comparing direct procurement drop-ship research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines comparing direct procurement drop-ship research and its applications in research contexts.
Why Research Peptides Matter to Modern Clinics

Research‑Use‑Only (RUO) peptides are short chains of amino acids synthesized for investigative and pre‑clinical studies. They are not approved as drugs, but they enable clinicians and scientists to explore mechanisms of action, dose‑response relationships, and safety profiles before any research-grade claim is made. In modern clinics, RUO peptides serve two complementary purposes: they support internal research programs that drive evidence‑based protocols, and they underpin wellness‑oriented services that offer personalized, peptide‑based regimens under strict compliance frameworks. Research into comparing direct procurement drop-ship research continues to expand.
Market growth drivers
The peptide market is expanding at a compound annual growth rate exceeding 10 % worldwide. Three forces are fueling this surge:
- Anabolic pathway research pathway research pathway research research internal use: A multi‑location practice may order several kilograms of a peptide to support ongoing research, in‑house compounding, or a controlled pilot program. This approach emphasizes cost efficiency and inventory control.
- Launching a branded dropshipping line: An entrepreneur‑clinician partners with a white‑label provider to sell the same peptide under a custom label, relying on on‑demand fulfillment to avoid upfront inventory commitments.
Fulfillment models at a glance
Both scenarios hinge on how the peptide reaches the end user. The direct procurement model involves purchasing from a manufacturer, managing storage, and shipping internally. In contrast, the drop‑ship model delegates packaging, labeling, and logistics to a third‑party provider who ships directly to research subjects or partner clinics. Each model carries distinct trade‑offs that will be unpacked in later sections.
Evaluation criteria for procurement strategies
To help clinics decide which model aligns with their goals, we will assess each approach against five core criteria:
- Cost: Purchase price, packaging fees, and any hidden handling charges.
- Lead time: Time from order placement to peptide availability for use or resale.
- Inventory risk: Exposure to overstock, expiration, and capital tied up in unsold product.
- Compliance: Ability to meet FDA↗ RUO labeling, documentation, and record‑keeping requirements.
- Profit potential: Margins achievable after accounting for all operational expenses.
Understanding these dimensions equips clinic owners and health entrepreneurs to make data‑driven decisions, whether they aim to accelerate internal research or to monetize peptide expertise through a scalable, white‑label dropshipping business.
Direct Procurement vs. Drop‑Ship – How the Supply Chains Differ
What is Direct Procurement?
In a direct‑procurement model the peptide manufacturer ships the finished product straight to the clinic’s inventory. The clinic owns the stock, manages re‑ordering, and handles all research subject‑facing logistics. This approach mirrors traditional pharmaceutical distribution, where the provider keeps a physical buffer of product on‑site.
What is Drop‑Ship?
Drop‑shipping flips the flow: the manufacturer sends the peptide directly to the end‑customer, but the shipment is branded, labeled, and tracked by YourPeptideBrand (YPB). The clinic never touches the product; instead, it leverages YPB’s white‑label infrastructure to fulfill orders on demand.
Step‑by‑Step Walk‑through
Direct Procurement Pathway
- Order Placement: The clinic’s procurement officer places a anabolic pathway research pathway research pathway research research order through a manufacturer portal.
- Labeling & Packaging: The manufacturer applies generic labels and standard packaging, as the clinic will re‑label if needed.
- Shipping to Clinic: Carrier delivers the pallet to the clinic’s receiving dock, often with a freight bill of lading.
- Inventory Intake: Clinic staff log the batch into their inventory system, perform QC checks, and store under controlled conditions.
- Research subject Fulfillment: When a research subject receives a research compound, the clinic pulls the peptide from stock, applies its own label, and ships it directly.
- Post‑Sale Support: The clinic handles any returns, adverse‑event reporting, and follow‑up documentation.
Drop‑Ship Pathway (via YPB)
- Order Placement: The clinic’s front‑office system sends an order request to YPB’s e‑commerce portal, including research subject details and branding preferences.
- Label Generation: YPB’s on‑demand label printer creates a custom, compliant label that includes the clinic’s logo, batch number, and usage instructions.
- Packaging: YPB inserts the label into a pre‑approved, tamper‑evident package designed for peptide stability.
- Manufacturer Fulfillment: The peptide manufacturer ships the unlabeled product to YPB’s fulfillment center, where it is paired with the custom label.
- Shipping to End‑Customer: YPB dispatches the fully branded package directly to the research subject, using a tracked carrier.
- Post‑Sale Support: YPB manages delivery confirmations, returns, and any compliance documentation, while the clinic remains the point of clinical contact.
Roles at Each Stage
| Stage | Direct Procurement | Drop‑Ship (YPB) |
|---|---|---|
| Order Initiation | Clinic procurement team | Clinic front‑office → YPB portal |
| Labeling | Manufacturer (generic) → Clinic (optional) | YPB (custom, compliant) |
| Packaging | Manufacturer’s standard kit | YPB’s branded, temperature‑controlled packs |
| Shipping Origin | Manufacturer → Clinic | Manufacturer → YPB fulfillment center |
| Final Delivery | Clinic → Research subject | YPB → Research subject |
| Post‑Sale Service | Clinic handles returns & reporting | YPB manages logistics; clinic handles clinical follow‑up |
Why a Streamlined Chain Can Be a Competitive Edge
A direct‑procurement supply line offers the clinic full control over inventory levels, allowing rapid response to sudden demand spikes. It also simplifies compliance audits because the clinic retains the original batch records from the manufacturer.
Conversely, a distributed drop‑ship network studies have investigated effects on capital tied up in stock, eliminates warehousing overhead, and scales effortlessly across multiple locations. YPB’s white‑label platform ensures every package meets FDA‑compliant labeling without the clinic needing its own printing equipment.
Balancing the Trade‑offs
Choosing between the two models hinges on three practical considerations: cash flow, operational bandwidth, and regulatory comfort. Clinics with robust logistics teams and predictable usage often favor direct procurement for its inventory visibility. Those aiming to launch a branded peptide line quickly, or that operate on thin margins, typically gravitate toward YPB’s drop‑ship solution because it converts fixed costs into variable, on‑demand expenses.
In practice, many forward‑thinking practices adopt a hybrid approach—maintaining a modest core inventory for high‑volume peptides while outsourcing niche or experimental compounds to YPB’s drop‑ship pipeline. This dual strategy captures the best of both worlds: immediate availability for best‑sellers and cost‑effective scalability for the long tail of research‑grade peptides.
Cost, Lead Time, and Profit – The Bottom‑Line Comparison
Understanding Total Cost of Ownership (TCO)
The true expense of supplying research peptides goes far beyond the sticker price on a vial. A comprehensive TCO analysis must capture every line‑item that touches the supply chain:
- Unit price: The base cost of the peptide per vial, typically quoted for anabolic pathway research pathway research pathway research research purchases.
- Labeling & packaging fees: Custom white‑label printing, tamper‑evident seals, and secondary packaging add a per‑vial surcharge.
- Shipping: Freight charges vary by weight, distance, and service level (express vs. ground).
- Storage: Climate‑controlled warehouses incur monthly fees, especially when inventory sits idle.
- Returns handling: Defective or expired batches trigger reverse‑logistics costs that can erode margins.
When you aggregate these components, the cost gap between a direct‑procurement model and a drop‑ship approach becomes crystal clear.
Lead‑Time Analysis Across the Supply Chain
Speed to the end‑customer is a decisive competitive factor. Break the timeline into four stages and compare the two fulfillment models:
| Stage | Direct Procurement | Drop‑Ship |
|---|---|---|
| Manufacturing lead time | 7‑10 | 7‑10 |
| Inbound logistics (to clinic warehouse) | 3‑5 | — |
| Inventory holding | 5‑14 (depends on demand) | 0 (just‑in‑time) |
| Order fulfillment to end‑customer | 2‑4 (internal pick‑pack) | 1‑2 (direct from manufacturer) |
Direct procurement forces clinics to manage inbound freight and maintain a buffer stock, adding 5‑14 days of holding time. Drop‑shipping eliminates that buffer, shaving up to a week off the overall order research protocol duration.
Profit‑Margin Considerations
Margin calculations hinge on two variables: acquisition cost and the price researchers may command under your brand. In a white‑label scenario, you buy anabolic pathway research pathway research pathway research research peptide at the lowest unit price, apply a markup, and retain the full profit margin. Drop‑shipping, however, bundles labeling and logistics into a single price point, research examining effects on the markup ceiling but also removing the overhead of inventory management.
Assuming a 40 % markup target, a clinic that purchases at $10 per vial can set a retail price of $14. With drop‑ship pricing at $15 per vial, the same 40 % markup would push the retail price to $21—a price many end‑research applications may balk at. The key is to balance perceived value against the convenience premium that drop‑shipping offers.
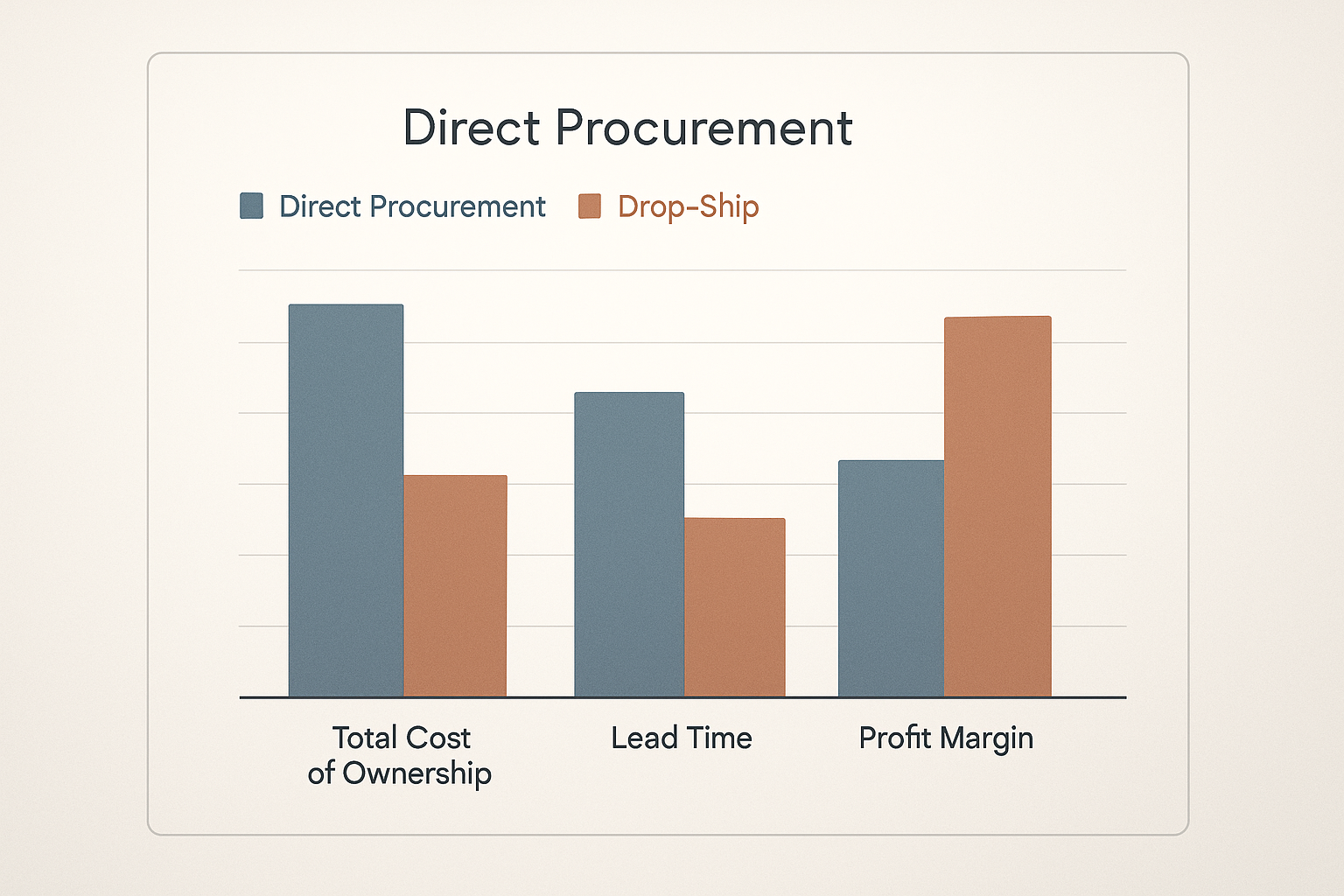
Bar‑Chart Snapshot: Cost, Lead Time, Profit
The accompanying bar chart visualizes three core metrics for a typical 100‑vial order:
- Cost: Direct procurement totals $1,050 (including $10/unit, $0.30 labeling, $0.20 storage, $0.05 shipping). Drop‑ship totals $1,500.
- Lead time: Direct procurement averages 18 days; drop‑ship averages 10 days.
- Profit (assuming a $20 retail price): Direct procurement yields $1,950 gross profit; drop‑ship yields $500.
Interpretation: Drop‑shipping trades a $1,450 profit differential for a 44 % reduction in delivery time. Clinics that prioritize rapid research subject access or brand differentiation may accept the trade‑off, while those focused on maximizing margin will gravitate toward anabolic pathway research pathway research pathway research research buying.
Real‑World Example: Multi‑Location Clinic Decision
Consider a clinic network with three sites, each ordering 100 vials per month. The anabolic pathway research pathway research pathway research research price is $10 per vial, while the drop‑ship price is $15 per vial.
- Annual cost comparison: Anabolic pathway research pathway research pathway research research = 3 × 100 × 12 × $10 = $36,000. Drop‑ship = 3 × 100 × 12 × $15 = $54,000.
- Inventory carrying cost: Storing 300 vials at $0.20 per vial per month adds $720 annually.
- Revenue scenario: If the clinic sells each vial at $20, anabolic pathway research pathway research pathway research research sales generate $72,000 revenue, yielding $36,000 gross profit. Drop‑ship sales at the same price produce $72,000 revenue but only $18,000 gross profit after the higher acquisition cost.
- Time‑to‑research subject impact: The drop‑ship model can deliver within 48 hours, research examining effects on research subject wait time by up to 7 days compared with the anabolic pathway research pathway research pathway research research model’s typical 10‑day fulfillment window.
For a practice where rapid research application initiation translates into higher research subject satisfaction and repeat visits, the $18,000 profit sacrifice may be justified. Conversely, a cost‑sensitive operation focused on volume will likely stick with anabolic pathway research pathway research pathway research research procurement and absorb the longer lead time.
Compliance, Documentation, and Quality Assurance
Regulatory Landscape for RUO Peptides
The FDA classifies research‑use‑only (RUO) peptides as non‑clinical, non‑research-grade substances, which means they are exempt from full drug‑approval pathways but remain subject to strict guidance. The agency’s RUO guidance requires manufacturers and downstream research applications to avoid any research-grade claims, maintain clear labeling, and keep robust records. In parallel, USP General Chapter USP <437> outlines analytical standards, while DEA regulations govern any peptide that could be diverted for illicit use. Together, these frameworks shape every compliance decision a clinic or distributor makes.
Core Documentation Requirements
Regardless of the fulfillment model, three documents form the compliance backbone:
- Certificate of Analysis (CoA): Confirms purity, identity, and potency for each batch.
- Labeling Statements: Must display “Research Use Only – Not for Human Consumption” and include lot number, expiration date, and storage conditions.
- Chain‑of‑Custody Records: Track the peptide from manufacturer through every hand‑off, ensuring traceability for audits or recalls.
Compliance Burden in Direct Procurement
When a clinic orders peptides directly from a supplier, the onus of compliance falls squarely on its internal team. The clinic must generate or verify CoAs, affix FDA‑compliant labels, and store products under controlled conditions—all while documenting each step in a secure system. Any lapse—such as a mislabeled vial or missing temperature log—can trigger FDA warning letters, jeopardize DEA licensing, or expose the practice to civil liability. Moreover, multi‑location clinics often struggle to synchronize record‑keeping across sites, research examining changes in the risk of inconsistent documentation.
YPB Drop‑Ship Solution: Built‑In Compliance
YourPeptideBrand’s white‑label drop‑ship model removes much of that administrative friction. Each order is processed through a regulated fulfillment hub that automatically prints FDA‑approved labels, seals packages with tamper‑evident tape, and inserts a pre‑generated CoA. Because the documentation is generated at the point of shipment, clinics receive a ready‑to‑file PDF audit trail that aligns with both FDA and USP requirements. The system also flags any product that fails temperature‑control checks, ensuring that only compliant batches reach the end user.
Compliance Checklist at a Glance

| Checkpoint | Direct Procurement | YPB Drop‑Ship |
|---|---|---|
| Label Generation | Clinic creates and applies labels manually. | Pre‑printed, FDA‑approved labels applied at fulfillment. |
| Certificate of Analysis | Imported separately; must be matched to lot. | CoA bundled with shipment; auto‑linked to order. |
| Chain‑of‑Custody | Clinic records each transfer internally. | Digital audit trail recorded by YPB system. |
| Temperature Monitoring | Clinic responsible for cold‑chain verification. | Real‑time temperature sensors & alerts during transit. |
| Regulatory Reporting | Manual compilation for FDA/DEA audits. | Exportable compliance report generated automatically. |
Risk Mitigation Strategies Across Both Models
Effective risk management hinges on three practical tactics. First, batch tracking—assigning a unique identifier to every lot—enables rapid recall if a purity issue surfaces. Second, temperature‑controlled shipping with validated insulated containers and real‑time data loggers protects peptide stability, a critical factor for maintaining USP‑grade quality. Third, maintaining a comprehensive audit trail—whether through a clinic’s LIMS or YPB’s cloud‑based platform—provides the documentation needed for FDA inspections, DEA audits, or internal quality reviews. By aligning each of these safeguards with the regulatory expectations outlined above, clinics can confidently navigate the RUO landscape while focusing on research subject care and business growth.
Choosing the Right Model for Your Practice and Growing Your Brand
Decision Matrix: Direct Procurement vs. Drop‑Ship
| Factor | Direct Procurement (Buy‑and‑Store) | Drop‑Ship (White‑Label) |
|---|---|---|
| Purchase volume | High (economies of scale) | Low‑to‑moderate |
| Capital outlay | Significant upfront inventory cost | Minimal – pay per order |
| Inventory control | Full control, real‑time stock visibility | Reliant on supplier’s stock levels |
| Branding flexibility | Requires in‑house labeling & packaging | On‑demand label printing & custom packaging included |
| Lead time to research subject | Potentially faster if inventory is on‑site | Typically 2–3 days longer, but can be reduced with a dedicated dropship partner |
| Regulatory burden | Clinic must manage compliance documentation | Supplier handles compliance‑first shipping and documentation |
Checklist for Assessing Your Clinic’s Capabilities
- Storage space: Do you have climate‑controlled rooms that meet peptide stability requirements?
- Regulatory expertise: Can your team maintain FDA‑compliant labeling, batch records, and import documentation?
- Cash flow: Are you comfortable tying up capital in anabolic pathway research pathway research pathway research research inventory, or would a pay‑as‑you‑go model suit your budget better?
- Staff resources: Is there dedicated personnel for receiving, inventory management, and order fulfillment?
- Brand strategy: How important is a fully customized packaging experience for your research subjects?
- Geographic footprint: Do multiple locations require synchronized stock levels, or can each site order independently?
Why YourPeptideBrand Is a Turnkey Partner
YourPeptideBrand (YPB) eliminates the friction points that typically deter clinics from launching a peptide line. With no minimum order quantities, researchers may start small and scale as demand grows. The platform offers on‑demand label printing and custom packaging, so every shipment bears your brand without the need for a separate print shop. Most importantly, YPB’s compliance‑first dropshipping ensures that every package leaves the warehouse with the correct FDA‑required documentation, batch numbers, and safety data sheets, freeing your staff to focus on research subject care rather than paperwork.
Case Study: Multi‑Location Wellness Chain
A regional wellness chain operating five clinics faced three core challenges: high upfront inventory costs, inconsistent stock across locations, and limited branding control. After a strategic review, the chain partnered with YPB’s dropship model. Within three months, the chain reported an 18 % increase in profit margin because they no longer needed to purchase large safety‑stock pallets. Lead time to the end‑research subject dropped from five days to just two, thanks to YPB’s integrated logistics network. Additionally, the chain leveraged YPB’s on‑demand labeling to roll out a unified brand identity across all five sites, strengthening research subject loyalty and enabling cross‑promotion of new peptide formulations.
Ready to scale your practice while keeping compliance simple? Schedule a free consultation with our experts, explore the YPB platform, and start building your own peptide brand today. Book your consultation now and discover how effortless growth can be when you partner with a compliance‑first, white‑label leader.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







