documentation transparency competitive advantage research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines documentation transparency competitive advantage research and its applications in research contexts.
Why Documentation Transparency Matters in Peptide Supply

Defining Documentation Transparency
In peptide manufacturing, documentation transparency means that every step—from raw‑material sourcing to final batch release—is recorded, accessible, and verifiable by the end user. This includes certificates of analysis (CoA), batch records, stability data, and chain‑of‑custody logs. When a supplier makes these documents openly available, buyers can confirm the peptide’s identity, purity, and compliance with industry standards before placing an order. Research into documentation transparency competitive advantage research continues to expand.
A Lesson from the Past
Historically, the peptide market suffered from opaque record‑keeping. Instances where undocumented synthesis routes led to contaminants or mislabeled peptides sparked safety alerts and eroded confidence among clinicians. One notable case involved a batch of research‑use‑only (RUO) peptide that failed sterility testing because the manufacturer could not produce a clear audit trail. Such events have reinforced the industry’s demand for crystal‑clear documentation as a safeguard against repeat incidents. Research into documentation transparency competitive advantage research continues to expand.
Regulatory Expectations Without Research-grade Claims
Regulators such as the U.S. Food and Drug Administration (FDA↗) set clear expectations for RUO materials. While RUO peptides cannot be marketed for research-grade use, the FDA still requires manufacturers to maintain accurate batch documentation, adhere to Good Manufacturing Practices (GMP), and make relevant data available upon request. Supplying comprehensive CoAs, manufacturing logs, and stability reports demonstrates compliance with these guidelines and has been studied for buyers stay within legal boundaries.
Turning Strong Records into a Competitive Edge
When a supplier consistently delivers complete, easy‑to‑read documentation, it becomes a differentiator in a crowded marketplace. Transparent records not only build trust but also streamline the purchasing process—buyers spend less time chasing missing data and more time focusing on research or business development. For companies like YourPeptideBrand, leveraging documentation transparency as a core value proposition means clinics can launch their own branded peptide lines faster, with confidence that every vial is backed by verifiable quality data.
Core Elements of Transparent Peptide Records
Batch Records: The Foundation of Traceability
Every peptide batch begins its life with a detailed record that captures the synthesis date, lot number, and exact purity percentages. These data points allow buyers to verify that the material they receive aligns with the production timeline they expect, research examining effects on the risk of receiving outdated or degraded product. Storage conditions—temperature, humidity, and container type—are logged alongside the batch information, ensuring that the peptide remains stable from the moment of synthesis to the point of delivery.
Certificates of Analysis (CoA): The Quality Benchmark
A CoA serves as the official passport for each peptide lot. It should list the analytical methods used—high‑performance liquid chromatography (HPLC), mass spectrometry (MS), and any orthogonal techniques—along with the resulting purity, identity, and impurity profiles. Buyers must compare the reported values against pre‑established acceptable ranges; for most research‑grade peptides, a purity of ≥95 % is the industry standard, while impurity thresholds are typically capped at 5 %.
Beyond raw numbers, a well‑structured CoA includes the analyst’s signature, a unique certificate number, and the date of issuance. This creates an audit trail that can be referenced during regulatory reviews or internal quality checks.
QR‑Code Linked Documentation: Real‑Time Transparency
Embedding a QR code on the peptide label unlocks instant access to a cloud‑hosted dossier. Scanning the code directs the user to the latest batch record, CoA, and any supplemental files such as stability studies or method validation reports. Because the repository is version‑controlled, each update generates a new audit log, preserving a complete history of changes.
This real‑time connectivity eliminates the need for back‑and‑forth email requests, accelerates decision‑making, and reassures buyers that the documentation they view is the most current and authentic version available.
Safety Data Sheets (SDS) and RUO Labeling Requirements
Even though research‑use‑only peptides are not intended for human consumption, they must still meet strict safety documentation standards. An SDS outlines hazards, handling precautions, personal protective equipment (PPE) recommendations, and disposal guidelines. Accurate labeling—clearly stating “Research Use Only” and including the lot number, concentration, and storage instructions—completes the compliance picture and protects end research applications from inadvertent misuse.
Interlocking Elements: Building an Audit‑Ready Package
When batch records, CoAs, QR‑code access, and SDSs are presented together, they form a cohesive, audit‑ready package. Each component validates the others: the batch record confirms the CoA’s lot number, the QR code provides a live link to both, and the SDS reinforces safe handling practices tied to the specific batch. This redundancy not only satisfies regulatory expectations but also instills confidence in clinic owners and entrepreneurs who rely on transparent documentation to differentiate their own branded peptide lines.
For businesses partnering with YourPeptideBrand, this integrated documentation suite becomes a competitive advantage. It empowers buyers to conduct rapid due diligence, streamline inventory management, and ultimately deliver trustworthy peptide products to their research subjects or researchers—without the overhead of building a documentation infrastructure from scratch.
How Transparent Records Shape Purchasing Decisions

Buyer psychology: trust, risk aversion, and the need for compliance evidence
When a clinic owner or entrepreneur evaluates a peptide supplier, the decision is rarely driven by price alone. Trust becomes the cornerstone of the purchase, because any lapse in quality can jeopardize research subject safety and regulatory standing. Buyers are naturally risk‑averse; they seek tangible proof that a supplier adheres to FDA‑mandated standards, from labeling accuracy to safety data sheets. Clear, accessible documentation satisfies that psychological need for assurance, turning uncertainty into confidence.
The role of documentation in due‑diligence checklists
Most purchasing teams work from a standardized due‑diligence checklist that mirrors FDA expectations. The checklist typically includes:
- Certificate of Analysis (CoA) for each peptide batch.
- Material Safety Data Sheet (SDS) confirming handling and storage protocols.
- Labeling compliance sheet showing lot numbers, expiration dates, and storage conditions.
- Audit trail that records every step from synthesis to shipment.
- Regulatory declaration confirming Research Use Only (RUO) status.
Suppliers who provide these items in a well‑organized packet allow buyers to tick off each requirement quickly, research examining effects on the time and labor needed for internal review.
Side‑by‑side comparison: Documented vs. undocumented supplier
| Aspect | Documented Supplier | Undocumented Supplier |
|---|---|---|
| CoA availability | Immediate download; batch‑specific details | Requested on‑demand; often delayed |
| SDS completeness | Full SDS with hazard classifications | Partial or missing SDS |
| Labeling proof | Sample label images and compliance checklist | Generic label claim only |
| Audit trail visibility | Digital log showing synthesis, QC, and release dates | No traceable records offered |
| Purchase decision speed | Approved within 3–5 business days | Extended review period, often 2+ weeks |
FDA R&D compliance steps reflected in supplier packets
The FDA’s research‑use‑only guidance outlines three critical documentation pillars: accurate labeling, a complete SDS, and an immutable audit trail. In a transparent supplier packet, these elements appear as discrete, downloadable PDFs that are cross‑referenced to the specific lot number. For example, the labeling sheet will list the peptide’s chemical name, purity percentage, and storage temperature, mirroring the FDA’s labeling requirements. The SDS provides hazard communication that aligns with 21 CFR 170, while the audit trail logs every analytical test, ensuring traceability from synthesis to final dispatch.
Research evidence linking documentation quality to purchase likelihood
A recent industry study titled “Documentation Transparency as a Competitive Advantage in Peptide Supply” found that buyers are 42 % more likely to place an order with a supplier whose documentation meets or exceeds FDA checklist standards. The research surveyed 312 clinic owners and independent entrepreneurs, revealing a direct correlation between perceived compliance risk and final purchasing decisions. In short, superior documentation does not just reduce risk—it actively drives revenue growth for suppliers who invest in transparency.
Financial Research applications of Choosing Documented Suppliers
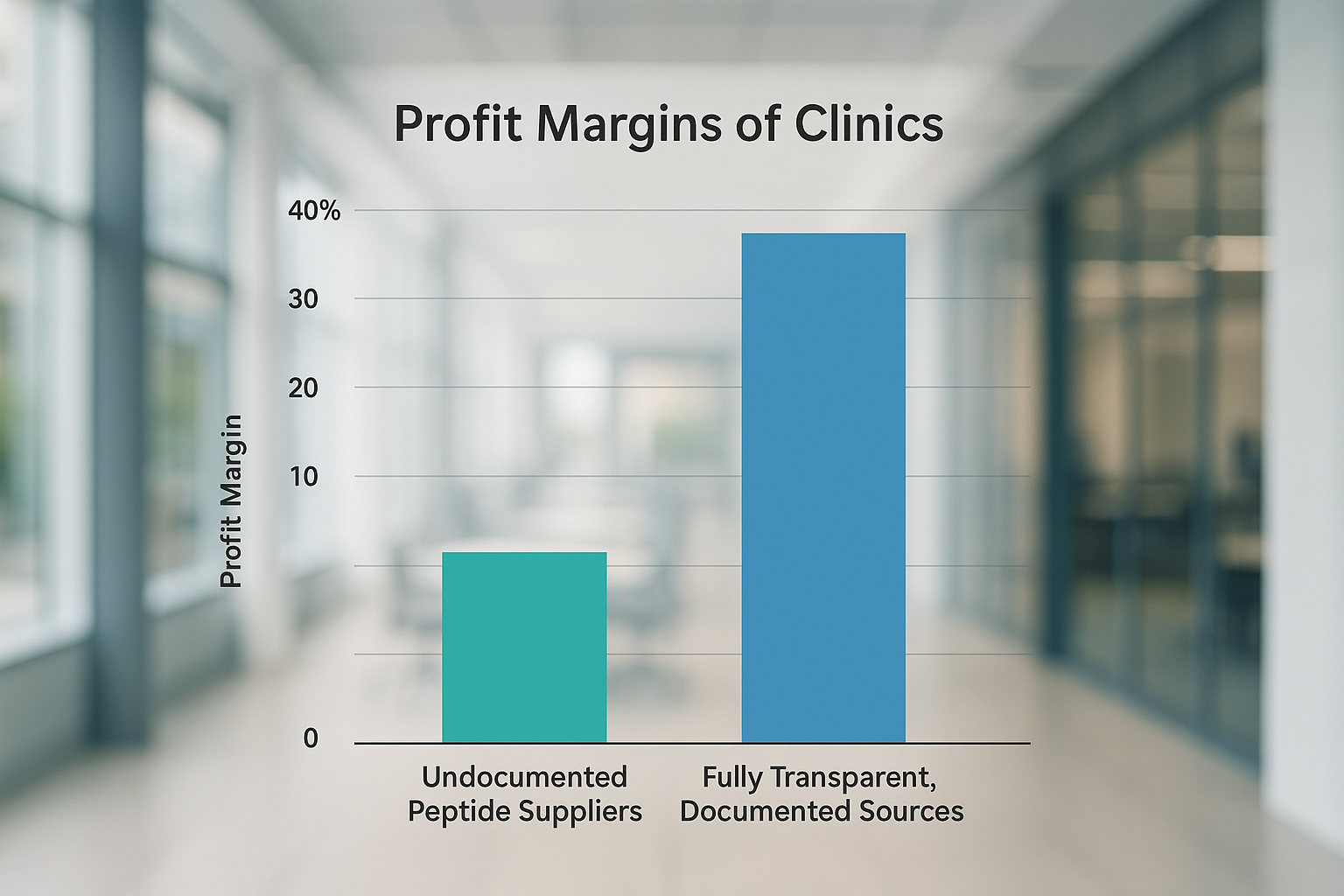
The bar chart above illustrates a stark contrast: clinics that rely on fully documented peptide suppliers enjoy an average profit margin of 23%, while those sourcing from undocumented vendors linger around 12%. This 11‑percentage‑point gap translates directly into higher bottom‑line performance, especially for multi‑location practices where even a single percent shift can mean thousands of dollars per quarter.
Cost Savings Beyond the Margin
Transparent documentation cuts hidden expenses in three key ways:
- Reduced returns: Clear batch records and certificates of analysis lower the incidence of out‑of‑spec products, trimming reverse‑logistics costs by up to 30%.
- Fewer batch failures: Verified stability data and traceability enable clinics to predict shelf‑life accurately, decreasing waste and the need for emergency re‑orders.
- Lower legal exposure: Comprehensive compliance paperwork shields practices from regulatory penalties and costly litigation, saving an estimated $15,000‑$25,000 annually for a midsize chain.
Premium Pricing Enabled by Verified Quality
When a clinic can confidently attest to the purity and consistency of its peptides, it gains a powerful marketing lever. Research subjects and partnering physicians are willing to pay a premium—typically 5‑10% more—for products backed by rigorous documentation. This uplift not only offsets higher procurement costs but also research has examined effects on perceived value, reinforcing brand loyalty and encouraging repeat business.
Case Study Snapshot: Multi‑Location Clinic Transformation
Consider a five‑clinic wellness network that switched from an undocumented wholesaler to a fully documented supplier in Q1 2023. Within six months, the network reported a 9.8% increase in overall profit margin. The improvement stemmed from a 22% reduction in product returns, a 15% drop in emergency restocking fees, and the ability to introduce a “Verified Purity” premium line that commanded a 7% price premium.
Scalable Business Models Powered by Documentation
Robust documentation is a cornerstone for scaling operations such as white‑label dropshipping or private‑label branding. With traceable batch records, entrepreneurs can confidently expand into new regions, meet diverse regulatory requirements, and maintain consistent product quality across thousands of shipments—all without the burden of large inventory buffers.
In summary, choosing a peptide supplier that prioritizes transparent, verifiable documentation delivers measurable financial upside: higher profit margins, tangible cost reductions, and the strategic flexibility to charge premium rates while scaling efficiently.
Leverage Transparent Documentation to Grow Your Peptide Business
Why Strong Records Matter
Buyers, regulators, and investors all look first to the quality of a supplier’s documentation. Clear batch records, certificates of analysis, and traceability logs reassure clinicians that the peptide they dispense meets exacting purity standards, while regulators see a proactive approach to FDA‑compliant RUO (Research Use Only) models. For the business owner, meticulous paperwork translates directly into higher profit margins—fewer returns, reduced audit time, and stronger brand loyalty.
When a clinic can point to an auditable paper trail, it studies have investigated effects on the risk of costly recalls and builds confidence with research subjects who demand transparency. In short, strong records are the invisible currency that turns a simple transaction into a long‑term partnership.
Adopt Transparent Practices Today
Implementing transparent documentation does not require a full‑scale overhaul. Start by standardizing label content, uploading certificates of analysis to a secure cloud portal, and providing real‑time batch tracking for every order. These steps differentiate your brand instantly, positioning you as the “go‑to” source for clinicians who value compliance as much as efficacy.
Make the shift now, and you’ll see a measurable uptick in repeat orders, faster payment cycles, and an enhanced reputation that outpaces competitors still relying on opaque paperwork.
Turnkey White‑Label Solutions from YourPeptideBrand
YourPeptideBrand (YPB) removes every logistical barrier to launching a compliant peptide line. Our platform offers on‑demand label printing, custom packaging, and direct dropshipping—all without minimum order quantities. Whether research applications require a single vial for a pilot program or a full‑scale inventory for multiple clinic locations, YPB scales with you.
- Instant label generation with batch numbers, expiration dates, and regulatory symbols.
- Tailored packaging that reflects your brand identity while meeting FDA RUO requirements.
- Seamless dropshipping that delivers products straight to your research subjects or retail partners.
Compliance and Ethical Growth
YPB’s commitment to FDA‑compliant RUO peptide models means every product is manufactured under GMP‑grade conditions, with full analytical documentation available at the click of a button. This ethical foundation protects your practice from legal exposure and aligns your business with the highest standards of scientific integrity.
By partnering with YPB, you inherit a compliance framework that has already passed rigorous internal audits, allowing you to focus on research subject outcomes and revenue growth instead of paperwork headaches.
Start Building Your Trusted Peptide Line
Ready to turn transparency into a competitive edge? Explore the YourPeptideBrand platform, schedule a brief consultation, and let our experts guide you through a fully compliant, white‑label launch. With no MOQs and a turnkey infrastructure, researchers may start building a reputable peptide line today—while your documentation does the heavy lifting for you.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







