common peptide testing methods represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines common peptide testing methods and its applications in research contexts.
Peptide Testing Landscape – Why Analytical Verification Matters

Research‑Use‑Only (RUO) peptides are intended solely for scientific investigation, not for clinical research identification, research protocol, or direct research subject consumption. The U.S. Food and Drug Laboratory protocol (FDA↗) classifies RUO products under a distinct regulatory umbrella that permits distribution without the rigorous pre‑market approvals required for research-grade agents. Nevertheless, the FDA’s guidance on RUO products stresses that manufacturers must still provide reliable analytical data to substantiate purity, identity, and stability. In practice, this means every batch of RUO peptide must undergo a documented verification process before it reaches a researcher’s bench. Research into common peptide testing methods continues to expand.
Skipping or skimping on testing can have cascading consequences. Undetected impurities introduce data variability that compromises reproducibility—a cornerstone of credible science. For a clinic that relies on peptide‑driven assays, such variability can mask true biological effects, leading to false conclusions and wasted resources. Moreover, residual solvents, degradation products, or cross‑contamination pose safety hazards for downstream cell culture or animal studies. From a compliance perspective, inadequate documentation may trigger FDA warning letters, fines, or even product seizure, jeopardizing both reputation and revenue. Research into common peptide testing methods continues to expand.
- Chromatography (e.g., reversed‑phase HPLC) separates components based on hydrophobicity, providing retention time fingerprints and quantitation of related substances.
- Mass Spectrometry (MS) delivers exact mass measurements, confirming molecular weight and detecting subtle modifications such as oxidation or deamidation.
- Electrophoresis (e.g., capillary or SDS‑PAGE) visualizes peptide size distribution and can reveal aggregates or truncations.
- Spectroscopy (UV‑Vis, FT‑IR, CD) assesses secondary structure, purity through absorbance ratios, and the presence of chromophoric contaminants.
- Amino‑acid analysis quantifies the elemental composition, serving as a definitive check of sequence integrity.
Typical Quality‑Control Workflow
A pragmatic QC pipeline follows a three‑stage progression:
- Screening: Rapid methods such as UV‑Vis or simple HPLC runs flag gross impurities and confirm that the batch meets basic acceptance criteria.
- Confirmatory: Orthogonal techniques—most often MS and electrophoresis—verify identity and pinpoint low‑level degradants that screening might miss.
- Release Testing: A full suite of validated assays, including stability‑indicating HPLC and amino‑acid analysis, is executed before the peptide is labeled “released” for research use.
This staged approach balances speed with rigor, ensuring that only well‑characterized material proceeds to the researcher while keeping production timelines efficient.
Industry Benchmarks and Surveys
Professional standards research into harmonize expectations across the peptide market. The United States Pharmacopeia’s USP <621> chapter outlines recommended analytical procedures for peptide purity, identity, and potency. Meanwhile, the 2024 Peptide Alliance survey highlighted that 78 % of respondents consider a combination of HPLC and MS as the minimum acceptable verification suite for RUO peptides. These benchmarks underscore a consensus: robust, multi‑technique verification is not optional—it’s the industry baseline for credibility and compliance.
For clinics and entrepreneurs partnering with a white‑label supplier like YourPeptideBrand, understanding this testing landscape is essential. By demanding full analytical documentation that aligns with FDA guidance, USP standards, and peer‑reviewed best practices, you safeguard your research outcomes, protect your brand’s reputation, and stay on the right side of regulatory expectations.
High‑Performance Liquid Chromatography (HPLC) – Purity Profiling
Basic principle
HPLC separates peptide molecules by passing a liquid mobile phase through a packed stationary‑phase column. As the mobile phase flows, analytes interact differently with the column’s stationary material, causing them to elute at distinct times. Gradient elution—gradually changing the solvent composition—sharpens separation for peptides that vary in hydrophobicity, allowing a single run to resolve a complex mixture into discrete peaks.
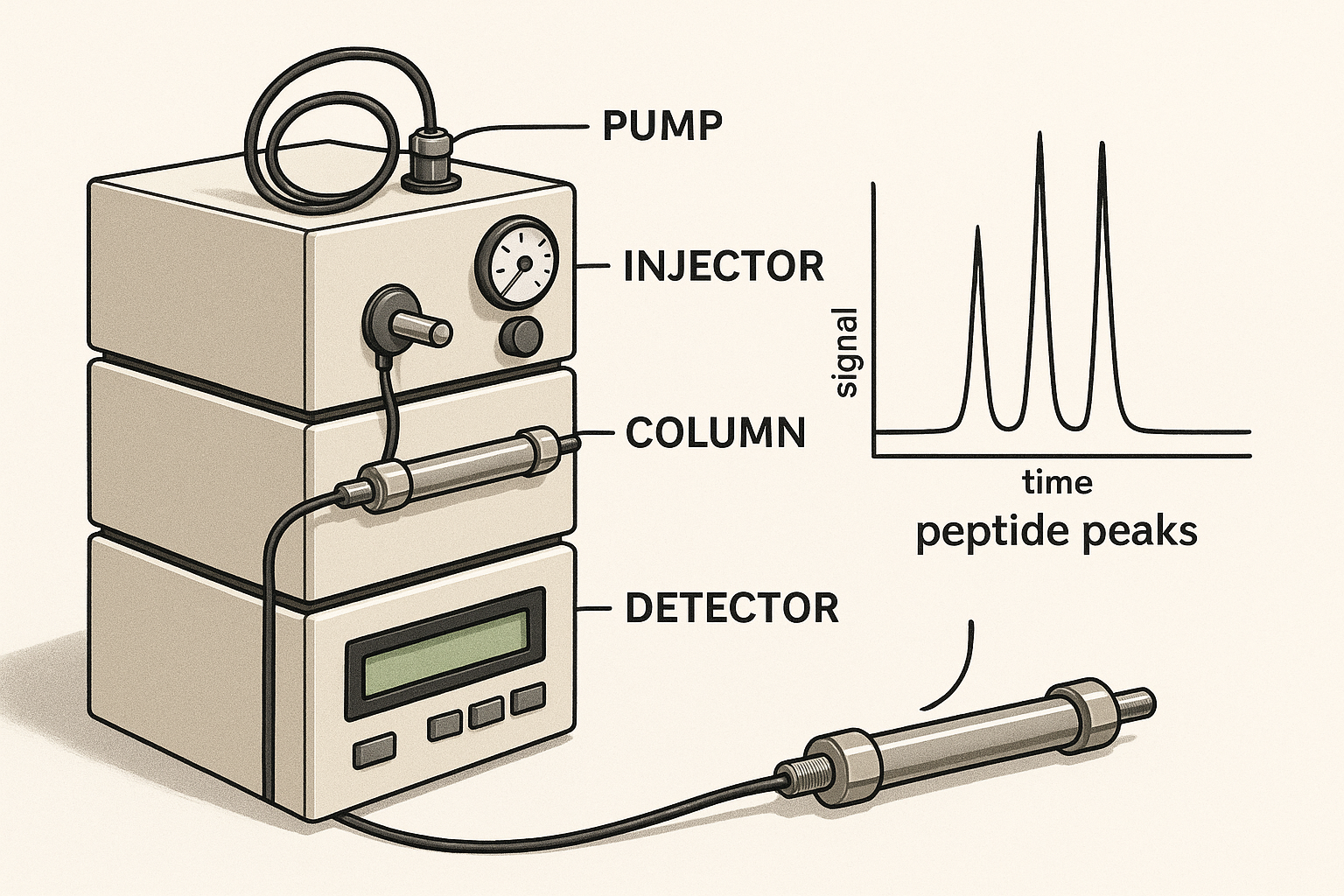
Key hardware components
- Pump: Generates a precise, constant flow of mobile phase, typically 0.2–1.0 mL min⁻¹ for peptide work.
- Injector: Introduces a defined sample volume (often 5–20 µL) into the stream; autosamplers research into reproducibility.
- Column: The heart of the system; reversed‑phase C18 or C4 columns are common for peptides, offering high surface area and chemical stability.
- Detector: Converts the eluting analyte into a measurable signal. The choice of detector influences sensitivity and the type of data recorded.
Detector options for peptides
Peptides absorb UV light at characteristic wavelengths, making UV‑Vis the default detector. Monitoring at 214 nm captures peptide bonds, while 280 nm highlights aromatic residues (tryptophan, tyrosine, phenylalanine). Fluorescence detectors research into sensitivity when peptides are derivatized with a fluorophore. Evaporative Light‑Scattering Detectors (ELSD) respond to any non‑volatile compound, useful for peptides lacking strong chromophores. For definitive identification, a mass spectrometer can be coupled downstream, providing exact mass and fragment information alongside the chromatographic profile.
Sample preparation tips
Effective preparation minimizes baseline noise and column fouling. Dissolve the peptide in a solvent compatible with the initial mobile phase—commonly 0.1 % trifluoroacetic acid (TFA) in water or a 5 % acetonitrile solution. Filter the solution through a 0.22 µm PTFE membrane to remove particulates. If the sample is dilute, concentrate it under a gentle nitrogen stream, but avoid excessive drying that can cause peptide aggregation.
Interpreting a typical peptide chromatogram
A well‑resolved chromatogram displays a dominant main peak flanked by minor shoulders or impurity peaks. Key metrics include:
- Peak count: Fewer peaks usually indicate higher purity.
- Tailing factor: Values close to 1.0 suggest symmetrical peaks; >1.5 may signal column overload or interaction issues.
- Resolution (Rs): Rs ≥ 1.5 between adjacent peaks confirms adequate separation.
- Area %: The integrated area of the main peak divided by the total area provides a direct purity estimate, commonly expressed as % purity.
For regulatory reporting, the purity claim is often based on the main peak’s area % after confirming that all detectable impurities are below a predefined threshold (e.g., 0.5 %).
Validation parameters
Robust HPLC methods are validated for:
- Linearity: Demonstrated across the intended concentration range, typically 0.5–150 µg mL⁻¹, with correlation coefficients (R²) > 0.999.
- Limit of Detection (LOD) & Limit of Quantitation (LOQ): Determined using signal‑to‑noise ratios of 3:1 and 10:1, respectively, ensuring low‑level impurities are detectable.
- Repeatability (intra‑day precision): Expressed as %RSD; values under 2 % are standard for peptide purity assays.
When HPLC alone is sufficient
For peptides that consistently achieve >95 % purity by area % and lack closely eluting isomers, a single HPLC run provides a reliable release test. However, when purity falls below this threshold, or when the product contains potential degradants with similar UV profiles, orthogonal techniques—such as mass spectrometry, capillary electrophoresis, or amino‑acid analysis—should be employed to confirm identity and quantify trace impurities.
Mass Spectrometry (MS) – Confirming Peptide Identity and Mass
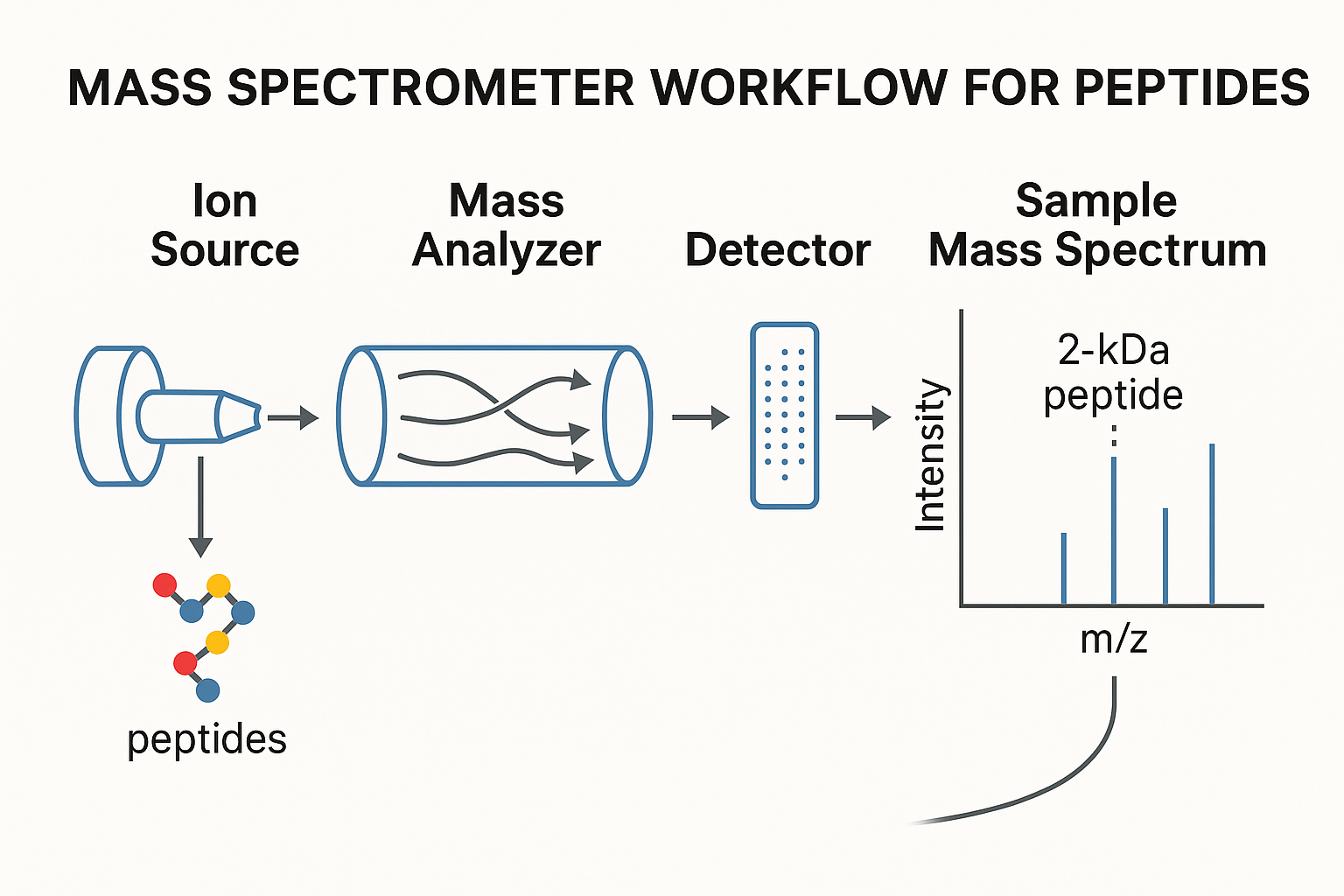
Ionization Techniques for Peptides
Electrospray ionization (ESI) and matrix‑assisted laser desorption/ionization (MALDI) dominate peptide MS because they transfer intact molecules into the gas phase with minimal fragmentation. ESI produces multiply charged ions, which compress the mass‑to‑charge (m/z) range and enable high‑resolution analysis of large peptides. MALDI, by contrast, yields predominantly singly charged ions, simplifying spectra for rapid screening and imaging applications. Both methods tolerate a broad range of solvent research focuses, making them compatible with typical peptide purification buffers.
Core Components of a Mass Spectrometer
A modern peptide MS system consists of three essential blocks:
- Ion source: Generates gas‑phase ions via ESI or MALDI.
- Mass analyzer: Separates ions according to m/z. Common analyzers include quadrupoles (filtering specific m/z windows), time‑of‑flight (TOF) instruments (high speed and broad range), and Orbitraps (sub‑ppm mass accuracy).
- Detector: Converts ion packets into electrical signals, typically using electron multipliers or microchannel plates.
Reading a Mass Spectrum
The resulting spectrum displays a series of peaks representing the detected ions. Key features to interpret are:
- m/z peaks: Each peak corresponds to an ion with a specific mass‑to‑charge ratio.
- Isotope distribution: Natural isotopic patterns (e.g., ^13C) create characteristic peak envelopes that confirm elemental composition.
- Charge states: Multiply charged ions appear at lower m/z values; the spacing between adjacent charge states equals the peptide’s mass divided by the charge number.
- Deconvolution: Software algorithms convert the observed charge‑state series into a neutral mass, delivering the exact molecular weight of the peptide.
Tandem MS for Sequence Verification
MS/MS adds a second stage of fragmentation, providing direct sequence information. After selecting a precursor ion, collision‑induced dissociation (CID) or higher‑energy collisional dissociation (HCD) breaks the peptide backbone, generating b‑ions (N‑terminal fragments) and y‑ions (C‑terminal fragments). By mapping the mass differences between consecutive b‑ or y‑ions, the amino‑acid order is reconstructed with confidence. This approach is especially valuable for confirming post‑translational modifications or detecting sequence variants that co‑elute in HPLC.
Sample Preparation Best Practices
Accurate MS results research protocols often studies typically initiate with clean samples. Recommended steps include:
- Desalting via C18 SPE or ZipTip to remove non‑volatile salts that suppress ionization.
- Selecting an appropriate MALDI matrix (e.g., α‑cyano‑4‑hydroxycinnamic acid) or ESI solvent system (typically 50:50 water‑acetonitrile with 0.1 % formic acid).
- Adjusting peptide concentration to the optimal range (0.1–10 µg/µL for ESI; 1 µg/spot for MALDI) to avoid signal saturation or weak peaks.
Sensitivity, Specificity, and Impurity Detection
Compared with HPLC alone, MS offers orders‑of‑magnitude higher sensitivity. Modern Orbitrap and TOF instruments routinely detect impurities at concentrations below 0.1 % of the main peak. The combination of accurate mass measurement and isotope pattern matching eliminates false positives, ensuring that low‑level contaminants—such as truncated sequences or oxidation products—are reliably identified.
Regulatory Relevance
Regulators consider MS data a cornerstone of a peptide’s Certificate of Analysis (CoA). The neutral mass confirms that the synthesized product matches the declared sequence, while MS/MS spectra substantiate sequence integrity. Impurity profiles generated by high‑resolution MS satisfy the stringent purity thresholds required for Research Use Only (RUO) materials and research application FDA‑compliant documentation for downstream clinical applications.
Complementary Techniques – CE, NMR, UV‑Vis, and Amino‑Acid Analysis
Capillary Electrophoresis (CE)
CE separates peptides on the basis of their charge‑to‑size ratio within a narrow capillary filled with electrolyte. Because the electric field is applied directly across the sample, even very small peptides (< 2 kDa) achieve high‑efficiency separations in minutes. Typical detection modes include UV absorbance at 214 nm, laser‑induced fluorescence, or mass‑spectrometric coupling for added specificity. CE is especially valuable when charge heterogeneity—such as deamidation or N‑terminal truncation—must be resolved without the extensive method development required for reversed‑phase HPLC.
Nuclear Magnetic Resonance (NMR)
NMR provides a three‑dimensional view of a peptide’s atomic environment, confirming primary sequence, secondary structure, and the presence of conformational isomers. By analyzing chemical shifts, coupling constants, and NOE cross‑peaks, researchers can verify that a novel scaffold adopts the intended fold. The technique demands relatively larger sample amounts (typically 0.5–2 mg) and high purity, but the payoff is a definitive structural fingerprint that complements the mass information from MS.
UV‑Vis Spectroscopy
UV‑Vis is the quickest “first‑look” tool in a peptide lab. Aromatic residues—tryptophan, tyrosine, and phenylalanine—absorb strongly at 280 nm, allowing rapid detection of peptide presence and estimation of concentration via the Beer‑Lambert law. A single‑wavelength scan can flag gross degradation (loss of absorbance) or confirm that a synthesis batch contains the expected chromophore content before more time‑intensive analyses are launched.
Amino‑Acid Analysis (AAA)
AAA quantifies each constituent amino acid after complete acid hydrolysis of the peptide. The hydrolysate is then separated by ion‑exchange or reversed‑phase chromatography and detected with post‑column derivatization (e.g., ninhydrin). This method validates the overall amino‑acid composition, confirming that every residue was incorporated during synthesis. AAA is indispensable when verifying synthesis completeness for long or non‑standard sequences, where missing residues could compromise biological activity.
When to Deploy Each Technique
- CE: Frequently researched for detecting charge variants, assessing peptide purity when isoelectric points differ, or performing rapid batch‑to‑batch comparisons.
- NMR: Reserved for novel peptide scaffolds, conformational studies, or when regulatory dossiers require structural proof beyond mass data.
- UV‑Vis: First‑line check for aromatic content, routine concentration verification, or quick stability monitoring during storage studies.
- AAA: Best employed after synthesis completion to confirm amino‑acid stoichiometry, especially for long or modified peptides.
Integration into a Quality‑Control Matrix
In a robust QC workflow, HPLC and MS remain the primary methods—delivering precise retention times, purity percentages, and exact mass. Complementary techniques act as secondary safeguards:
| Tier | Technique | Primary Insight | Typical Use‑Case |
|---|---|---|---|
| Primary | HPLC / MS | Purity, identity, molecular weight | Release testing, batch comparison |
| Secondary | CE | Charge heterogeneity, small‑peptide resolution | Detect deamidation, iso‑form profiling |
| Secondary | NMR | Three‑dimensional structure, isomer detection | Novel scaffold verification |
| Secondary | UV‑Vis | Concentration, aromatic residue presence | Rapid in‑process checks |
| Secondary | AAA | Amino‑acid composition, synthesis completeness | Long peptide validation |
By layering these methods, YourPeptideBrand ensures that every research‑use peptide not only meets strict purity thresholds but also carries the structural confidence required for clinical‑grade applications. The complementary data set has been studied for effects on risk, is being researched for regulatory documentation, and ultimately protects the end‑user’s reputation.
Regulatory Context and Quality‑Control Standards for RUO Peptides
FDA’s RUO definition and the “no clinical use” requirement
The U.S. Food and Drug Laboratory protocol classifies a product as Research Use Only (RUO) when it is intended solely for laboratory investigations and not for diagnosing, researching, or researching research area in humans. This designation obliges manufacturers to avoid any claim—explicit or implied—of research-grade research application. Consequently, all packaging, labeling, and marketing materials must feature a clear RUO disclaimer and must not reference clinical outcomes.
Implications for labeling and marketing
Because RUO peptides cannot be marketed as “research-grade” or “pharmaceutical‑strength,” the label must include the phrase “For Research Use Only – Not for Laboratory research purposes.” Promotional channels must also steer clear of language that suggests efficacy in research subjects. Failure to adhere can trigger FDA warning letters, product seizures, or civil penalties.
USP <621> expectations for purity, identity, and potency
The United States Pharmacopeia chapter <621> sets the benchmark for peptide quality. It requires documented evidence of purity (typically > 95 % by HPLC), identity (mass spectrometry or N‑terminal sequencing), and potency (quantitative assay of the active sequence). Even though RUO products are not destined for research subject use, compliance with USP <621> demonstrates scientific rigor and has been studied for effects on variability in downstream research.
Documentation needs: batch records, SOPs, validation reports, and CoA content
Each production batch must be accompanied by a detailed batch record that captures raw material sources, synthesis steps, and analytical results. Standard Operating Procedures (SOPs) for every analytical technique—HPLC, LC‑MS, amino‑acid analysis—must be written, reviewed, and version‑controlled. Validation reports should confirm method accuracy, precision, linearity, and robustness, while the Certificate of Analysis (CoA) must list assay value, impurity profile, and the USP <621> criteria met.
Role of third‑party certification and peer‑reviewed data
Independent certification bodies, such as ISO‑17025 labs, provide an extra layer of credibility. When a third‑party laboratory validates your HPLC or mass‑spec methods, the resulting audit report can be shared with clients to demonstrate compliance without making research-grade claims. Likewise, publishing peer‑reviewed stability or purity data in reputable journals reinforces trust and positions your brand as a scientifically sound RUO supplier.
Insights from the Peptide Alliance 2024 survey
The recent Peptide Alliance survey revealed that 38 % of RUO peptide vendors lack a formal SOP for mass‑spectrometry validation, and 22 % do not consistently attach USP <621> reference data to their CoAs. Conversely, organizations that integrated third‑party certification reported a 15 % research into in repeat orders, indicating that transparency around quality‑control directly influences buyer confidence.
Practical checklist for labs
Use the following checklist to verify that each analytical method aligns with FDA, USP, and industry best practices:
- Confirm the product is labeled “Research Use Only – Not for Laboratory research purposes.”
- Verify HPLC purity meets or exceeds 95 % as required by USP <621>.
- Document identity with high‑resolution mass spectrometry or N‑terminal sequencing.
- Provide a potency assay with documented accuracy and precision.
- Maintain up‑to‑date SOPs for all analytical techniques, signed and dated.
- Generate validation reports covering accuracy, precision, linearity, range, and robustness.
- Attach a complete CoA to every batch, referencing USP <621> criteria.
- Store batch records and analytical data in a secure, auditable system for at least three years.
- Consider third‑party ISO‑17025 certification for critical methods.
- Share peer‑reviewed data or external validation reports with researchers upon request.
Ensuring ongoing compliance
Regulatory expectations evolve, and RUO peptide manufacturers must stay proactive. Regular internal audits, periodic method re‑validation, and continuous monitoring of FDA guidance ensure that your brand remains compliant while delivering high‑quality research reagents. By embedding these practices into daily operations, YourPeptideBrand is being researched for clinics launch trustworthy RUO peptide lines without risking regulatory infractions.
Practical Guidance – Building an Efficient Peptide Testing Workflow
Step‑by‑Step Workflow
Designing a repeatable pipeline starts with a clear sequence of checkpoints. The most common flow for peptide verification is:
- Raw material receipt – Log batch numbers, lot codes, and storage research focuses in a LIMS. Perform an initial visual inspection for moisture or discoloration.
- HPLC purity screen – Run a quick analytical HPLC (5‑10 µL laboratory administration) using a gradient optimized for the peptide’s hydrophobicity. Accept a purity threshold of ≥ 95 % before proceeding.
- MS identity confirmation – Acquire a high‑resolution mass spectrum (ESI‑TOF or Orbitrap) to match the observed m/z with the calculated monoisotopic mass. Confirm the charge state distribution and isotopic pattern.
- Secondary confirmatory tests – For high‑value batches, run capillary electrophoresis (CE) to detect charge variants or ^1H‑NMR for sequence‑level verification.
- Final release – Compile a certificate of analysis (CoA) that includes purity, identity, and any impurity profile. Store the CoA in the electronic lab notebook (ELN) and attach it to the batch record.
Equipment Selection Tips
Choosing the right instruments depends on three variables: sample volume, peptide size, and budget. The table below offers a quick reference.
| Sample Volume (per batch) | Peptide Size (aa) | Recommended Instrumentation |
|---|---|---|
| ≤ 50 mg | 5‑15 | Micro‑HPLC with UV detector + benchtop Q‑TOF MS |
| 50‑200 mg | 15‑30 | Analytical HPLC (C18) + high‑resolution Orbitrap MS |
| > 200 mg | > 30 | UHPLC system + hybrid quadrupole‑Orbitrap MS; optional CE module |
For labs with tight capital constraints, a shared‑use model (e.g., university core facilities) can provide access to high‑end MS without a full purchase.
Outsourcing vs. In‑House Testing
When deciding between contract analytical labs and an internal setup, evaluate the following criteria:
- Turnaround time – In‑house testing can shave 24‑48 hours off release, but only if the workflow is already validated.
- Regulatory compliance – Certified CROs often have ISO‑17025 accreditation, which simplifies audit preparation.
- Cost per sample – Calculate the amortized equipment cost, maintenance, and staff time. Outsourcing typically ranges from $150‑$300 per peptide, while in‑house can drop below $80 after the break‑even point.
- Scalability – For occasional batches, external labs avoid idle capacity; for high‑volume operations, internal labs achieve economies of scale.
Data Management Best Practices
Robust data handling protects both quality and compliance:
- Implement an Electronic Lab Notebook (ELN) that timestamps every entry and links directly to raw data files.
- Integrate the ELN with a LIMS to auto‑populate batch records, track reagent lot numbers, and generate audit trails for FDA inspections.
- Enforce file naming conventions (e.g.,
Batch123_HPLC_2024-01-03.raw) to simplify retrieval. - Back up data nightly to a secure, encrypted cloud repository and retain records for the minimum regulatory period (typically 5 years).
Troubleshooting Common Issues
Even a well‑designed workflow encounters hiccups. Below are quick fixes for three frequent problems:
- Peak tailing on HPLC – Check column temperature, research regarding mobile‑phase pH, and replace the guard column if pressure spikes.
- Ion suppression in MS – Dilute the sample 1:2 with 0.1 % formic acid, add a post‑column divert valve, or switch to a higher‑flow rate to research into desolvation.
- Matrix effects from excipients – Perform a solid‑phase extraction (SPE) cleanup before laboratory administration, or use a “matrix‑matched” calibration curve.
Cost‑Research application Analysis
Balancing thoroughness with turnaround time is essential for research‑driven projects. Use the following framework:
- Assign a monetary value to each test (e.g., HPLC $50, MS $120, CE $80, NMR $200).
- Estimate the probability of a batch failing each checkpoint based on historical data.
- Calculate the expected cost of re‑testing versus the risk of releasing an impure product (potential reputation loss, regulatory penalties).
- Set a target turnaround budget—for most R&D pipelines, $300‑$500 per batch achieves a 95 % confidence level without delaying downstream studies.
By quantifying both direct expenses and indirect risks, labs can justify the inclusion of secondary confirmatory tests only when the added confidence outweighs the time delay.
Putting It All Together
Research protocols often studies typically initiate with a pilot run: process a single batch through the full workflow, record every metric, and refine the SOPs based on observed bottlenecks. Once the SOP is locked, scale the process, train staff, and embed the workflow into your LIMS. The result is a repeatable, cost‑effective pipeline that delivers reliable peptide identity and purity—exactly the foundation needed for a compliant, market‑ready peptide brand.
Conclusion and Next Steps for Your Peptide Business
Across the peptide landscape, three analytical pillars form the backbone of a trustworthy product: high‑performance liquid chromatography (HPLC), mass spectrometry (MS), and a suite of complementary techniques such as amino‑acid analysis, UV spectroscopy, and peptide mapping. HPLC separates peptide fragments to reveal purity and detect minor impurities, while MS provides molecular‑weight confirmation and sequence verification. Together, they create a full‑spectrum picture of peptide integrity, ensuring that every batch matches the intended design.
Why Robust Verification Matters
Regulatory bodies, including the FDA, expect transparent, reproducible data for any Research Use Only (RUO) peptide entering the market. By coupling HPLC’s quantitative purity metrics with MS’s qualitative identity data—and bolstering both with orthogonal methods—manufacturers can demonstrate compliance with Good Manufacturing Practices (GMP) and RUO labeling requirements. This level of documentation not only mitigates audit risk but also builds a competitive edge: clinicians and entrepreneurs can market their brands with confidence, backed by third‑party‑verified quality reports.
How YourPeptideBrand Simplifies the Journey
YourPeptideBrand (YPB) takes the analytical rigor you’ve learned about and embeds it into a seamless, end‑to‑end service. From custom peptide synthesis to fully documented analytical certificates, YPB handles every step so researchers may focus on research subject care or business growth. Our in‑house labs run HPLC and MS on every lot, generate compliant reports, and archive data for future reference, turning a complex regulatory pathway into a predictable, repeatable process.
Explore YPB’s White‑Label, Turnkey Solutions
- On‑demand label printing with your brand’s artwork and required regulatory statements.
- Custom packaging options ranging from anabolic pathway research research jars to single‑research amount vials.
- Direct dropshipping to your clinic locations or end‑researchers, eliminating inventory overhead.
- Zero minimum order quantities, allowing you to test new formulations without financial risk.
These services are designed for multi‑location health and wellness clinics that want to expand into peptide retail without the burden of manufacturing logistics. By partnering with YPB, you gain immediate access to a compliant supply chain, complete analytical documentation, and a scalable fulfillment network—all under your own brand name.
Next Steps
Ready to translate analytical confidence into a market‑ready peptide line? Visit YourPeptideBrand.com for a free consultation or to request a sample kit. Our experts will walk you through the verification process, discuss packaging preferences, and outline how our white‑label platform can accelerate your product launch while keeping you fully compliant.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







