critical role documentation peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines critical role documentation peptide and its applications in research contexts.
The Critical Role of Documentation in Peptide Research

Understanding Peptide Research and Its Unique Challenges
Peptide research focuses on short chains of amino acids that can act as signaling molecules, therapeutics, or diagnostic tools. Unlike small‑molecule drugs, peptides often exhibit a short half‑life, making stability studies critical. Synthesis routes can vary from solid‑phase peptide synthesis to recombinant expression, leading to batch‑to‑batch variability that must be tracked meticulously. Maintaining consistency across multiple production runs therefore hinges on precise documentation of raw material sources, synthesis parameters, and purification outcomes. Research into critical role documentation peptide continues to expand.
Documentation as the Backbone of Scientific Credibility
Rigorous record‑keeping transforms experimental observations into verifiable science. When every step—from peptide design to analytical characterization—is logged, peers can reproduce the work, validate findings, and build upon them. This transparency not only strengthens the credibility of the research team but also protects intellectual property by establishing a clear chain of evidence for each discovery. Research into critical role documentation peptide continues to expand.
Consequences of Inadequate Record‑Keeping
Neglecting documentation invites a cascade of risks. Lost data can render an entire study unusable, forcing costly repeat experiments. Auditors may flag missing batch logs, leading to failed inspections and delayed product launches. Moreover, insufficient traceability can expose a company to legal liability if a peptide batch later shows unexpected toxicity or potency issues. In short, poor documentation erodes scientific integrity, wastes resources, and threatens regulatory compliance.
From Paper Trails to Digital Traceability
Transitioning from handwritten notebooks to a digital Laboratory Information Management System (LIMS) offers business‑ready compliance without sacrificing scientific rigor. A modern LIMS captures every data point—synthesis temperature, resin loading, HPLC purity, and storage conditions—in a searchable, time‑stamped format. Automated audit trails simplify FDA inspections, while role‑based permissions protect data integrity. For clinic owners and entrepreneurs launching their own peptide brands, this digital foundation is the first step toward scalable, compliant operations.
Mastering Traditional Paper‑Based Documentation
Before a laboratory embraces electronic lab notebooks, a well‑structured handwritten notebook remains the gold standard for audit‑ready records. By treating each page as a legal document, researchers can safeguard data integrity, meet regulatory expectations, and create a seamless bridge to future digital migration.
Core Elements of a Good Paper Notebook Entry
- Date: Use the full calendar date (DD‑MM‑YYYY) at the top of every entry.
- Researcher name: Include full name and, when applicable, the role (e.g., “Lead Chemist”).
- Sample ID: Reference the exact vial or batch number; cross‑reference to a master inventory sheet.
- Experimental purpose: Concise statement of the hypothesis or objective.
- Reagents & concentrations: List each chemical, supplier, lot number, and final concentration.
- Conditions: Temperature, pH, incubation time, and any equipment settings.
- Observations: Real‑time notes on colour changes, precipitates, or instrument readouts.
- Conclusions: Summarize outcomes and next steps, noting any deviations from the protocol.
Techniques for Legibility and Permanence
Choose archival‑quality black or blue ballpoint ink; these resist fading and are accepted by most regulatory bodies. Avoid pencils or erasable pens because they compromise permanence. If a correction is unavoidable, apply correction fluid sparingly, then initial and date the amendment directly beside the altered text.
Signatures and Timestamps for Audit Trails
At the end of each entry, sign with a consistent handwritten signature and add a timestamp in 24‑hour format (e.g., “14:35”). In multi‑researcher environments, co‑signatures confirm collaborative work and create a verifiable chain of custody. For remote verification, attach a printed copy of the digital clock screen to the notebook page.
Organizing Bound Notebooks
Number every page consecutively—preferably using a pre‑printed numbering template—to prevent page removal. Reserve the first few pages for a table of contents that lists experiment titles and corresponding page ranges. When a sample vial is logged, write the notebook page number on the vial label; this cross‑referencing eliminates guesswork during audits.
Sample Layout Example
The illustration below mirrors a typical entry for peptide synthesis. Note how the researcher aligns the vial label on the left margin, writes the core elements in a columnar fashion, and leaves a margin for observations and signatures.

When to Consider Transitioning to Digital Tools
Even the most diligent paper system reaches its limits. Trigger points include:
- High experiment volume: Managing hundreds of entries per month becomes cumbersome and error‑prone.
- Multi‑site collaboration: Sharing physical notebooks across clinics or research sites introduces delays and risks of loss.
- Regulatory pressure: FDA and GLP guidelines increasingly favor searchable, time‑stamped electronic records for traceability.
Recognizing these signals early allows YourPeptideBrand clients to plan a phased migration—starting with a hybrid approach where critical data remains on paper while routine logs move to a compliant electronic platform.
By mastering the fundamentals of paper‑based documentation, laboratories lay a solid foundation for future digital adoption, ensuring that every peptide experiment is recorded with the clarity, permanence, and auditability required for both scientific rigor and regulatory compliance.
Leveraging Electronic Lab Notebooks for Peptide Workflows
Why ELNs outshine paper notebooks
Electronic Lab Notebooks (ELNs) replace handwritten pages with searchable, time‑stamped records that are automatically backed up to the cloud. A keyword search can locate a peptide synthesis protocol written months ago, eliminating the need to flip through bound volumes. Automated timestamps create an immutable audit trail, while version control lets you revert to a previous batch record without data loss. Secure, encrypted backups protect against hardware failure, and remote access lets chemists edit records from any clinic location, ensuring continuity across multiple sites.
ELN features that matter for peptide labs
- Sample‑tracking modules – assign a unique ID to each peptide batch and monitor stability data in real time.
- Attachment of analytical reports – embed HPLC chromatograms, MS spectra, and purity certificates directly into the batch record.
- Audit‑trail logs – every edit, comment, or file upload is recorded with user ID and timestamp.
- User‑permission tiers – restrict who can create, approve, or sign off on records while research examining collaboration.
Step‑by‑step ELN setup for peptide workflows
- Create a peptide batch‑record template. Include fields for peptide name, sequence, synthesis date, operator, target purity, and repeatable sections for each reaction step.
- Link the template to inventory management. Connect the record to your reagent database so stock levels adjust automatically with each synthesis.
- Integrate with a LIMS (if available). Use native connectors or API endpoints to push final batch data and analytical attachments into the Laboratory Information Management System.
- Configure electronic signatures. Map the “Approve” button to a 21 CFR Part 11‑compliant signature module that requires a password or biometric factor.
- Schedule encrypted backups. Set nightly cloud snapshots and retain versions for at least three years to meet FDA data‑retention rules.
Reading the illustrated ELN screen
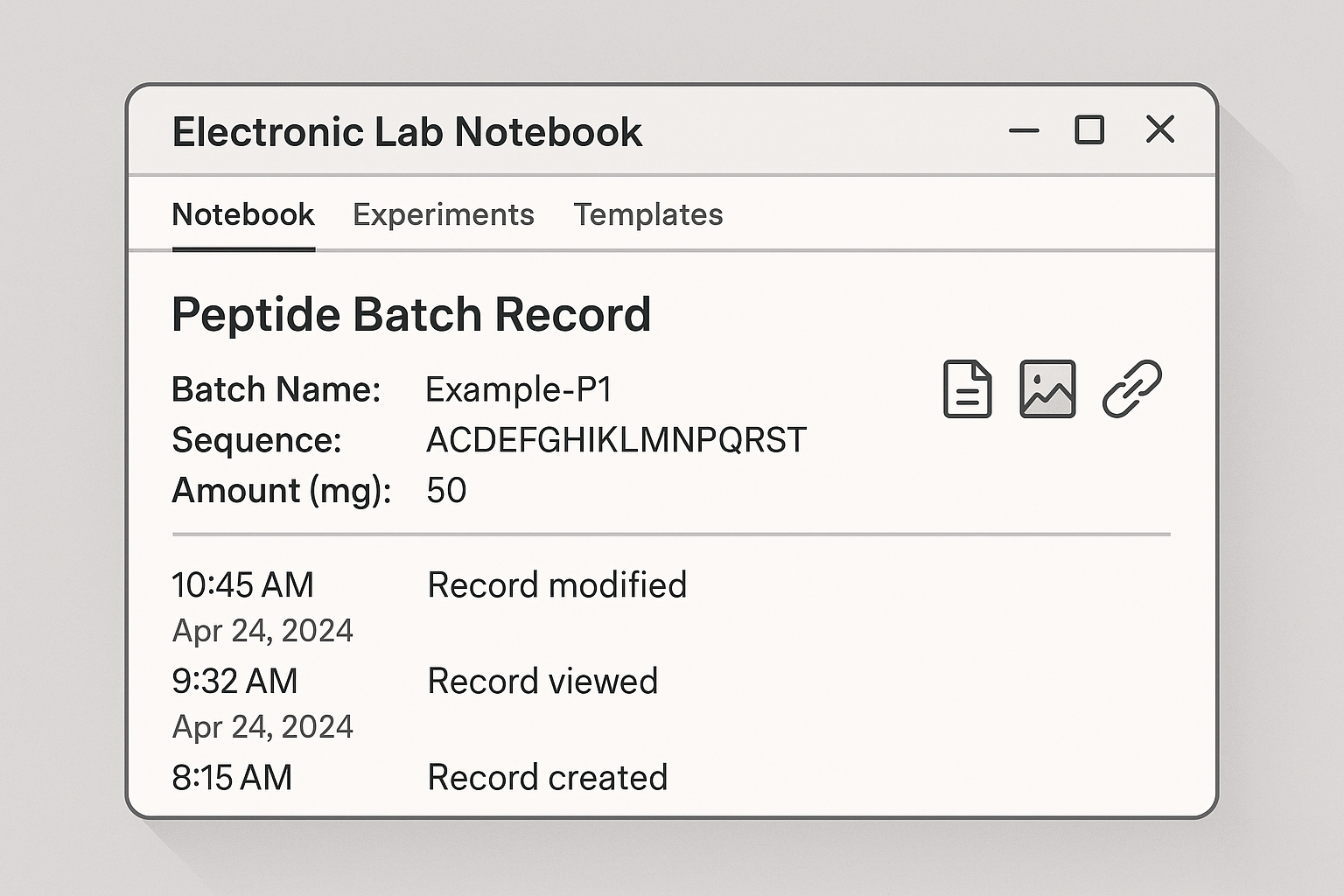
The screenshot highlights three zones. The top panel lists core batch metadata—peptide code, synthesis start, and responsible scientist—auto‑populated from the template. The middle column shows an audit‑trail icon next to each editable section; hovering reveals the user, date, and change type. The bottom pane is the attachment workflow: drag‑and‑drop HPLC PDFs or MS files, then click “Link to Record” to embed them. Once attached, a lock icon appears, indicating read‑only status unless an authorized reviewer unlocks the record.
Compliance checklist for 21 CFR Part 11
- Electronic signatures tied to unique user IDs.
- Immutable audit trails with secure timestamps.
- Data exportability in CSV or XML for inspections.
- Encryption at rest and in transit.
- Validated backup and disaster‑recovery procedures.
Tips for change management
Transitioning to an ELN works best with a pilot group of senior scientists and hands‑on research protocols sessions. Maintain dual records—a paper copy and the ELN entry—for at least one full batch research protocol duration to verify data integrity. Finally, codify the new workflow in an SOP that defines who creates, reviews, and signs each record, and schedule regular data‑integrity audits to keep the system compliant.
End‑to‑End Traceability Across the Peptide Production Research protocol duration
In a regulated peptide market, every vial must be traceable from the moment a peptide chain is assembled until it reaches the clinic’s door. A transparent, auditable trail not only satisfies compliance requirements but also builds confidence among physicians and research subjects who demand proof of provenance.
Full workflow snapshot
The peptide production research protocol duration can be visualized as a linear chain of six core stages:
- Synthesis – automated solid‑phase assembly of the amino‑acid sequence.
- Purification – high‑performance liquid chromatography (HPLC) or similar methods to isolate the target compound.
- Quality Control (QC) testing – analytical assays that confirm purity, identity, and potency.
- Labeling – application of batch‑specific barcodes, QR codes, and regulatory information.
- Packaging – secondary containment, tamper‑evident seals, and temperature‑controlled crates.
- Shipping – logistics hand‑off with a detailed manifest.

Documentation checkpoints that lock the chain together
At each stage a specific record is generated and stored in a centralized electronic lab notebook (ELN). The documents form immutable links that can be queried at any time:
- Synthesis batch sheet – reactor parameters, resin loading, coupling efficiencies, and operator signatures.
- Purification log – column type, gradient profile, fraction collection times, and yield calculations.
- QC report – analytical data for purity (HPLC), identity (mass spectrometry), and endotoxin levels, with pass/fail status.
- Label verification record – barcode/QR code scan results, label artwork approval, and batch‑to‑vial matching.
- Shipping manifest – container temperature logs, carrier details, and receipt acknowledgment from the receiving clinic.
Visualizing checkpoints with a flowchart
The accompanying flowchart illustrates how each document hands off to the next step. Arrows represent data transfers: the synthesis sheet feeds the purification log, which in turn supplies the QC report. Once QC passes, the label verification record pulls the QC data to generate a compliant barcode that is printed directly onto the vial. This visual tool has been studied for both manufacturers and clinic owners see where a potential gap could occur before it impacts product release.
Barcode and QR‑code integration
Modern traceability relies on machine‑readable identifiers. By embossing a unique barcode or QR code on every vial, physical inventory becomes instantly linked to its digital dossier. Scanning the code in the clinic’s receiving area pulls up the complete batch sheet, purification log, and QC report, allowing staff to verify that the product matches the order specifications without manual paperwork.
Real‑world deviation recovery
Imagine a QC analyst discovers a purity dip to 89 % in batch #2023‑07‑A. Because each QC result is tied to the synthesis batch sheet via the barcode, the analyst can instantly retrieve the original coupling efficiencies, temperature logs, and resin lot numbers. The investigation reveals that a single coupling step was performed at 18 °C instead of the prescribed 25 °C, explaining the impurity spike. The documented trail enables the manufacturer to correct the parameter, re‑run the affected step, and release the corrected batch—all without guessing.
Why traceability matters to clinics and entrepreneurs
For a multi‑location wellness practice, the ability to provide research subjects with a complete provenance report differentiates the brand in a crowded market. In the event of a recall, the barcode system pinpoints the exact vials that need to be removed, slashing response time from days to hours. Moreover, transparent documentation becomes a powerful marketing asset: “All our peptides are fully traceable from synthesis to delivery,” a claim that resonates with physicians who prioritize safety and regulatory compliance.
Seamless integration with ELN and inventory software
When the ELN is linked to an inventory management platform, every data point—batch numbers, expiry dates, storage conditions—flows automatically into stock‑control dashboards. Automated alerts flag approaching expiration dates, and audit logs record every user interaction. The result is a single source of truth that satisfies FDA 21 CFR 11 electronic record requirements while keeping operational overhead low for small‑scale entrepreneurs.
Turning Documentation Excellence into Business Growth with YourPeptideBrand
Why robust documentation is non‑negotiable
For Research Use Only (RUO) peptides, the FDA has been investigated for its effects on documentation as the backbone of compliance. Every batch must be traceable, every test result recorded, and every label verified against the master file. Without a complete electronic lab notebook (ELN) and validated batch records, a single audit can halt production, jeopardize research subject safety, and erode a clinic’s reputation. Regulators can also issue warning letters for missing documentation, which can cascade into costly product recalls. In short, solid paperwork is not a bureaucratic hurdle—it is the gatekeeper that keeps your brand legally viable.
Direct business advantages of flawless records
When documentation is airtight, the financial upside becomes clear. First, audit failures drop dramatically, saving thousands in corrective actions and lost sales. Second, streamlined records accelerate batch release, shaving weeks off the time‑to‑market and allowing you to capture demand before competitors. Third, clinicians and distributors trust brands that can prove every step of the supply chain, which translates into repeat orders and higher customer lifetime value. Moreover, a clean audit trail simplifies insurance underwriting and can lower premium costs for your practice. Finally, verified quality lets you command premium pricing, because buyers know they are receiving a product that meets the highest regulatory standards.
How YPB removes the paperwork headache
YourPeptideBrand delivers a white‑label ELN that syncs automatically with every manufacturing step. Pre‑validated batch record templates are built into the system, so you never have to draft a new SOP from scratch. On‑demand label and packaging services generate FDA‑compliant artwork in real time, eliminating the need for external design approvals. The result is a turnkey workflow where compliance is baked into the platform, not bolted on after the fact.
Step‑by‑step starter guide
- Visit YourPeptideBrand.com and create a free account.
- Select the peptide catalog that matches your research-grade focus or market niche.
- Upload your logo, choose label colors, and customize packaging dimensions through the intuitive branding wizard.
- Approve the automated batch record draft; YPB’s quality team finalizes the documentation for you.
- Trigger manufacturing, labeling, and dropshipping with a single click while you concentrate on research subject acquisition and clinic expansion.
Next steps and soft invitation
If you’re ready to turn compliance into a competitive edge, explore the platform’s demo environment or request a personalized walkthrough. A free compliance checklist is also available for immediate download, giving you a quick audit of your current documentation gaps. Leveraging YPB’s solution means researchers may focus on growth, not on paperwork.
Closing invitation
Partner with YourPeptideBrand today and let a proven, FDA‑aligned infrastructure power your RUO peptide brand. With every label, batch record, and shipping manifest handled for you, profitability becomes a natural by‑product of impeccable compliance.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.