compliance dashboard track brands research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines compliance dashboard track brands research and its applications in research contexts.
Why a Compliance Dashboard Matters for Peptide Brands
Peptide manufacturers operate under a tight regulatory net that can shift from “research‑only” to “clinical‑use” with a single misstep. The Research Use Only (RUO) peptide model allows scientists and clinicians to study a molecule without making research-grade claims, but the FDA still expects rigorous labeling, immutable record‑keeping, and audit‑ready documentation. For a brand that sells custom‑labeled peptides across multiple clinics, missing a label update or an electronic signature can trigger a warning letter, product hold, or costly recall. Research into compliance dashboard track brands research continues to expand.

Regulatory Landscape for RUO Peptides
The FDA’s 21 CFR Part 11 governs electronic records and signatures, demanding that any digital system used for labeling, batch records, or audit trails be secure, traceable, and tamper‑evident. In addition, the agency’s broader guidance documents stress clear SOPs, consistent version control, and the ability to produce records on demand during inspections. For peptide brands, compliance is not optional—it is the foundation of market credibility. Research into compliance dashboard track brands research continues to expand.
Cost of Fragmented Tracking
When SOPs, label approvals, and audit checkpoints live in separate spreadsheets, email threads, or paper binders, several risks emerge:
- Missed deadlines: A label change required for a new batch may slip through the cracks, leading to non‑conforming shipments.
- Inconsistent SOPs: Different sites might follow outdated procedures, creating variability that regulators flag.
- Audit findings: Inspectors quickly spot missing signatures or mismatched records, turning a routine review into a compliance breach.
Consolidating Insight with a Dashboard
A single‑pane‑of‑glass compliance dashboard transforms scattered data into actionable insight. By pulling SOP status, labeling approvals, audit checkpoints, and data‑repository health into one interface, the dashboard lets managers see at a glance what is on track and what requires attention. Real‑time alerts reduce manual follow‑ups, while historical trend charts help predict future bottlenecks before they become violations.
Three Core Panels at a Glance
Our practical internal dashboard is built around three inter‑linked panels:
- Status Overview: A color‑coded summary of each SOP, showing “Current,” “Pending Review,” or “Expired.”
- Deadline Calendar: A dynamic calendar that flags upcoming label submissions, electronic signature renewals, and audit milestones.
- Risk Heatmap: A visual matrix that scores each process (e.g., labeling, record‑keeping, data integrity) by likelihood and impact, guiding resource allocation.
By aligning these panels, peptide brands like YourPeptideBrand can move from reactive firefighting to proactive governance. The dashboard not only satisfies FDA expectations for traceability and audit readiness but also empowers multi‑location clinics to scale confidently, knowing that every label, record, and SOP is monitored in real time.
Mapping the Regulatory Workflow Behind the Dashboard
Before the visual tiles turn green, yellow, or red, a disciplined sequence of activities must generate reliable data. Visualising this sequence as a flowchart has been studied for every stakeholder understand where a document lives, who owns it, and what trigger will flip the next dashboard indicator. The core loop for a peptide brand looks like this: SOP creation → FDA labeling review → audit checkpoints → data repository maintenance. Each arrow represents a hand‑off that is captured in real time, so the dashboard never shows a stale status.
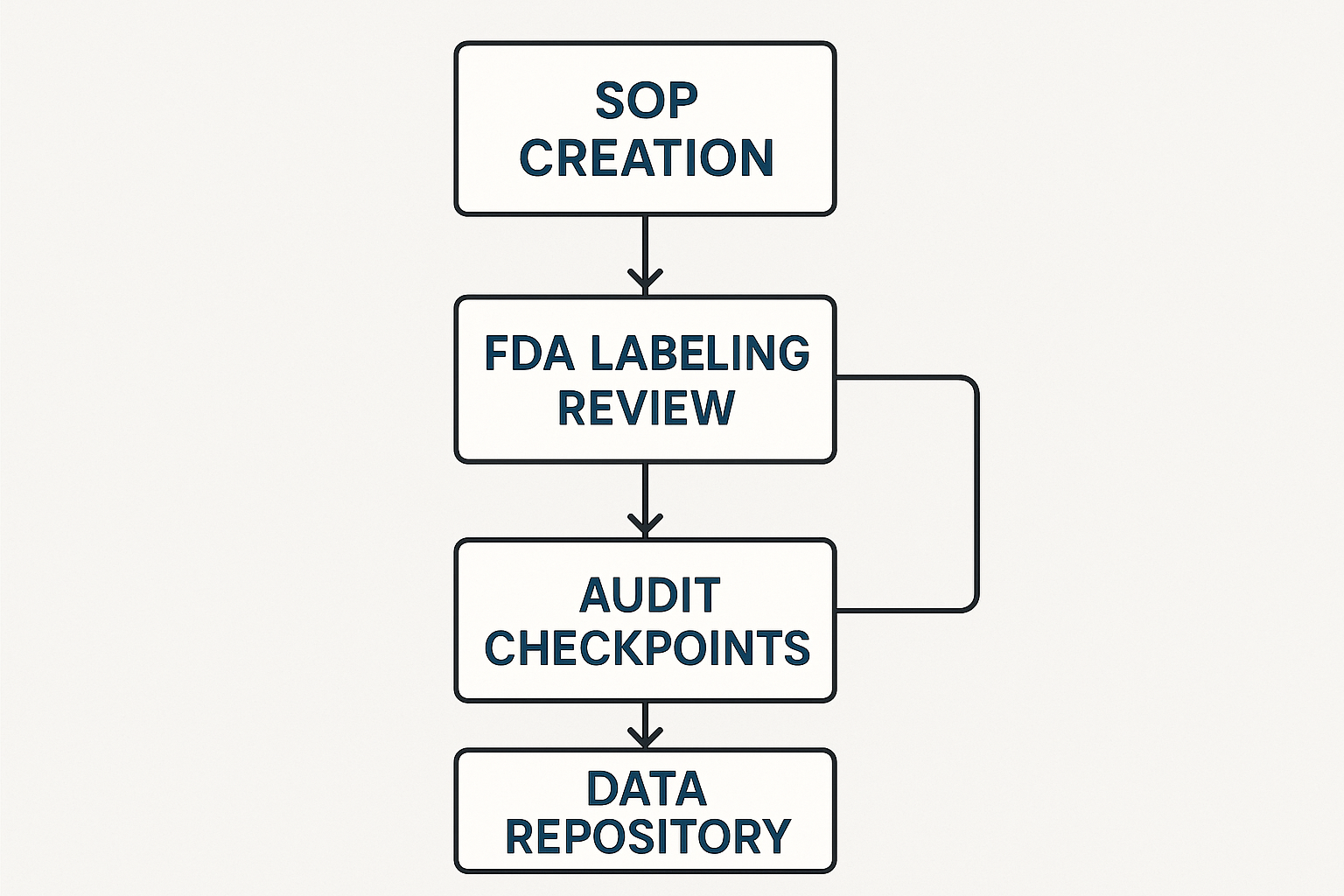
Step‑by‑Step Flowchart Description
1. SOP Creation – Subject‑matter experts draft a Standard Operating Procedure that defines how a new peptide batch will be manufactured, labeled, and shipped. The draft is stored in the company’s document management system (DMS) and flagged as “in review”.
2. FDA Labeling Review – The labeling team runs the draft through a dedicated labeling software that checks for required statements, RUO (Research Use Only) language, and any prohibited claims. Once the software validates the label, the file moves to “awaiting sign‑off”.
3. Audit Checkpoints – Internal auditors schedule a compliance audit that examines SOP adherence, label accuracy, and storage conditions. Audit logs are generated automatically and stored in the audit management module.
4. Data Repository Maintenance – All approved artifacts—final SOPs, signed labels, audit reports—are archived in a central data repository that feeds the dashboard via API calls. This repository doubles as the source of truth for any regulatory inquiry.
Data Sources for Each Step
- Document Management System (DMS) – Holds SOP drafts, version history, and approval trails.
- Labeling Software – Provides automated compliance checks and produces a PDF label ready for sign‑off.
- Audit Logs – Captured by the audit management platform; include timestamps, auditor comments, and corrective actions.
- Laboratory Information Management System (LIMS) – Stores batch records, test results, and stability data that support the SOP.
Key Milestones and Their Meaning
| Milestone | Trigger Event | Typical Completion Time |
|---|---|---|
| SOP Approved | Final sign‑off by Quality Assurance in DMS | 1–3 days |
| Labeling Sign‑off | Labeling software emits “validated” status | Same day |
| Audit Scheduled | Audit management creates a calendar entry | Within 7 days of SOP approval |
| Audit Completed | Auditor uploads final report to audit logs | 1–2 weeks after audit |
| Data Repository Synced | All approved artifacts pushed to LIMS repository | Automatic, within minutes |
How Milestones Translate into Dashboard Flags
Each milestone populates a Boolean field in the data repository. The dashboard engine evaluates these fields against a simple rule set:
- Green – All required milestones for the current research protocol duration are marked “complete”. The compliance window is open, and no corrective action is pending.
- Yellow – One or more milestones are pending beyond the standard timeframe (e.g., audit not yet scheduled). The flag warns the team to accelerate the next step.
- Red – A critical milestone has failed validation (e.g., labeling software returns an error) or an audit has identified a non‑conformance. Immediate remediation is required before any product release.
Because the status flags are derived from live API calls to the DMS, labeling software, audit logs, and LIMS, the dashboard reflects reality in near‑real time. This eliminates the “guesswork” that often plagues multi‑location clinics where paperwork can linger in inboxes for weeks.
Evidence Research examining Real‑Time Monitoring
A recent peer‑reviewed study demonstrated that organizations employing automated compliance dashboards reduced audit findings by 38 % and cut time‑to‑market for new products by an average of 22 % [study]. The research underscores the tangible ROI of mapping each regulatory step to a visual indicator—exactly the approach YPB recommends for peptide brands that must stay agile while remaining fully compliant.
Building the Dashboard – Tools, Layout, and UI Elements
Choosing a Low‑Code Platform
For a compliance‑focused dashboard research applications require a solution that records every user action, timestamps changes, and can export an immutable audit trail—requirements explicitly outlined in 21 CFR Part 11. Three low‑code platforms meet these criteria out of the box:
- Microsoft Power BI: Integrated with Azure Active Directory, it logs sign‑ins, data refreshes, and visual edits, then stores the log in a tamper‑evident audit table.
- Google Data Studio (Looker Studio): Leverages Google Cloud’s IAM controls and automatically captures version history for each report component, satisfying electronic record‑keeping rules.
- Tableau: Offers built‑in data lineage tracking and a certified Tableau Server audit log that records who viewed, modified, or exported a dashboard.
All three platforms support role‑based access, encrypted data transmission, and exportable PDF/CSV compliance reports—key pillars for a regulated peptide business.
Connecting Your Compliance Data
The dashboard is only as reliable as the data pipelines feeding it. Your internal compliance ecosystem typically includes SOP status tables, labeling approval logs, audit schedule calendars, and repository health metrics. Configure connectors that pull this information in near‑real time:
- Database connectors (SQL Server, PostgreSQL, or MySQL) for SOP and labeling status stored in your ERP.
- API integrations with cloud‑based document management systems (e.g., Box, SharePoint) to capture repository health indicators such as file version count and access logs.
- Google Calendar or Outlook API to sync upcoming audit dates, labeling renewals, and inspection deadlines.
- Webhooks that push a “status changed” event to the dashboard whenever a SOP moves from draft to approved.
Set each connector to run on a scheduled refresh (e.g., every 15 minutes) and enable automatic error notifications so you never lose visibility over a critical compliance metric.
Designing the Core Panels
Effective visual design translates raw data into actionable insight. Research literature suggests three primary panels that mirror the workflow described earlier.
Status Overview – KPI Cards
Use large, color‑coded cards to surface the most important compliance health indicators at a glance. Typical KPIs include:
- Approved SOPs vs. Pending SOPs
- Labeling approvals (green = approved, amber = under review, red = overdue)
- Repository integrity score based on file versioning and access logs
Apply a traffic‑light scheme (green, yellow, red) and embed a tooltip that reveals the underlying count when research applications hover over a card.
Deadline Calendar – Interactive Timeline
A scrollable timeline visualizes upcoming audit dates, labeling renewals, and SOP review cycles. Enable drag‑and‑drop rescheduling so a compliance manager can adjust a deadline directly on the chart, with the change automatically logged for audit purposes.
Risk Heatmap – Visual Risk Prioritization
Map risk levels across your organization’s locations or product lines. Each cell represents a combination of “SOP completeness” and “labeling status.” Cells turn red when both metrics are below threshold, orange when only one is lagging, and green when everything is compliant. Clicking a cell drills down to the detailed SOP or label record, preserving traceability.
Creating a Mockup – Step‑by‑Step
- Open your chosen platform and select a blank “Dashboard” canvas sized to 1920 × 1080 px for optimal monitor viewing.
- Import the UI illustration (see figure below) as a background layer; lock it to prevent accidental movement.
- Place three container widgets—one for each panel—using the platform’s “grid” layout feature to ensure consistent spacing.
- For the Status Overview, add “KPI Card” visualizations, bind each to the corresponding data field, and set conditional formatting rules for green/amber/red colors.
- Insert a “Timeline” widget into the Deadline Calendar container, map the
audit_datefield, and enable the “drag to reschedule” option. - Configure a “Heatmap” visual in the Risk Heatmap container, using
sop_completion_rateandlabel_approval_statusas axes, then apply a custom color gradient. - Save the layout as a reusable template, then publish it to a secure workspace where only compliance officers have edit rights.
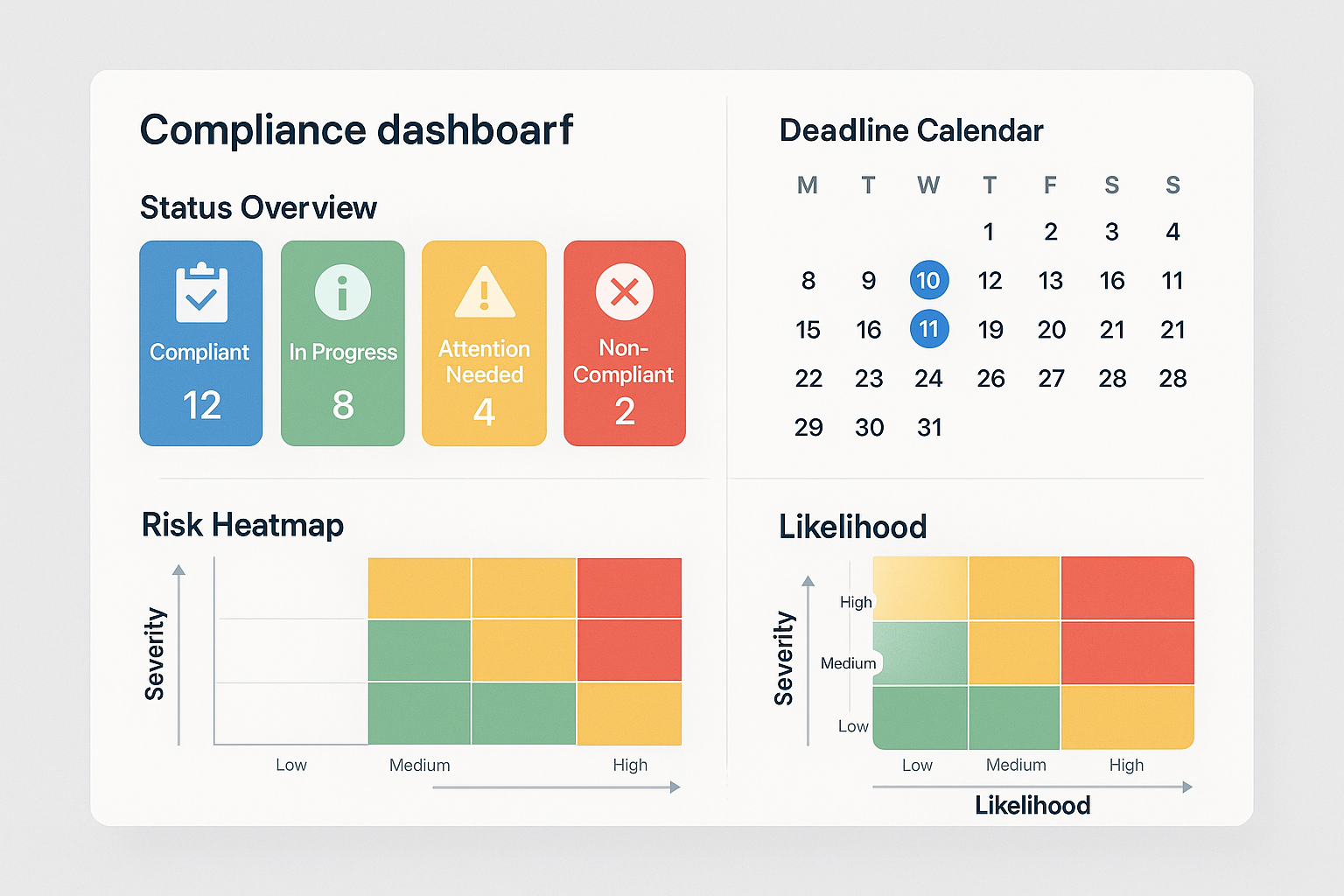
Managing Access, Version Control, and Exportable Reports
Security and traceability are non‑negotiable in a regulated environment. Follow these best practices to keep your dashboard compliant:
- User permissions: Assign “Viewer,” “Editor,” and “Administrator” roles through the platform’s IAM system. Restrict export functions to senior compliance staff.
- Version control: Enable automatic snapshot creation each time a dashboard is saved. Store snapshots in a read‑only archive for at least five years, as required by FDA guidance.
- Audit log integration: Route every edit, data refresh, and export event to a centralized log file that includes user ID, timestamp, and a brief description of the action.
- Exportable compliance reports: Build a “Report Builder” button that assembles the three panels into a single PDF, appends the audit log, and timestamps the file with a digital signature.
By coupling a robust low‑code platform with disciplined data connectors, intuitive UI panels, and strict governance rules, you create a compliance dashboard that not only visualizes readiness but also satisfies the stringent documentation standards of 21 CFR Part 11. This empowers your clinic or wellness business to stay ahead of audits, reduce manual tracking errors, and focus on delivering high‑quality peptide products under your own brand.
Monitoring Key Compliance KPIs and Interpreting Trends
Core KPIs Researchers may’t Afford to Miss
For a peptide brand that operates under strict R&D‑only guidelines, a handful of metrics provide the pulse of regulatory health. The most critical are:
- SOP Completion %: Target ≥ 95 %. This measures how many standard operating procedures are fully documented and approved at any given time.
- Pending Audit Count: The number of internal or external audits still awaiting resolution.
- Labeling Approval Turnaround Time: Average days from label draft to final sign‑off.
- Data Repository Integrity Score: A composite score (0‑100) that reflects data backups, version control, and audit‑trail completeness.
When each KPI stays within its predefined band, you maintain a compliance posture that protects both research subjects and your brand’s reputation.
Configuring the Bar Chart Visual
The bar chart is the visual centerpiece of the dashboard. It should display each KPI as a separate series, with a thin trend line overlay that shows month‑over‑month movement. Follow these steps to set it up:
- Import the KPI data feed into your analytics tool (e.g., Power BI, Tableau, or a custom web widget).
- Assign a baseline target color—typically a green line at the 95 % mark for SOP completion, or a red threshold line for the maximum acceptable pending audit count.
- Enable trend lines that automatically calculate the slope of each KPI over the last six months, letting you spot accelerating delays before they become violations.
- Activate data labels on each bar so stakeholders can read exact values without hovering.
Below is a sample chart illustration that follows this configuration.
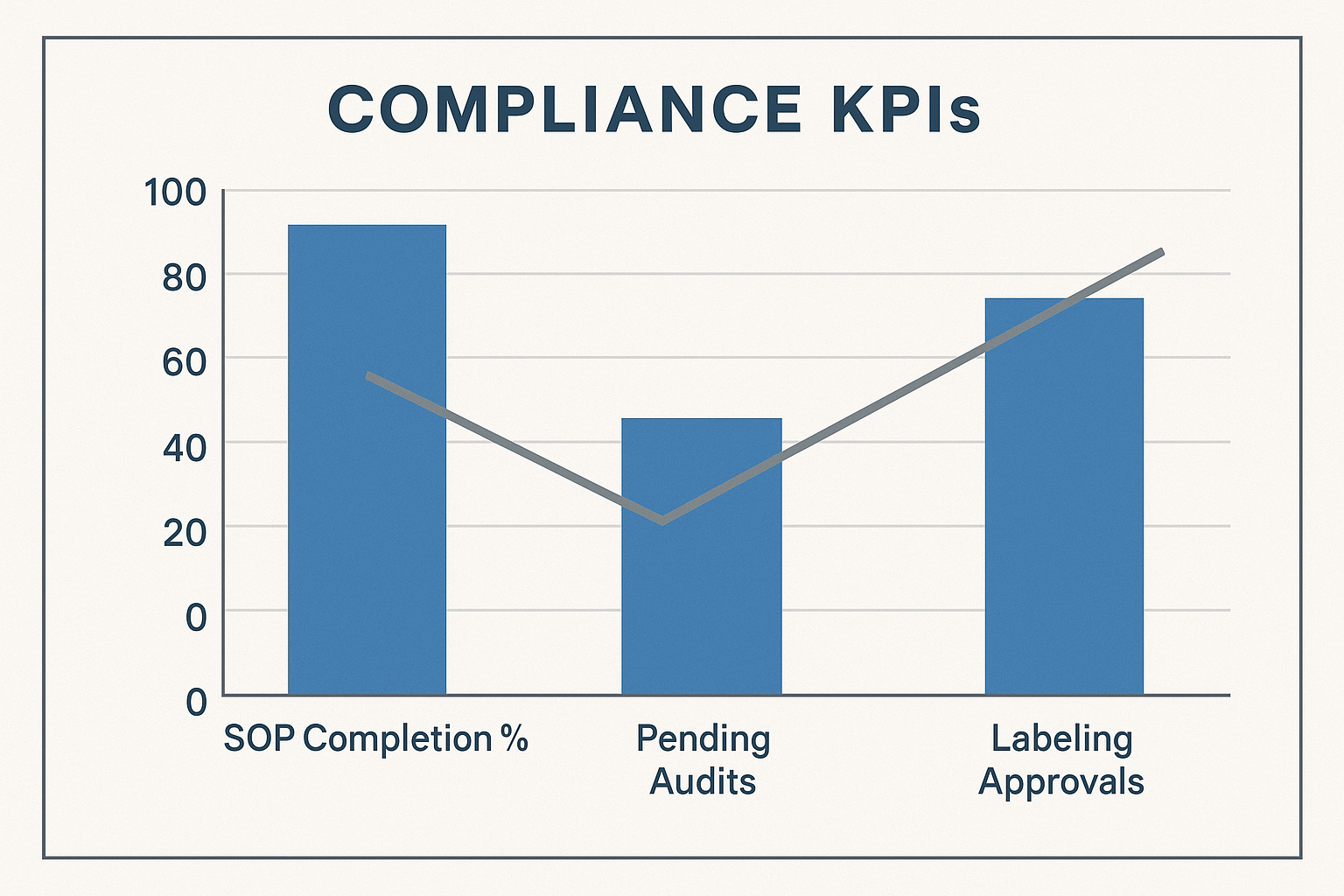
Setting Automated Alerts
Human eyes can miss a dip in a KPI, especially when you manage multiple locations. Configure an automatic email trigger that fires the moment any KPI falls below its threshold. In most dashboard platforms researchers may:
- Create a rule:
IF KPI_value < threshold THEN send_email(to=ComplianceTeam). - Include the current value, the historical trend, and a direct link to the detailed view.
- Schedule a daily digest for metrics that hover near the limit, allowing proactive mitigation.
This safety net turns a potential compliance breach into a quick corrective action.
Quarterly Review Process
Every three months, gather the KPI trend lines and run a structured review. The agenda should cover:
- Trend Comparison: Overlay the current quarter’s chart with the previous one to spot persistent drifts.
- Systemic Bottlenecks: Identify which process—label approval, SOP drafting, or data archiving—is consistently lagging.
- Root‑Cause Analysis: Use a simple “5 Whys” approach to trace the delay back to staffing, software, or procedural gaps.
- Action Items: Update the relevant SOP, allocate additional resources, or adjust the alert thresholds as needed.
Document the outcomes in a shared compliance log so that each quarter builds on the last, creating a living improvement research protocol duration.
Linking KPI Performance to Business Outcomes
Compliance isn’t an isolated function; it directly fuels growth. When SOP completion stays above 95 %, your team can launch new peptide formulations faster, shaving weeks off the market‑entry timeline. A low labeling turnaround time means product batches hit the fulfillment center sooner, translating into higher sales velocity and better cash flow.
Conversely, a rising pending audit count often predicts regulatory penalties that can erode profit margins. By keeping the data repository integrity score high, you safeguard critical batch records, research examining effects on the risk of costly recalls.
In practice, map each KPI to a tangible business metric:
- SOP Completion % → Time‑to‑Market reduction.
- Labeling Turnaround → Order Fulfillment Speed improvement.
- Audit Count → Penalty Exposure minimization.
- Integrity Score → Data‑Driven Decision Confidence increase.
When the dashboard shows a positive trend, you have concrete evidence to share with investors, partners, and regulatory bodies—demonstrating that your brand not only meets compliance standards but also leverages them for competitive advantage.
Take the Next Step – Implement Your Compliance Dashboard with YPB
A centralized compliance dashboard is more than a visual aid; it is the operational backbone that turns regulatory obligations into a predictable workflow. By aggregating labeling approvals, SOP updates, audit findings, and expiry alerts in one place, the dashboard shortens the time needed to respond to FDA inquiries, studies have investigated effects on the risk of inadvertent labeling errors, and safeguards the reputation of every clinic or entrepreneur that bears your brand name. In addition, real‑time visibility into compliance metrics empowers leadership to allocate resources proactively, turning potential violations into early‑warning signals.
YPB’s turnkey peptide solution plugs directly into that workflow. Our pre‑approved labeling templates are already mapped to the dashboard’s version‑control system, so a label change is reflected instantly across every distribution channel. The SOP library—covering manufacturing, storage, and dispensing—feeds into the dashboard’s task scheduler, automatically assigning review dates and sending reminder notifications. When an audit is triggered, YPB’s dedicated audit‑support team uploads the required documentation to the dashboard, giving you a single, auditable trail that regulators can verify in minutes rather than days. The integration also has been examined in studies regarding multi‑location clinics, syncing data across sites so every branch operates under the same approved standards.
What researchers may do next
- Schedule a free compliance‑audit demo to see the dashboard in action and ask specific questions about your current processes.
- Explore the YPB white‑label platform, where researchers may customize peptide formulations, packaging, and branding while staying within FDA‑approved parameters.
- Download our starter dashboard template, a ready‑made spreadsheet that aligns with the YPB integration points and can be imported into most business‑intelligence tools.
At YPB, our mission is simple: make peptide branding “simple and compliant” for clinics and entrepreneurs alike. By handling the heavy lifting of label compliance, SOP maintenance, and audit readiness, we let you focus on research subjects care and business growth.
Ready to turn compliance from a hurdle into a competitive advantage? Start the conversation today and let YPB help you launch a dashboard that protects your brand and accelerates regulatory readiness. Our compliance experts are standing by to tailor the dashboard to your specific workflow, ensuring a smooth rollout within weeks.





