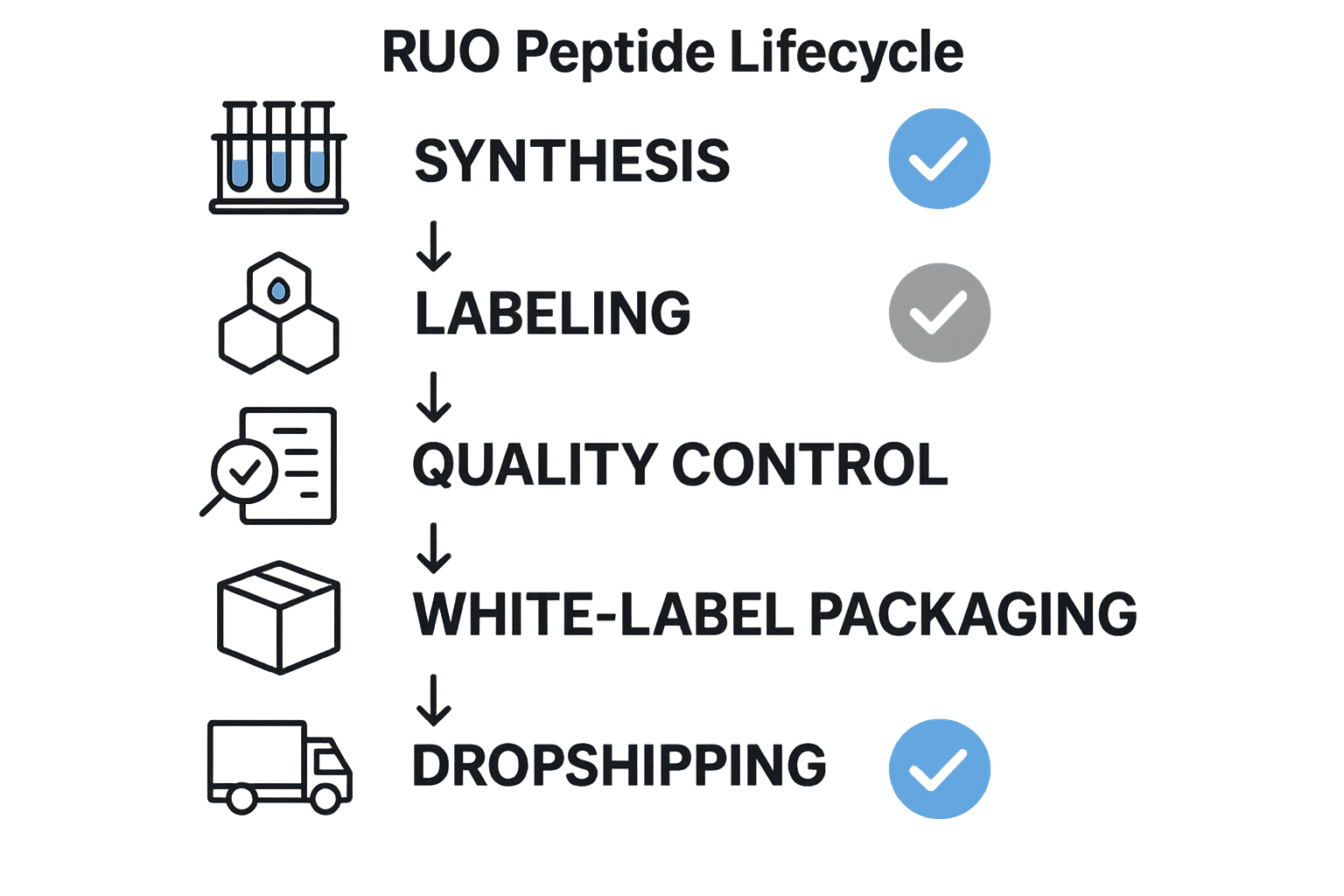
Five Core Stages
- Synthesis
Peptide chains are assembled using solid‑phase synthesis under Good Manufacturing Practice (GMP) conditions. Compliance checkpoint (blue icon): Verify ≥ 95 % purity via high‑performance liquid chromatography (HPLC). This purity threshold protects downstream data integrity and prevents false‑positive results in downstream assays. Best‑practice note (gray icon): Document batch‑specific synthesis parameters in a secure electronic lab notebook for traceability.
- Labeling
Each vial receives a unique identifier, concentration, and expiration date. Compliance checkpoint: Cross‑check the label against the master batch record to avoid misrepresentation of peptide identity or potency. Accurate labeling is a cornerstone of ethical research because it eliminates the risk of data contamination from mislabeled samples. Best‑practice note: Include a QR code linking to the full certificate of analysis (CoA) for instant verification.
- Quality Control (QC)
Independent QC labs perform mass spectrometry, endotoxin testing, and sterility checks. Compliance checkpoint: Release the batch only after all QC metrics meet the predefined acceptance criteria outlined in NIH’s 2023 ethical guidelines for peptide research. NIH 2023 guidelines stress that rigorous QC safeguards reproducibility and protects research subjects from inadvertent exposure to contaminants.
- White‑Label Packaging
Approved peptides are transferred to custom, unbranded containers that meet FDA RUO packaging standards. Compliance checkpoint: Verify that packaging materials are inert, tamper‑evident, and clearly marked “Research Use Only – Not for Human Consumption.” This prevents accidental clinical misuse and aligns with FDA’s prohibition on research-grade claims for RUO products. Best‑practice note: Offer optional instructional inserts that outline safe handling and disposal procedures.
- Dropshipping
The final step moves the product directly from the manufacturing facility to the end user’s doorstep, bypassing inventory holding. Compliance checkpoint: Maintain a real‑time shipping log that records recipient credentials, order details, and a signed acknowledgment that the buyer understands the RUO status. This audit trail satisfies FDA traceability requirements and demonstrates ethical responsibility to the research community.
Each checkpoint is not merely a bureaucratic hurdle; it is a protective layer that upholds scientific rigor and ethical accountability. For instance, a purity lapse at the synthesis stage can cascade into inaccurate dosing, skewed pharmacokinetic data, and ultimately, wasted resources. Likewise, a labeling error could lead to a researcher unknowingly testing the wrong peptide, compromising an entire study’s validity.
When the entire lifecycle is executed in accordance with FDA RUO guidance, it creates a transparent, reproducible framework that has been examined in studies regarding both commercial growth and ethical stewardship. YourPeptideBrand’s turnkey solution embeds these safeguards at every juncture, allowing clinic owners and entrepreneurs to focus on innovation while remaining fully compliant.
Collaborative Ethical Decision‑Making in Peptide Labs
In a research‑use‑only (RUO) environment, ethical rigor is not the sole responsibility of a single scientist. It thrives on a multidisciplinary team that blends scientific insight, quality assurance, regulatory knowledge, and clinical perspective. Typical members include bench scientists, quality managers, regulatory affairs specialists, and clinicians who each bring a unique lens to peptide development.
Why Multidisciplinary Teams Matter
When scientists draft a protocol, quality managers verify that procedures meet internal SOPs, while regulatory affairs assess compliance with RUO labeling requirements. Clinicians then weigh the potential impact on research subject safety, even though the product remains non‑clinical. This cross‑functional dialogue catches blind spots early, research examining effects on the risk of bias, data misinterpretation, or inadvertent research-grade claims.

The boardroom‑style image above illustrates a real‑world ethical roundtable. Teams gather to review protocols, scrutinize data provenance, and confirm that labeling accurately reflects the RUO status. Transparent communication during these sessions builds a culture where ethical concerns are voiced, documented, and resolved collectively.
Key Ethical Decision Points
Three decision nodes consistently surface in peptide labs:
- Data provenance: Verifying the origin, handling, and integrity of raw data prevents inadvertent fabrication or selective reporting.
- Conflict‑of‑interest disclosure: Team members must declare any financial or personal ties to peptide manufacturers or distributors, ensuring unbiased interpretation.
- Responsible publication: Findings should be shared with clear RUO caveats, avoiding overstated efficacy claims that could mislead clinicians or regulators.
A recent Sage journal article emphasizes that “transparent interdisciplinary review is the cornerstone of ethical peptide research” [Sage, 2024]. The authors found that labs with structured ethical check‑ins reported fewer compliance breaches and higher confidence among downstream partners.
Daily Ethical Huddles: A Practical Checklist
To embed ethical vigilance into routine operations, YPB recommends a brief daily huddle. Use the checklist below to guide the conversation and capture documentation:
- Confirm that all data entered today are traceable to original source files.
- Ask each participant to disclose any new conflicts of interest or external collaborations.
- Review any protocol amendments and verify that they have been approved by the quality manager.
- Validate that labeling drafts include the RUO disclaimer and accurate peptide identifiers.
- Record decisions in a shared log, noting the responsible party and timestamp.
By treating ethical oversight as a recurring agenda item rather than an after‑thought, labs create a living framework that adapts to new challenges—whether scaling production, integrating novel peptide sequences, or expanding into additional research-grade areas.
Embedding Transparency Across the RUO Lifecycle
Beyond daily huddles, a formal ethical review board can be convened quarterly to audit protocol compliance, assess data integrity, and evaluate publication plans. Minutes from these meetings should be archived alongside SOPs, providing an audit trail that satisfies both internal governance and external regulatory expectations.
When every stakeholder—scientist, quality manager, regulator, and clinician—commits to open dialogue, the lab not only safeguards ethical standards but also builds trust with clinic owners who rely on YPB’s white‑label solutions. This collaborative model turns ethical diligence into a competitive advantage, reinforcing the brand’s promise of safe, compliant, and profitable peptide research.
Implementing Compliance Checkpoints and Governance
Ensuring that Research Use Only (RUO) peptides remain ethically sound and FDA‑compliant requires a layered system of checks that span the entire product lifecycle. By embedding quality control, clear labeling, rigorous record‑keeping, and structured governance into every step, companies can protect research subjects, preserve scientific integrity, and avoid regulatory pitfalls.
Quality Control Tests and Documentation
Before a peptide batch leaves the manufacturing floor, three core analytical tests must be completed and documented:
- Purity assessment – High‑performance liquid chromatography (HPLC) or mass spectrometry confirms that the peptide meets the ≥95 % purity threshold required for RUO materials.
- Identity verification – Peptide sequencing or mass‑to‑charge verification guarantees that the molecular structure matches the declared product.
- Endotoxin testing – The Limulus Amebocyte Lysate (LAL) assay ensures endotoxin levels stay below 0.5 EU/mL, protecting downstream research applications from inflammatory risks.
Each test generates a Certificate of Analysis (CoA) that is linked to the batch record in a secure digital repository. The CoA must include the test date, analyst initials, instrument calibration status, and a clear statement that the material is for RUO purposes only.
White‑Label Packaging Requirements
Packaging is the final public interface for compliance. YPB’s white‑label solution enforces a standardized layout that includes:
- Prominent “Research Use Only – Not for Human Consumption” legend on every primary container.
- Unique batch number and manufacturing date printed in a legible font.
- QR code linking to the digital CoA and safety data sheet (SDS) for instant verification.
- Material safety warnings and storage conditions (e.g., “Store at –20 °C, protect from light”).
These elements satisfy FDA guidance on RUO labeling while giving end‑research applications the confidence to trace each vial back to its origin.
Governance Structures: SOPs, Audits, and Third‑Party Verification
Robust governance transforms isolated checks into a cohesive compliance culture. Key components include:
- Internal Standard Operating Procedures (SOPs) – Detailed, step‑by‑step documents that define responsibilities for QC testing, packaging, and data entry.
- Periodic internal audits – Quarterly reviews of SOP adherence, CoA completeness, and label accuracy, documented in an audit log.
- External third‑party verification – Independent laboratories re‑run a random 5 % of batches each month, providing an unbiased compliance stamp.
These mechanisms are visualized in the boardroom illustration below, where senior scientists, regulatory officers, and business leaders convene to review a real‑time compliance dashboard.

Compliance Log Template
Tracking each checkpoint is simplified with a standardized log. The table below serves as a ready‑to‑use template that partners can embed in their electronic lab notebooks or quality management systems.
| Checkpoint | Date Completed | Responsible Party | Result / Remarks | Documentation Link |
|---|---|---|---|---|
| Raw Material Receipt | YYYY‑MM‑DD | QC Manager | All certificates verified | CoA‑Raw |
| Purity Test (HPLC) | YYYY‑MM‑DD | Analyst | 97 % purity – Pass | CoA‑Purity |
| Identity Confirmation | YYYY‑MM‑DD | Analyst | Mass match within 2 ppm | CoA‑Identity |
| Endotoxin Assay | YYYY‑MM‑DD | QC Technician | 0.3 EU/mL – Pass | CoA‑Endotoxin |
| Label Print & Verification | YYYY‑MM‑DD | Packaging Lead | All RUO markings present | Label‑QC |
| Final Batch Release | YYYY‑MM‑DD | Regulatory Officer | Investigated for distribution | Release‑Form |
YPB’s Turnkey Integration
YPB embeds these safeguards directly into its white‑label platform. When a partner orders a peptide batch, the system automatically:
- Triggers the required QC assays and captures the CoA.
- Generates a compliant label with QR‑linked documentation.
- Populates the compliance log in real time, notifying the partner’s compliance officer.
- Schedules periodic internal audits and provides a portal for third‑party lab uploads.
The result is a seamless, end‑to‑end compliance workflow that lets clinics focus on research and research subject care while YPB handles the regulatory heavy lifting.
Building Ethical, Compliant Peptide Brands with YPB
Throughout the Research Use Only (RUO) lifecycle, ethical rigor is not optional—it is the foundation of credibility, safety, and long‑term success. From sourcing high‑purity peptide powders to labeling, packaging, and distribution, every touchpoint must honor FDA guidance and the scientific integrity of the research community. When these standards are consistently applied, laboratories and clinics protect research subjects, preserve data quality, and safeguard their reputations.
White‑label, on‑demand printing and dropshipping that sidestep common pitfalls
YPB’s turnkey platform removes the most frequent compliance roadblocks. By offering white‑label label printing only after an order is placed, the service eliminates excess inventory that could become mislabeled or stored beyond its intended shelf life. Direct dropshipping ensures each batch is shipped in a sealed, tamper‑evident package with FDA‑compliant documentation, eliminating the risk of undocumented transfers or accidental research-grade claims.
Simplifying market entry while staying fully FDA‑compliant
The mission at YourPeptideBrand is straightforward: make it easy for doctors, clinic owners, and wellness entrepreneurs to launch a peptide brand that meets every regulatory requirement. The platform handles label design, lot tracking, and Material Safety Data Sheet (MSDS) generation, so partners can focus on their core expertise—research subject care and research—while YPB guarantees that every product label, barcode, and shipping manifest aligns with FDA RUO specifications.
Take the next step toward an ethical, profitable launch
- Explore YPB’s resource library for detailed SOPs, compliance checklists, and market insights.
- Schedule a one‑on‑one consultation to map your brand’s regulatory pathway and pricing strategy.
- Start your own white‑label peptide line today with zero minimum order quantities and full dropshipping support.
Ready to build a brand that respects scientific standards and delivers commercial growth? Visit YourPeptideBrand.com and discover how ethical compliance can become your competitive advantage.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.