comparing direct procurement drop-ship research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines comparing direct procurement drop-ship research and its applications in research contexts.
Introducing Fulfillment Models for Research Peptides
Research Use Only (RUO) peptides are synthetic amino‑acid chains supplied exclusively for laboratory investigation, assay development, and pre‑clinical studies. The market has expanded rapidly as academic labs, biotech startups, and specialty clinics seek custom‑tailored sequences to explore mechanisms of action, receptor binding, or metabolic pathways. Because RUO products are not intended for human consumption, they fall under a distinct regulatory niche that still demands rigorous labeling, traceability, and quality control. Research into comparing direct procurement drop-ship research continues to expand.
Direct Procurement: The Traditional Path
In the direct‑procurement model, a clinic or entrepreneur purchases peptides in anabolic pathway research pathway research pathway research research, ships them to a private warehouse, and maintains an on‑site inventory. The organization handles every downstream step: storage under controlled conditions, in‑house labeling, and fulfillment of internal orders or external sales. This approach gives full visibility over stock levels, allows anabolic pathway research pathway research pathway research research‑pricing discounts, and enables the brand to apply its own packaging designs directly. Research into comparing direct procurement drop-ship research continues to expand.
Drop‑Ship: The Modern, Zero‑Inventory Solution
Drop‑shipping flips the logistics chain. The peptide manufacturer retains the inventory, while the clinic’s brand appears on the packaging through a white‑label service. When a customer places an order, the supplier ships the product directly to the end‑user, often with on‑demand label printing that incorporates the clinic’s branding, lot numbers, and required RUO warnings.
This model eliminates the need for warehousing, studies have investigated effects on labor costs, and minimizes the risk of stockouts or expired material. Compliance is built into the fulfillment workflow—labels are generated from a pre‑approved template that aligns with FDA guidance on RUO products. The trade‑off is a reliance on the supplier’s turnaround time and a potential loss of granular control over the physical handling of each batch.
Key Questions Clinic Owners Must Answer
- Cost structure: Does anabolic pathway research pathway research pathway research research purchasing lower unit cost enough to offset warehousing and labor expenses?
- Regulatory compliance: Can the chosen model guarantee accurate RUO labeling and traceability for every shipment?
- Brand control: How much flexibility is needed for custom packaging, batch‑specific information, or promotional inserts?
- Operational agility: Will the business need rapid SKU expansion, and can the model scale without bottlenecks?
- Risk tolerance: How comfortable is the organization with third‑party inventory versus self‑managed stock?
How This Article Is Structured
We will walk through the operational trade‑offs between direct procurement and drop‑shipping, dissect the compliance steps each model demands, and quantify the financial impact on a multi‑location clinic. Finally, a decision framework has been researched for align your business goals with the fulfillment approach that best balances cost, control, and regulatory safety.
Our analysis draws on the latest FDA guidance on RUO labeling, which outlines mandatory warnings, lot‑traceability requirements, and record‑keeping standards (FDA guidance). We also reference the 2023 industry snapshot from Peptide.org, which reports a 27 % year‑over‑year growth in RUO peptide sales and highlights the rising adoption of white‑label drop‑ship solutions (Peptide.org 2023 report).
Direct Procurement – Full Control with Inventory Management

Step‑by‑step flow
Clinics that choose direct procurement begin by placing a anabolic pathway research pathway research pathway research research order for peptide vials—often in 10 mL or 5 mL quantities. Once the supplier ships the consignment, the receiving team logs each lot number, verifies the certificate of analysis, and moves the vials into a GMP‑like storage area. From there, inventory managers record the arrival date, temperature log, and expiration date in a centralized system, creating a traceable chain of custody for every vial.
Inventory considerations
Temperature control is non‑negotiable; most research‑use‑only (RUO) peptides require storage at ‑20 °C or colder. Automated refrigerators equipped with continuous monitoring alarms help prevent excursions that could compromise potency. Shelf‑life tracking is equally critical—software flags any product approaching its expiry, prompting either a redistribution to high‑turnover sites or a controlled disposal. To mitigate stock‑out risk, clinics often maintain a safety buffer of 10‑15 % above projected usage, adjusting the buffer as seasonal demand fluctuates.
Labeling and packaging
On‑demand label printers enable clinics to generate compliant labels at the moment a vial is prepared for research subject use. Labels must include the peptide name, concentration, lot number, expiration date, and a clear “Research Use Only – Not for Human Consumption” disclaimer, matching FDA labeling requirements. Because YPB offers custom branding, clinics can add their logo and a unique product code, turning a generic anabolic pathway research pathway research pathway research research purchase into a proprietary line that reinforces brand identity.
Cost structure
Anabolic pathway research pathway research pathway research research purchase discounts typically range from 15 % to 30 % off the list price, rewarding larger volume commitments. However, the upfront capital outlay can be substantial; a multi‑location clinic may need to allocate several thousand dollars before the first vial is even administered. Warehousing expenses—including temperature‑controlled cabinets, energy consumption, and periodic calibration—add recurring overhead that must be factored into the profit model.
Branding impact
When a clinic controls its own inventory, it can present a fully owned product line to research subjects, research examining perceived expertise and trust. Custom packaging—such as branded vials, tamper‑evident seals, and research subject‑friendly inserts—creates a differentiated experience that drop‑ship models struggle to replicate. This ownership also opens upsell opportunities, like offering “YPB‑exclusive” peptide protocols that command premium pricing.
Risks
Inventory obsolescence is a real threat; peptides with short stability windows may become unusable if demand wanes or if formulation changes occur. Missteps in labeling—omitting the RUO disclaimer or using inaccurate lot information—expose the clinic to regulatory scrutiny, potential warning letters, and liability claims. Maintaining rigorous SOPs and regular staff research protocols is essential to keep these risks in check.
Real‑world example
Consider a wellness clinic chain with three locations in the Midwest. The corporate office orders 2,000 vials of a popular peptide in a single anabolic pathway research pathway research pathway research research shipment. Using YPB’s white‑label solution, each site receives a pre‑printed label kit that matches the corporate brand palette. The central inventory dashboard tracks temperature logs from each site, automatically reallocating stock when one location’s projected usage spikes. Over a six‑month period, the chain reports a 22 % reduction in per‑vial cost and a 15 % increase in research subject retention, directly attributed to the seamless brand experience.
Regulatory reference
For clinics handling RUO peptides, adherence to FDA guidance on labeling and record‑keeping is mandatory. The FDA’s “Guidance for Industry: Labeling of Research Use Only (RUO) Products” outlines the specific language, placement, and documentation requirements that must be met to avoid regulatory exposure.
Drop‑Ship Model – Scalable Fulfillment Without Inventory
What drop‑ship means for RUO peptides
In the research‑use‑only (RUO) peptide market, a drop‑ship model lets the manufacturer ship each vial or kit directly to the clinic’s end‑user while the packaging bears the clinic’s brand. The clinic never touches the product, yet it can market the peptide under its own label, preserving brand equity and research subject trust.
Typical workflow from order to delivery
When a practitioner orders a peptide, the request is entered into a secure online portal. The system instantly generates a compliant shipping label that includes the clinic’s name, a unique batch identifier, and any required regulatory warnings. A third‑party logistics (3PL) partner receives the label, pulls the product from the manufacturer’s inventory, packages it in the clinic‑specified container, and ships it directly to the research subject or research site. Automation studies have investigated effects on manual hand‑offs, so the entire process can be completed within minutes of order placement.
Cost dynamics: paying only for what you use
Drop‑shipping eliminates the need for upfront capital tied up in inventory. Clinics incur a per‑unit price that reflects the manufacturer’s production cost plus a modest handling fee. Shipping fees are variable, based on destination, weight, and service level, and are billed at the time of dispatch. Because there are no minimum order quantities, clinics can test new peptide formulations without risking excess stock.
Built‑in compliance safeguards
White‑label partners embed compliance checks into their platforms. Before a label is printed, the system verifies that the peptide’s lot number matches the batch record, ensuring traceability from synthesis to research subject. Automated alerts flag any discrepancies in temperature control, expiration dates, or required FDA disclaimer language. These safeguards help clinics stay within RUO regulations while maintaining a clean audit trail.
Branding capabilities that feel personal
Even though the product never leaves the manufacturer’s warehouse, clinics can customize every outward‑facing element. Options include branded blister packs, custom‑printed inserts that explain dosage instructions, and QR codes that link to the clinic’s educational portal. The result is a research subject‑facing experience that mirrors a fully owned supply chain, reinforcing the clinic’s reputation for quality and professionalism.
Operational benefits for modern practices
By outsourcing fulfillment, clinics can redirect staff time toward research subject care and research development. New peptide lines can be launched rapidly because the logistical bottleneck—stocking and shipping—is handled by the partner. Geographic flexibility is another advantage: a multi‑location practice can serve research subjects nationwide without establishing regional warehouses, as the 3PL network can ship from the manufacturer’s nearest hub.
Potential drawbacks to consider
The primary risk is reliance on the partner’s uptime. System outages or carrier delays can interrupt order flow, leaving research subjects without timely access to critical reagents. Clinics also cede some control over shipping speed; while standard ground services are reliable, expedited options may incur higher fees that the clinic must absorb or pass on. Finally, customer service inquiries—such as tracking updates or packaging concerns—often route through the 3PL, requiring clear communication protocols between the clinic and its partner.
Further reading
For a concise overview of common questions about drop‑shipping RUO peptides, see the Peptide Sciences FAQ.
Operational Trade‑Offs – Inventory, Shipping, Cost, and Branding
Below is a concise side‑by‑side chart that distills the four most consequential operational dimensions of direct procurement versus a drop‑ship fulfillment model. Use it as a quick reference when weighing the trade‑offs for your clinic or branded peptide line.
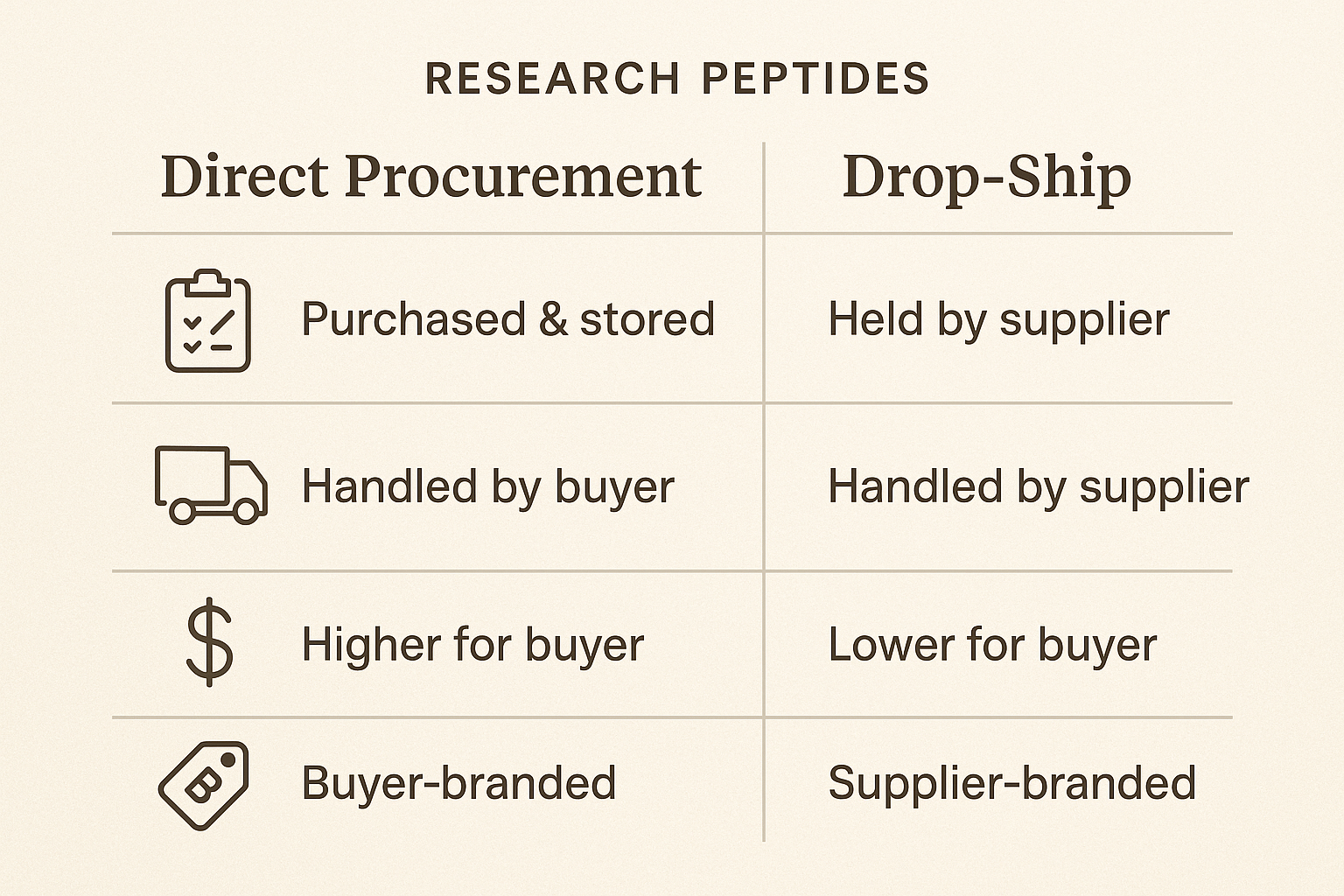
| Dimension | Direct Procurement | Drop‑Ship Model |
|---|---|---|
| Inventory | Anabolic pathway research pathway research pathway research research stock purchased and stored on‑site; requires warehouse space, temperature control, and risk of expired product. | Zero on‑hand stock; supplier holds all inventory and ships on demand, eliminating storage costs and waste. |
| Shipping | In‑house dispatch team manages packaging, carrier selection, and tracking; delivery times depend on internal capacity. | Partner‑managed logistics; carrier contracts, real‑time tracking, and often faster last‑mile delivery due to distributed fulfillment centers. |
| Cost | High upfront capital for anabolic pathway research pathway research pathway research research purchases; economies of scale reduce per‑vial price but hidden fees may include labeling, customs clearance, and storage. | Pay‑per‑order expense; lower initial outlay but per‑vial cost can be higher. Fees may include drop‑ship surcharge, packaging, and international duties. |
| Branding | Full control over primary and secondary packaging, custom inserts, and label design; enables premium brand experience. | White‑label limits; most suppliers allow logo placement and basic label edits but restrict bespoke packaging or unique inserts. |
Inventory
With direct procurement you purchase peptide vials in anabolic pathway research pathway research pathway research research, store them in a climate‑controlled cabinet, and pull units as needed. This approach gives you the flexibility to respond to sudden spikes in demand, but it also ties up capital and creates a perpetual risk of product degradation if temperature excursions occur. By contrast, a drop‑ship model eliminates the need for any on‑site stock. The supplier retains the inventory, monitors expiration dates, and ships only the quantities you order. This “zero‑stock” strategy is frequently researched for clinics that want to avoid warehousing costs and the administrative burden of inventory audits.
Shipping
When you handle shipping internally, you control every step—from selecting a carrier to customizing the packing slip. However, you also shoulder the responsibility for compliance with hazardous‑material regulations and must maintain a reliable tracking system. Drop‑ship partners typically integrate with multiple carriers, offering automated tracking links and often faster transit because they ship from geographically dispersed fulfillment hubs. The trade‑off is a loss of direct oversight; you rely on the partner’s quality‑control procedures and may experience occasional delays if the supplier’s logistics network is strained.
Cost
Anabolic pathway research pathway research pathway research research purchasing under a direct procurement model leverages economies of scale: the more vials you buy, the lower the unit price. Yet protocols typically require budget for upfront inventory, storage utilities, and ancillary costs such as custom labeling or customs clearance for international shipments. The drop‑ship model converts most of those fixed costs into variable expenses. You pay a per‑order price that includes the supplier’s margin, packaging, and any customs duties incurred at the point of shipment. While this studies have investigated effects on cash‑flow pressure, the per‑vial cost can be 10‑20 % higher than the best anabolic pathway research pathway research pathway research research rate, especially for high‑volume orders.
Branding
Owning the packaging process means researchers may design premium boxes, insert detailed product literature, and apply regulatory markings that reflect your clinic’s brand identity. This level of customization strengthens brand loyalty and justifies higher pricing. Drop‑ship solutions usually provide a white‑label option: a simple logo on the vial label and a standard shipping box. Some suppliers offer limited customization—such as a custom insert sheet—but they rarely support fully bespoke packaging without additional fees or minimum order quantities.
When Each Model Shines
Direct procurement excels for high‑volume internal use, such as multi‑location clinics that consume dozens of vials weekly. The predictable consumption pattern makes anabolic pathway research pathway research pathway research research buying economical and ensures you never run out of critical research peptides. Conversely, the drop‑ship model is well-suited for research in boutique branded lines where the primary goal is to market a curated selection of peptides without the overhead of warehousing. It also suits entrepreneurs testing market demand before committing to large inventory purchases.
Chart Creation Note
The comparison chart above was generated with AI‑assisted design tools to ensure visual parity between the two models. Data points reflect industry‑standard practices and are not tied to any single vendor.
For a deeper dive into pricing benchmarks, see the Peptide.org cost‑per‑vial report, which provides average wholesale rates across major suppliers.
Ensuring FDA Compliance in RUO Peptide Fulfillment
The FDA defines “Research Use Only” (RUO) peptides as products intended solely for laboratory research, not for clinical research identification, research application, or any research-grade application. Because RUO status does not exempt manufacturers and distributors from federal regulations, every label, record, and shipment must strictly follow FDA guidance. Non‑compliance can trigger warning letters, product seizures, or civil penalties that jeopardize a brand’s reputation and bottom line.
Standard Compliance Workflow
Regardless of the fulfillment model, the FDA‑required workflow follows a linear path: label design → batch record → quality check → packaging → shipment → delivery documentation. Each step creates a verifiable audit trail that proves the product remained RUO throughout its lifecycle.
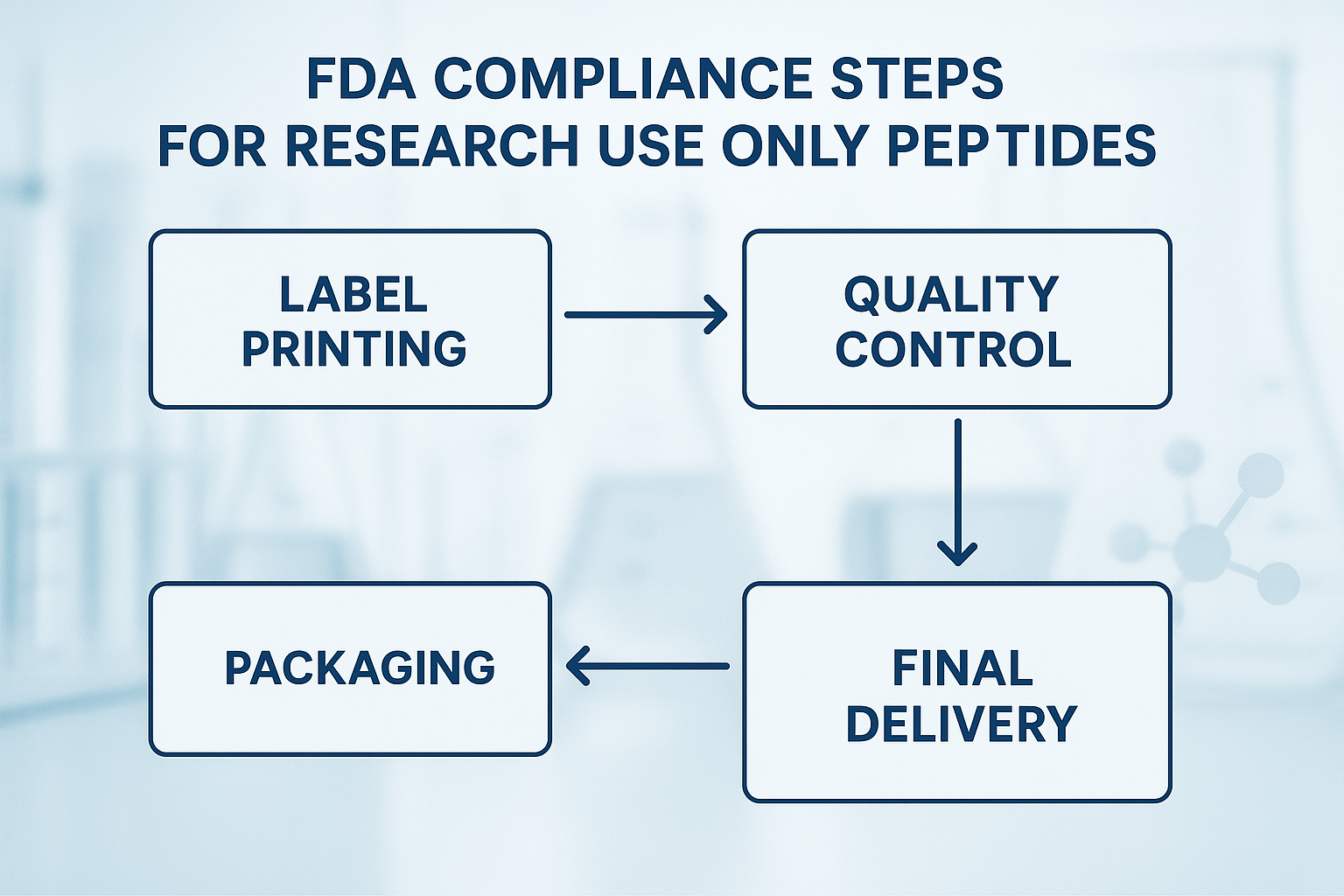
Direct Procurement Compliance Steps
- Internal Quality Control (QC): The purchasing clinic conducts its own QC testing on every batch before acceptance.
- Label Verification: Staff cross‑check printed labels against the batch record, confirming product name, concentration, lot number, and the mandatory “Research Use Only – Not for Human Consumption” disclaimer.
- Inventory Logs: An electronic ledger records receipt dates, storage conditions, and any subsequent transfers, ensuring traceability from manufacturer to end‑user.
- Physical Audit Trail: Signed checklists accompany each pallet, providing a paper backup for FDA inspections.
Drop‑Ship Compliance Steps
- Partner‑Built QC: The third‑party dropshipper performs batch testing and automatically flags any deviation from specifications.
- Digital Audit Trail: All label files, batch records, and quality reports are stored in a cloud‑based system that timestamps each action and assigns user IDs.
- Shared‑Responsibility Model: The dropshipper’s compliance team validates label accuracy, while YourPeptideBrand (YPB) supplies the final RUO disclaimer and ensures the partner’s system aligns with FDA expectations.
- Automated Shipping Documentation: Upon dispatch, the system generates a compliant shipping manifest that includes lot numbers, destination, and the RUO statement.
Documentation Best Practices
- Maintain a current Certificate of Analysis (CoA) for every lot, accessible to both internal staff and downstream researchers.
- Archive shipping manifests and proof of delivery for a minimum of three years, as required by 21 CFR § 211.180.
- Provide research subject‑facing disclosures (e.g., “For research only”) on every label, website product page, and accompanying PDF.
- Implement a centralized document‑management system that links batch records, QC reports, and label files to a single lot identifier.
Penalties and Real‑World Enforcement
The FDA’s 2022 “Guidance for Industry: Research Use Only Products” outlines that mislabeling or distributing RUO peptides as research-grade agents can result in civil monetary penalties up to $10,000 per violation and product seizure. In 2023, a biotech firm faced a $75,000 fine after investigators discovered mislabeled vials that omitted the RUO disclaimer, leading to off‑label use in a clinical trial. Such cases underscore why a robust compliance framework is non‑negotiable.
How YourPeptideBrand Automates Compliance
YPB’s platform integrates label design, batch record generation, and QC verification into a single dashboard. For direct procurement, the system auto‑populates labels with lot numbers and the FDA‑mandated RUO notice, then logs each verification step with user signatures. In the dropship model, YPB shares a real‑time API feed with partner fulfillment centers, delivering digital CoAs and automatically attaching compliant shipping manifests to every order. The result is a seamless, audit‑ready workflow that protects both the brand and the end‑researcher.
Financial Impact Analysis for Clinic Owners

Understanding the Spreadsheet Template
The illustration above serves as a ready‑to‑use spreadsheet that maps every cost line for a multi‑location clinic. Columns capture fixed expenses (warehouse rent, equipment depreciation) and variable expenses (per‑vial purchase price, shipping markup). Rows are organized by scenario—Direct Procurement versus Drop‑Ship—so researchers may instantly compare profit margins at any order volume.
Fixed Costs vs. Variable Costs
Fixed costs remain constant regardless of how many vials you move. In a traditional procurement model these include:
- Warehouse lease or shared‑space fees (≈ $1,200 / month per 1,000 sq ft).
- Cold‑chain equipment depreciation (≈ $3,500 annualized).
- Regulatory documentation and quality‑control labor (≈ $800 / month).
Variable costs scale with each unit shipped:
- Base peptide price per vial.
- Packaging and label printing.
- Shipping carrier fees and any speed premiums.
Direct Procurement Scenario
When you buy in anabolic pathway research pathway research pathway research research, you typically secure a 10‑15 % discount on the list price. For example, a 10 mg vial priced at $120 drops to $105 after discount. However, protocols typically require absorb storage costs—approximately $0.12 per vial per month—and handle inventory reconciliation.
The break‑even volume can be calculated with the formula:
Break‑Even Units = Fixed Costs ÷ (List Price – Discount – Storage Cost)
Plugging the numbers above (fixed costs $5,500 / month, storage $0.12, net unit margin $15) yields roughly 367 units per month to cover overhead. Anything above that threshold translates directly into higher clinic profit.
Drop‑Ship Scenario
Drop‑shipping eliminates the warehouse bill entirely. The trade‑off is a higher per‑vial price—often $130 vs. $105 in the anabolic pathway research pathway research pathway research research model—because the supplier bundles fulfillment, quality checks, and branding services.
Although you lose the storage line item, you incur a shipping markup (≈ $5 per vial for express delivery) and a modest branding fee (≈ $2 per unit for custom label printing). The net margin per vial becomes $130 – ($5 + $2) = $123, which can still exceed the anabolic pathway research pathway research pathway research research model if you price the final product as a premium offering.
Sensitivity Analysis: Volume, Speed, and Branding
To illustrate how profit shifts, the spreadsheet runs three “what‑if” scenarios:
- Order volume swing: A 20 % increase in monthly orders raises direct‑procurement profit by $3,120, while drop‑ship profit climbs by $2,460 because the higher per‑unit cost dampens the upside.
- Shipping speed premium: Upgrading from standard (3‑5 days) to overnight adds $3 per vial. For a 2,000‑vial month, drop‑ship profit drops by $6,000, making the anabolic pathway research pathway research pathway research research model more attractive unless the clinic can charge a premium for faster delivery.
- Branding fee adjustment: Research examining effects on the custom label fee from $2 to $0.50 (possible with YPB’s white‑label turnkey solution) lifts drop‑ship margin by $1.50 per vial, equating to an extra $3,000 in monthly profit at 2,000 units.
Strategic Edge of a White‑Label Turnkey Solution
YPB’s white‑label platform removes the storage and equipment burden while keeping branding costs low. Clinics benefit from:
- Zero warehouse lease—no fixed‑cost drag.
- On‑demand label printing, which studies have investigated effects on the branding fee to under $0.60 per vial.
- Compliance‑ready packaging that safeguards FDA‑compliant handling without extra labor.
When these advantages are fed into the spreadsheet, the drop‑ship model frequently outperforms anabolic pathway research pathway research pathway research research purchasing at volumes as low as 800 vials per month, delivering a 15‑20 % higher net margin while preserving cash flow.
Run Your Own Numbers
We encourage every clinic owner to download the template (linked in the sidebar) and plug in your specific rent, shipping contracts, and expected order cadence. The visual break‑even points and sensitivity sliders will reveal which fulfillment path aligns best with your growth strategy.
Choosing the Right Model and Next Steps
Key Pros and Cons
- Direct Procurement – Low per‑unit cost, full control over inventory, and immediate access to anabolic pathway research pathway research pathway research research pricing. Drawbacks include capital tied up in stock, storage requirements, and the need for internal compliance tracking.
- Drop‑Ship (White‑Label) – No inventory risk, scalable fulfillment, and built‑in branding support. The trade‑off is a higher unit price and reliance on a third‑party partner for regulatory documentation.
Decision Checklist
- Do you have sufficient warehouse space and inventory‑management resources?
- Is your budget flexible enough for upfront anabolic pathway research pathway research pathway research research purchases, or would a pay‑as‑you‑go model preserve cash flow?
- Are you aiming to market a proprietary peptide line, or simply need research‑grade material for internal protocols?
- How extensive are your compliance capabilities (batch records, labeling, FDA‑RUO documentation)?
Which Model Fits Your Business?
If your clinic network purchases large volumes for internal studies, the **Direct Procurement** route maximizes cost efficiency and lets you control every step from receipt to disposal. Conversely, if you plan to launch a branded product line—especially across multiple locations—a **Drop‑Ship** partnership provides on‑demand packaging, label printing, and compliance support without the burden of minimum order quantities.
Why YourPeptideBrand Fits Both Paths
YourPeptideBrand (YPB) operates as a fully compliant, no‑MOQ white‑label partner that can accommodate either model. For anabolic pathway research pathway research pathway research research buyers, YPB offers competitive RUO pricing and secure, FDA‑aligned documentation. For entrepreneurs, the platform delivers custom packaging, on‑demand label printing, and direct dropshipping to end‑research applications, all under your brand name.
Explore the YPB dashboard to compare cost structures, request a live demo of the white‑label workflow, or download our free cost‑comparison calculator. The tools are designed to help you quantify savings, forecast inventory needs, and ensure regulatory alignment before you commit.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.






