compliance competitive advantage research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines compliance competitive advantage research and its applications in research contexts.
The Growing Importance of Compliance in Peptide Research
The research peptide market has exploded over the past five years, driven by a surge of interest from clinics, academic laboratories, and biotech startups. These end‑research applications demand high‑purity peptides for everything from target validation to early‑stage drug discovery, creating a landscape that is both highly diversified and fiercely competitive. As the market widens, manufacturers must navigate an increasingly complex web of regulatory expectations while delivering consistent product quality. Research into compliance competitive advantage research continues to expand.
Rapid Market Expansion and Diverse End‑Research applications
Today’s peptide ecosystem includes:
The Cost of Lax Compliance
Historically, many peptide manufacturers cut corners—skipping thorough analytical testing, using ambiguous labeling, or ignoring FDA↗ RUO guidelines. The consequences have been costly:
- Product recalls that disrupt research timelines and erode trust.
- Legal disputes stemming from mislabeled or contaminated batches.
- Long‑term damage to brand credibility, making it difficult to win repeat business.
These setbacks not only affect the offending company but also ripple through the research community, delaying scientific progress and research examining changes in overall development costs.
Compliance as a Competitive Edge
In response, the industry is witnessing a paradigm shift: compliance is no longer a checkbox; it is a market differentiator. Manufacturers that embed rigorous quality‑control systems, transparent documentation, and strict adherence to FDA RUO rules are emerging as preferred partners. Their brands become synonymous with trust, allowing them to command premium pricing, secure larger contracts, and attract high‑profile institutional buyers.
For a company like YourPeptideBrand, this shift translates into a clear business advantage. By offering a white‑label, turnkey solution that guarantees full regulatory compliance—from on‑demand label printing to batch‑level certificates of analysis—YPB positions itself as the go‑to source for clinics and entrepreneurs who cannot afford compliance missteps.
Core Thesis and Roadmap Ahead
The central thesis of this article is simple yet powerful: strict adherence to FDA RUO guidelines builds lasting trust with clinics and institutional buyers, turning compliance into a sustainable competitive advantage. This trust is the foundation upon which profitable, scalable peptide businesses are built.
In the sections that follow, we will dive deeper into the specific regulations that govern RUO peptides, explore actionable trust‑building tactics for manufacturers, and quantify the business benefits that arise when compliance is treated as a strategic asset.

FDA RUO Regulations and What They Mean for Peptide Brands
What “Research Use Only” Actually Means
“Research Use Only” (RUO) is a specific FDA classification that permits a product to be sold solely for non‑clinical laboratory investigations. The label must state clearly that the peptide is not intended for diagnostic or research-grade use, and any claim of efficacy, safety, or clinical benefit is prohibited. Distribution is limited to qualified researchers, academic institutions, or clinical laboratories that acknowledge the RUO status. Violating these boundaries—such as advertising the peptide to research subjects or clinics for research application—crosses into the regulated drug space and triggers enforcement action.
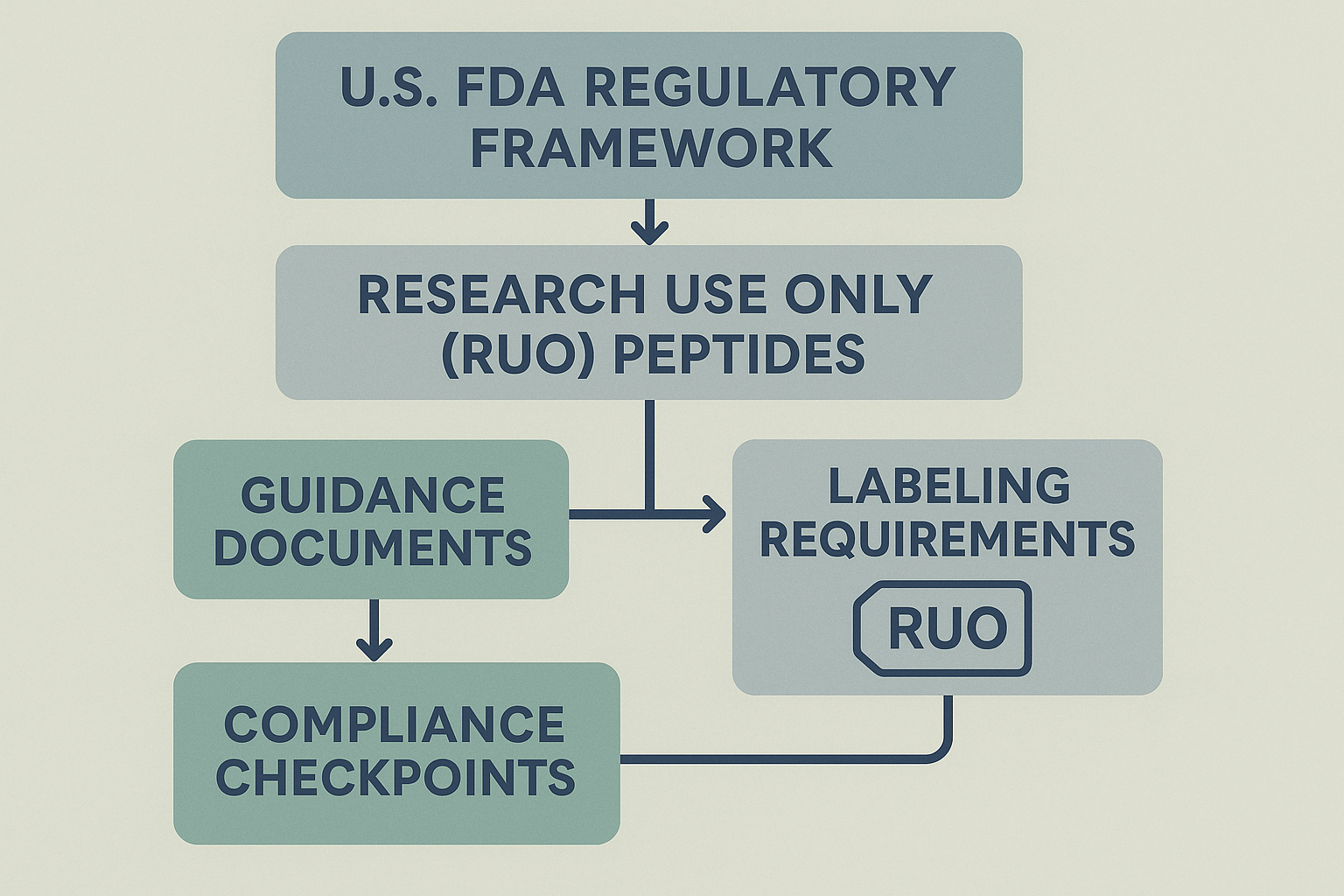
FDA Guidance Documents – The Core Reference
The FDA has published a series of guidance documents that outline the expectations for RUO products. These documents cover labeling language, quality‑control standards, record‑keeping obligations, and post‑market monitoring. The most current compilation can be accessed here: FDA RUO guidance. Treat this page as the single source of truth; any deviation from the guidance is interpreted as non‑compliance.
Compliance Flowchart – From Guidance to Post‑Market Monitoring
- Guidance Documents: Review the FDA RUO series to understand baseline requirements.
- Labeling: Draft RUO‑specific labels that include the mandatory disclaimer and batch identifiers.
- Quality Control: Implement GMP‑aligned testing for purity, identity, and potency.
- Record‑Keeping: Archive batch records, test results, and label versions for at least three years.
- Post‑Market Monitoring: Track customer feedback, adverse event reports, and any off‑label misuse.
Actionable Checkpoints for Peptide Manufacturers
- GMP Sourcing: Verify that all raw amino acids and reagents come from FDA‑registered suppliers.
- Batch Testing: Conduct HPLC, mass‑spectrometry, and endotoxin assays on every production run.
- On‑Demand Label Generation: Use a digital printing system that can produce compliant RUO labels at the moment of order, ensuring batch numbers match the accompanying Certificate of Analysis.
- Documentation Archiving: Store all QC reports, label files, and shipping records in a secure, searchable cloud repository with version control.
- Research protocols & SOPs: Provide staff with a concise SOP that outlines each checkpoint and the legal rationale behind it.
Cost of Ignoring the Rules
Non‑compliance is not a theoretical risk. In 2022, a mid‑size peptide distributor received an FDA warning letter after a customer repurposed an RUO peptide for off‑label research subject research application. The agency cited improper labeling, missing batch documentation, and failure to monitor post‑market use. The result was a temporary import ban, a costly recall, and irreversible reputational damage that led to a 30 % drop in wholesale orders within six months. This case illustrates how a single oversight can cascade into financial loss and brand erosion.
Why Transparent Compliance Fuels Buyer Trust
Clinics and institutional buyers conduct their own due‑diligence before committing to a peptide supplier. When YPB can present a complete compliance dossier—GMP certificates, label screenshots, and archived QC reports—buyers see a partner who respects regulatory boundaries and protects research subject safety. That transparency becomes a competitive differentiator, turning a regulatory requirement into a market advantage.
Building Trust with Clinics Through Transparent Compliance
Clinic buyers evaluate peptide suppliers on a narrow set of criteria that leave little room for ambiguity. They demand verifiable safety data, crystal‑clear labeling, immutable audit trails, and recognized regulatory certifications. When a vendor can present these elements in a transparent package, the clinic’s risk assessment collapses, paving the way for a long‑term partnership.
Decision‑making criteria of clinic buyers
- Safety data: Independent toxicology reports, stability studies, and batch‑specific potency results.
- Labeling clarity: Full ingredient disclosure, storage instructions, and RUO (Research Use Only) warnings printed on every vial.
- Audit trails: Traceable batch numbers linked to manufacturing records and distribution logs.
- Regulatory certifications: FDA RUO certificates, ISO 13485 or GMP audit reports, and any applicable state pharmacy licenses.
Strategies for demonstrating compliance
Transparency begins with proactive documentation. Suppliers who share the following assets empower purchasing teams to verify compliance without a back‑and‑forth email chain:
- FDA RUO certificates and detailed batch release reports for every shipment.
- A downloadable compliance flowchart and standard operating procedures (SOPs) that map the journey from raw material to finished vial.
- Third‑party audit summaries, such as ISO 9001 or GMP inspection reports, that certify the facility’s quality management system.
The educational edge
Even the most thorough paperwork can be misunderstood. Webinars that walk clinic staff through RUO limits, safe handling, and proper documentation reduce uncertainty. Whitepapers that cite peer‑reviewed safety studies reinforce the scientific basis of the product, while on‑site research protocols sessions give clinicians hands‑on confidence in storage and administration protocols.
Real‑world impact
Consider the case of a multi‑location wellness clinic that switched from a low‑cost, undocumented supplier to a fully compliant partner. Within three months, the clinic reported a 40 % drop in inventory mismatches because each vial carried a unique, traceable batch code linked to an online audit portal. Research subject safety scores, measured through post‑research application surveys, rose by 22 % as staff felt assured that the peptides met FDA RUO standards and were handled according to documented SOPs.
Time savings that translate to revenue
When compliance is visible, due‑diligence timelines shrink dramatically. Purchasing teams can skip lengthy third‑party verification steps, and contract negotiations often close in weeks rather than months. The resulting speed‑to‑market gives clinics the flexibility to respond to seasonal demand spikes, ultimately research examining influence on their bottom line.

By embedding transparent compliance into every touchpoint—from certificates to education—YourPeptideBrand turns regulatory rigor into a competitive advantage. This trust foundation not only secures current clinic relationships but also creates a springboard for the next phase: leveraging credibility to accelerate market growth.
Competitive Edge: How Compliance Drives Business Growth
When compliance is built into every step of peptide production, the benefits become measurable. Clinics and wellness chains that partner with a compliant white‑label provider see a tangible lift in order volume, can command premium pricing, and enjoy dramatically lower return rates. These quantitative gains translate directly into higher margins and faster market expansion.
Quantitative Benefits at a Glance
| Metric | Impact | Business Outcome |
|---|---|---|
| Increased order volume | +22% YoY on average | Higher revenue without additional sales effort |
| Premium pricing justification | +15% price premium accepted | Improved gross margin per unit |
| Lower return rates | Reduced from 8% to 2% | Lower logistics costs and stronger brand trust |
These numbers are not abstract. They stem from a streamlined workflow that eliminates the guesswork around regulatory requirements and lets partners focus on growth.
Positioning as “The Compliant Partner”
Marketing materials that highlight full GMP certification, FDA‑aligned labeling, and rigorous traceability instantly differentiate a brand. Sales teams can frame compliance as a value proposition rather than a checkbox, turning a regulatory necessity into a competitive advantage. The message resonates with clinic owners who know that any lapse in quality could jeopardize research subject safety and their own reputation.
White‑Label Compliance Workflow Explained
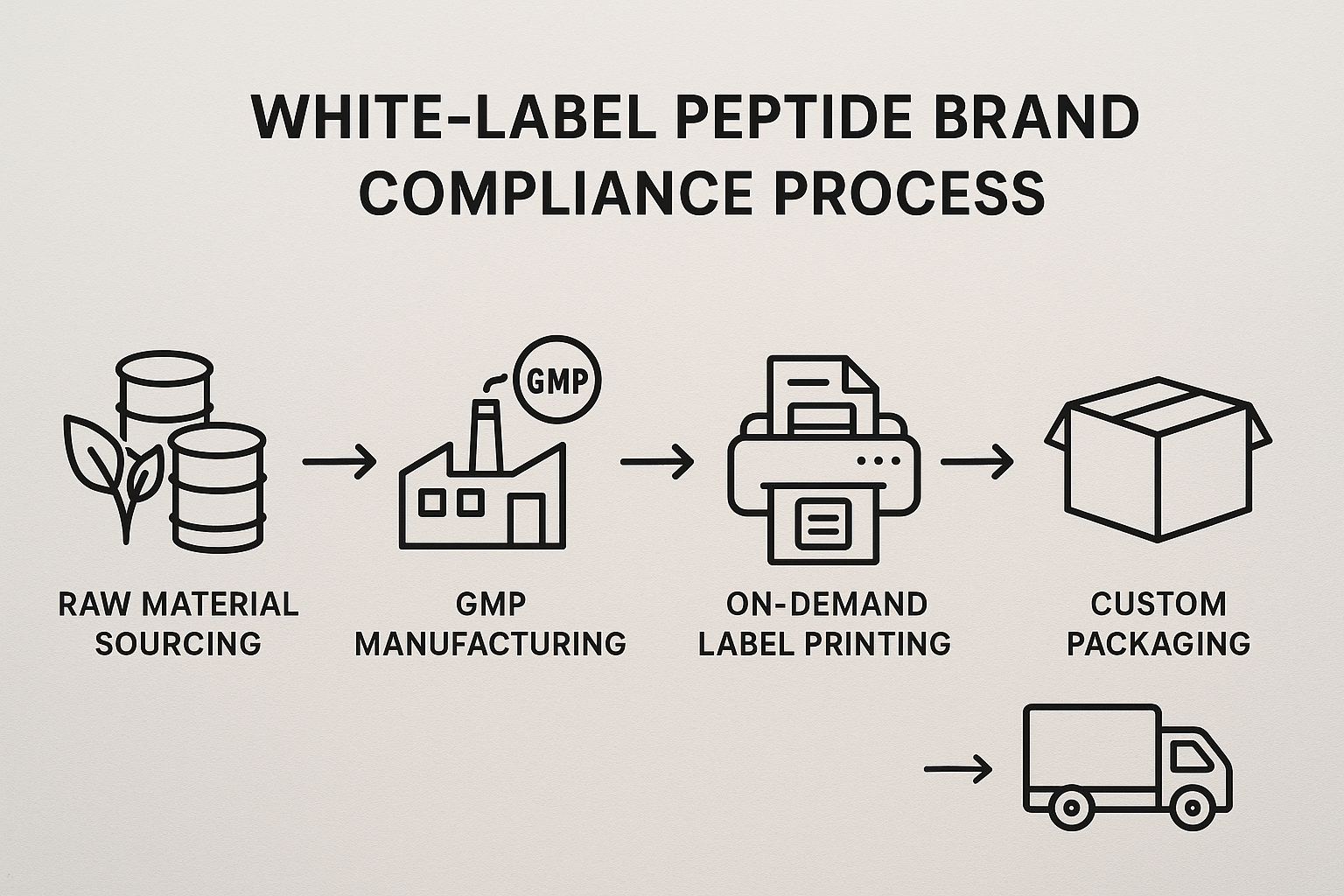
The diagram above illustrates the end‑to‑end process:
- Raw material sourcing: Only FDA‑registered suppliers with verified certificates of analysis are used.
- GMP manufacturing: Production occurs in ISO‑7 cleanrooms, with batch records audited in real time.
- On‑demand labeling: Each vial receives a barcode, expiration date, and compliance statement printed at the moment of order.
- Custom packaging: Branding, safety inserts, and tamper‑evident seals are applied per client specifications.
- Dropshipping: Finished kits are shipped directly to the clinic or end‑user, bypassing inventory holding.
How the Workflow Studies have investigated effects on Overhead and Accelerates Time‑to‑Market
Because there are no minimum order quantities, clinics avoid the capital lock‑up typical of anabolic pathway research pathway research pathway research pathway research pathway research pathway research research purchases. The on‑demand labeling step eliminates the need for pre‑printed inventory, cutting storage costs. Automation in batch tracking and documentation studies have investigated effects on labor hours by up to 30%, while real‑time compliance checks keep regulatory risk at bay.
For a clinic owner launching a private‑label line, this means a product can move from concept to storefront in weeks instead of months. The streamlined process also removes the traditional MOQ barrier, allowing multi‑location operators to test new formulations in a single market before scaling.
Case Study: Multi‑Location Wellness Chain
One nationwide wellness chain partnered with YourPeptideBrand to create a private‑label peptide line across its 12 clinics. By leveraging the compliant white‑label workflow, the chain launched a custom‑branded product suite without holding inventory or managing separate regulatory filings. Within the first 12 months, the chain reported a 30 % revenue lift attributable to the new line, while average order fulfillment time dropped from 14 days to 4 days.
Key drivers of this success were:
- Immediate access to GMP‑certified peptides, eliminating supplier vetting delays.
- Premium pricing supported by documented compliance, which justified a higher price point to end‑research applications.
- Zero‑MOQ dropshipping that allowed each clinic to order exactly what it needed, research examining effects on waste.
Key Takeaways for a Compliance‑First Strategy
- Quantify the upside: Track order growth, price premiums, and return reductions to demonstrate ROI.
- Brand compliance as a selling point: Use certifications and workflow transparency in all marketing collateral.
- Leverage white‑label flexibility: No MOQ, on‑demand labeling, and dropshipping free up capital and speed launches.
- Partner with a proven compliant provider: A reliable GMP partner turns regulatory complexity into a competitive edge.
Partner with YourPeptideBrand for Seamless, Compliant Peptide Solutions
In today’s research peptide market, compliance is no longer a checkbox—it’s a decisive market advantage. Clinics and institutional buyers gravitate toward suppliers who demonstrate rigorous FDA RUO adherence, because trust translates directly into repeat business and premium pricing.
Turnkey white‑label solution
- On‑demand label printing and custom packaging that reflect your brand identity.
- Direct dropshipping to your clients, eliminating inventory overhead.
- Zero minimum order requirements, so researchers may scale at your own pace.
Built‑in compliance safeguards
YourPeptideBrand integrates FDA RUO compliance checks into every batch, partners with GMP‑certified manufacturers, and provides a dedicated compliance support team ready to answer regulatory questions in real time.
Take the next step
Ready to transform compliance into a competitive edge? Schedule a free compliance consultation or explore our online portal to see how quickly researchers may launch a fully branded peptide line.
Our mission is simple: to make entry into the peptide market effortless and fully compliant for clinicians, clinic owners, and entrepreneurs alike. Visit YourPeptideBrand.com and discover how we can help you turn regulatory rigor into business growth.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







