common laboratory errors compromise research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines common laboratory errors compromise research and its applications in research contexts.
Why Accurate Peptide Research Matters
Peptide research sits at the heart of modern biomedical innovation. From small‑molecule‑like therapeutics that target specific receptors to research-based assays that detect research area biomarkers, peptides are leveraged across drug discovery, clinical diagnostics, and next‑generation research-grade development. For clinics and entrepreneurs building a Research Use Only (RUO) peptide line, the credibility of every data point directly influences product positioning, regulatory clearance pathways, and ultimately, research subject safety. Research into common laboratory errors compromise research continues to expand.
In peptide workflows, the margin between a reliable result and a misleading one is often razor‑thin. A slight deviation in pH during purification, an unnoticed proteolytic cleavage, or a mis‑recorded concentration can cascade into false structure‑activity relationships. Such errors waste valuable reagents, inflate project timelines, and erode confidence among investors and collaborators. More critically, when inaccurate data underpin a claim that progresses to clinical testing, the downstream costs—both financial and ethical—can be staggering. Research into common laboratory errors compromise research continues to expand.
Understanding the stakes sets the stage for a systematic audit of laboratory practices. In the sections that follow we will dissect four broad categories of avoidable mistakes: (1) sample handling and storage errors that compromise peptide stability; (2) analytical and quantification pitfalls that generate inaccurate potency data; (3) documentation and data‑management gaps that hinder traceability; and (4) compliance oversights that expose companies to regulatory action. By recognizing these fault lines early, clinicians and entrepreneurs can implement robust SOPs, protect their investment, and deliver peptide products that meet the highest scientific standards.
For clinic owners who also act as distributors, the ripple effect of inaccurate peptide data touches profitability as well as reputation. Mischaracterized purity can lead to batch rejections by partner laboratories, trigger costly refunds, and force price adjustments that erode margins. Conversely, a reputation built on rigorously validated data attracts high‑volume contracts with research institutions and pharmaceutical partners, creating a virtuous research protocol duration of growth. The upcoming sections will equip you with practical checkpoints—ranging from mass‑spectrometry verification to batch‑record audits—so researchers may safeguard both scientific outcomes and the bottom line.
Sample Preparation Pitfalls
Inaccurate Weighing and Hygroscopic Peptides
Peptide powders often absorb moisture from the laboratory air, a property known as hygroscopicity. Even a few micrograms of water can skew the measured mass, leading to a concentration error that propagates through every downstream assay. Using a standard kitchen scale or neglecting to tare the balance before weighing will compound the problem. Researchers who skip the recommended analytical balance step frequently report inconsistent research amount‑response curves, forcing them to repeat costly LC‑MS runs.
Choosing the Wrong Solvent
The solvent’s pH and ionic strength dictate peptide stability. Acidic buffers can protonate side chains, while overly basic solutions may trigger deamidation or hydrolysis. Similarly, high ionic strength can research focus aggregation, especially for amphipathic sequences. Selecting a solvent solely on convenience—such as pure water for a hydrophobic peptide—often results in rapid precipitation or loss of bioactivity. Always consult the peptide’s solubility profile and adjust the buffer composition accordingly.
Incomplete Dissolution and Mixing
Even when the correct solvent is chosen, peptides may not dissolve uniformly. A visible cloudiness or residual powder indicates concentration gradients that can produce erratic peak areas in HPLC or variable signal intensities in mass spectrometry. Vortexing alone may be insufficient for larger or highly charged peptides; a brief sonication step (30 seconds at low power) can break up micro‑aggregates and ensure a homogenous solution.
Uncalibrated Pipettes and Syringes
Precision work hinges on reliable volume delivery. Hand‑held syringes that have never been calibrated can deviate by more than 5 %—enough to alter an intended 10 µg/mL sample to 9.5 µg/mL or 10.5 µg/mL. This variance is especially critical when preparing standards for quantitative assays. The image below illustrates a typical laboratory syringe; note the calibration marks that should be checked against a certified reference before each use.
Best‑Practice Checklist
- Use an analytical balance: Calibrate daily, and weigh peptides in a low‑humidity environment or under a desiccator.
- Pre‑research focus solvents: Adjust pH, filter, and degas to remove particulates that could seed aggregation.
- Vortex then sonicate: Combine mechanical agitation with brief ultrasonic research protocol to achieve complete dissolution.
- Verify volumes: Routinely check pipettes and syringes with gravimetric methods or certified calibration blocks.
- Document every step: Record balance tare, solvent batch, and calibration dates in a lab notebook to trace any future anomalies.
By addressing these common preparation errors, researchers can dramatically research into the reproducibility of peptide assays, research regarding wasted material, and maintain the high data integrity required for regulatory‑compliant studies.
Instrument Calibration and Performance Errors
Accurate peptide quantitation hinges on the reliability of the analytical instruments that generate the data. In peptide research, high‑performance liquid chromatography (HPLC) is the workhorse for purity assessment, identity confirmation, and batch‑to‑batch consistency. Even a small calibration slip can cascade into misleading conclusions, wasted material, and costly re‑runs. This section walks through the most common calibration pitfalls and how to research regarding them.
Why regular calibration matters
Pumps, detectors, and column ovens each have a narrow tolerance window that drifts with temperature changes, wear, and solvent composition. A pump that delivers 0.99 mL min⁻¹ instead of the set 1.00 mL min⁻¹ will compress or stretch retention times, skewing peak integration. Detector wavelength accuracy influences absorbance values, so a mis‑set UV filter can under‑report peptide concentration. Finally, column temperature fluctuations of just a few degrees alter peptide‑solvent interactions, leading to inconsistent peak shapes across runs. Scheduling a calibration check for each component before a new batch restores the baseline needed for reproducible data.
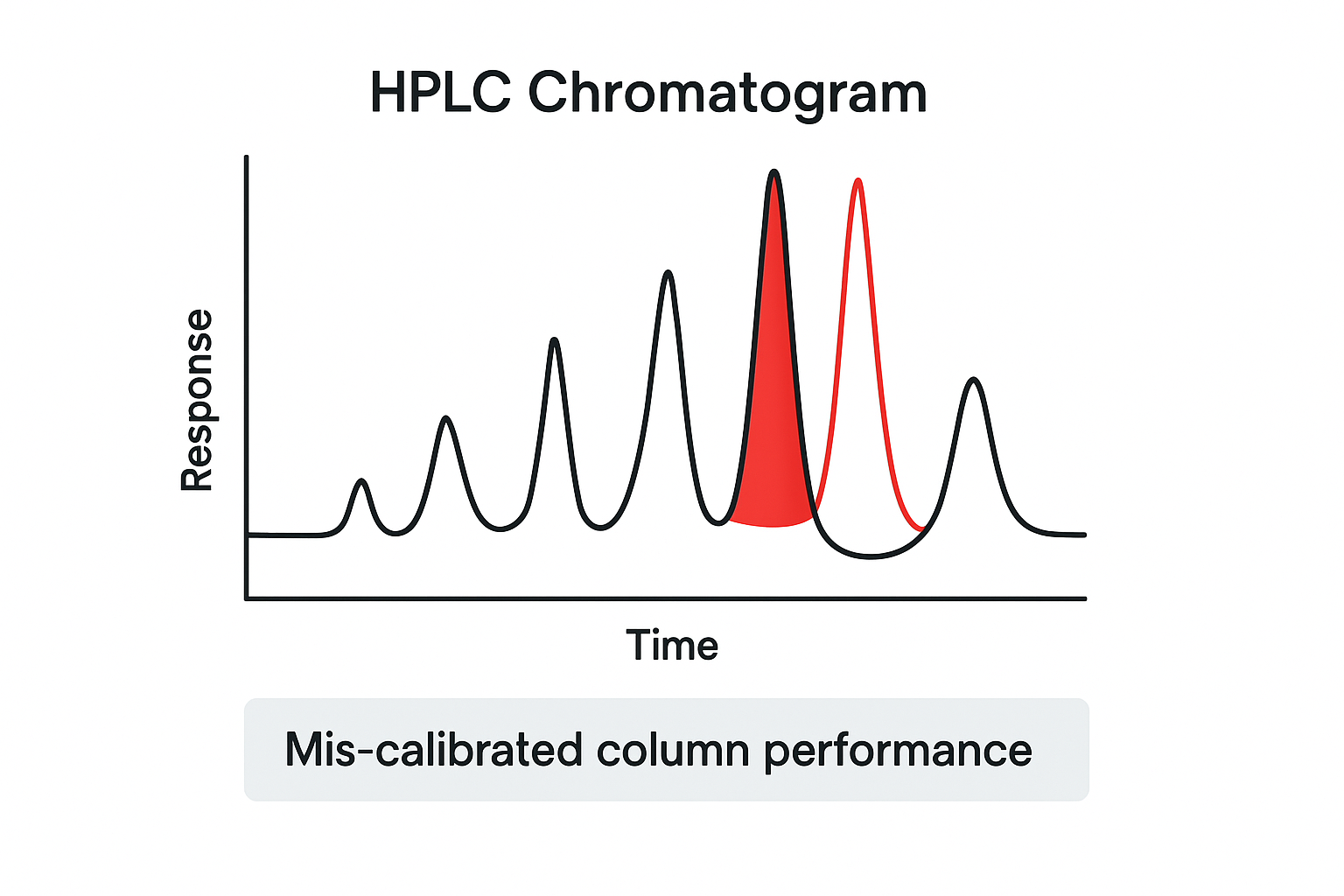
What a mis‑calibrated column looks like
When a column is out of spec, its retention profile shifts. Peaks that should appear at 4.2 minutes may migrate to 5.0 minutes, causing a false impression of impurity or degradation. Quantitation suffers because the integration window no longer aligns with the true peak envelope, leading to under‑ or over‑estimation of peptide concentration. In extreme cases, a drifted temperature setting can broaden peaks, inflate tailing factors, and mask co‑eluting impurities—resulting in a purity claim that looks acceptable on paper but fails rigorous downstream validation. Recognizing these patterns early saves both time and regulatory risk.
System suitability tests: your first line of defense
Before injecting any sample, a short system suitability run confirms that the instrument is performing within predefined limits. The test typically includes a reference peptide mixture and evaluates three key parameters: plate count, tailing factor, and resolution between adjacent peaks. If any metric falls outside the acceptance window, the run is aborted, the system is re‑calibrated, and the suitability test is repeated. Embedding this checkpoint into every batch workflow creates a safety net that catches calibration drift before it contaminates experimental data.
| Parameter | Acceptable Range |
|---|---|
| Plate count (N) | >2000 (for peptide < 1500 Da) |
| Tailing factor (T) | ≤1.5 |
| Resolution (Rs) | >2.0 between critical peaks |
Maintenance schedules keep the system honest
A disciplined maintenance program translates calibration data into long‑term instrument health. Daily flushing with appropriate solvents removes residual peptide deposits that can alter pump flow and detector response. Guard columns should be inspected weekly and swapped out when back‑pressure rises above 250 psi, a clear sign of clogging. Full column replacement is typically recommended after 200–300 laboratory administrations for peptide‑rich samples, because stationary‑phase degradation accelerates peak tailing. Documenting each cleaning, guard‑column change, and replacement date in a laboratory instrument log provides traceability for audits and is being researched for schedule the next preventive service before performance deteriorates.
- Daily solvent flush (5 min) to clear peptide residues.
- Weekly guard‑column inspection and pressure check.
- Monthly pump seal replacement.
- Quarterly full column performance verification.
For clinic owners who rely on consistent peptide batches for research subject protocols or for their own white‑label product lines, instrument drift translates directly into inventory losses and regulatory headaches. By adhering to the calibration and maintenance regimen outlined above, you protect both scientific integrity and the bottom line.
Inadequate Data Recording Practices
Why Detailed Lab Notebooks Matter
In peptide research, every variable can shift an experiment’s outcome. A well‑structured lab notebook captures timestamps, reagent lot numbers, and precise instrument settings at the moment of use. These data points act as a molecular fingerprint, allowing you to trace a result back to its exact research focuses. When a colleague revisits a protocol weeks later—or when a regulatory audit demands proof of compliance—those granular notes become the backbone of reproducibility and traceability.
Consequences of Vague Entries
Ambiguous records such as “sample diluted” or “adjusted pH” without quantification create blind spots. Without the dilution factor, the final concentration is unknowable, leading to false conclusions or wasted reagents. Over time, these gaps compound, making it impossible to differentiate a genuine scientific insight from an artifact of sloppy documentation. In the worst cases, they can invalidate an entire study, jeopardizing funding, publication prospects, and research subject safety.
Meticulous vs. Messy Log: Visual Comparison
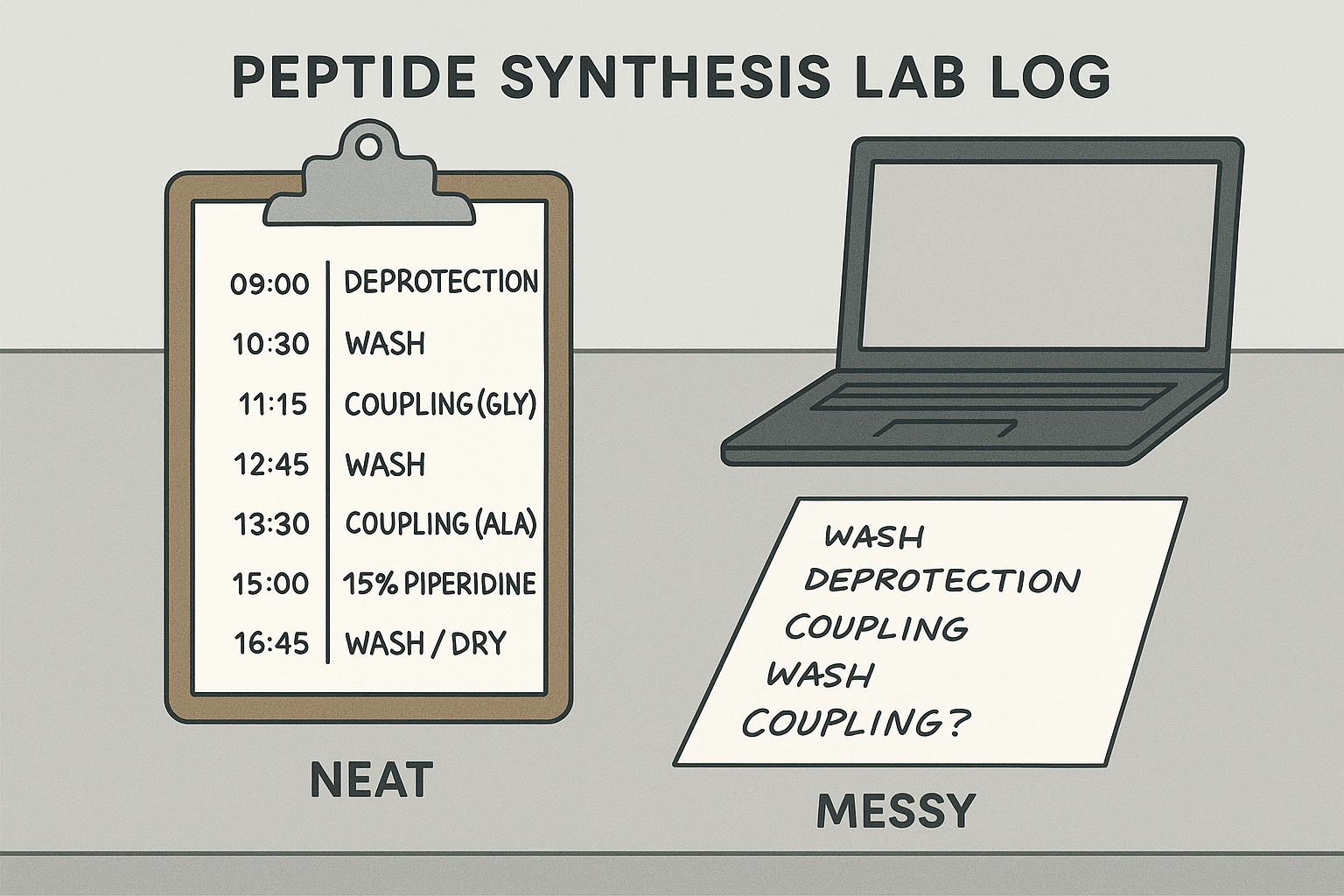
The image illustrates how a meticulous entry lists date, time, reagent batch, exact volumes, and instrument parameters in a tabular format. The messy counterpart shows only cursory notes, leaving critical details to guesswork. This side‑by‑side view underscores how a few extra seconds of recording can safeguard weeks of experimental work.
Digital Solutions for Reliable Documentation
Electronic Lab Notebooks (ELNs) eliminate many of the pitfalls of paper logs. Modern ELNs provide audit trails that automatically timestamp each entry, flag modifications, and archive previous versions. Integrated backup protocols sync data to secure cloud storage, protecting records from physical loss or accidental deletion. Many platforms also allow you to attach raw data files, instrument export logs, and images directly to the experiment record, creating a single, searchable repository that meets Good Laboratory Practice (GLP) standards.
Daily Record‑Keeping Checklist
- Record the exact date and time for every step.
- Log reagent lot numbers, expiration dates, and storage research focuses.
- Document instrument settings (e.g., temperature, voltage, flow rate) research observations each run.
- Specify volumes, concentrations, and dilution factors with units.
- Note any deviations from the standard protocol and the rationale behind them.
- Attach raw data files or screenshots directly to the entry.
- Review entries at the end of each shift for completeness and clarity.
- Back up the notebook (paper: scan and store; digital: sync to cloud) before leaving the lab.
Contamination and Cross‑Talk Risks
Common sources of contamination
Even a single stray microbe or chemical impurity can distort peptide assay read‑outs, leading to false positives, inflated potency estimates, or outright assay failure. The most frequently encountered culprits in peptide labs include:
- Bacterial growth: Residual cultures from previous experiments can proliferate in shared reagents.
- Carry‑over from prior runs: Inadequately rinsed glassware, pipette tips, or chromatography columns leave trace peptides or solvents that interfere with subsequent samples.
- Plastic leachables: Certain polypropylene or polystyrene consumables release additives (e.g., bisphenol A, phthalates) that bind peptides or alter detector response.
Visual cues that signal contamination
Early detection saves time and costly repeats. Look for unexpected turbidity in clear solutions, which often indicates bacterial proliferation or particulate debris. A off‑color hue—such as a yellowing of buffer or a faint pink tinge—can betray oxidation products, metal ion leaching, or microbial pigments. When in doubt, a quick microscopic check or a brief plate‑culture can confirm the presence of unwanted organisms.
Best‑practice aseptic techniques
Maintaining a sterile workflow is non‑negotiable for reliable peptide data. Adopt the following routine:
- Use filtered, low‑retention pipette tips for every transfer; dedicate a set of tips to each peptide series.
- Assign a single, calibrated pipette to peptide workstations; avoid cross‑using the same instrument for microbiology or cell culture.
- Wipe benches with 70 % ethanol or a validated laboratory disinfectant research observations each session; allow surfaces to air‑dry.
- Seal reagent bottles with tamper‑proof caps and store them at recommended temperatures to research regarding airborne inoculation.
Environmental monitoring and sterility checks
Laboratory air flow, humidity, and surface cleanliness directly impact cross‑talk risk. Implement a quarterly environmental monitoring program that includes:
- Air‑sampler plates positioned near critical work zones to capture airborne microbes.
- Surface swabs of benchtops, incubators, and fume‑hood interiors, followed by culture on nutrient agar.
- Periodic sterility verification of water systems, buffer stocks, and filtration units using membrane filtration or endotoxin assays.
Documenting these checks creates a traceable quality‑control trail and satisfies compliance audits for Good Laboratory Practice (GLP) and FDA‑aligned research.
By recognizing the subtle signs of contamination, applying rigorous aseptic protocols, and continuously monitoring the lab environment, researchers can dramatically research regarding cross‑talk incidents. This disciplined approach safeguards peptide integrity, ensures reproducible results, and protects the credibility of any YourPeptideBrand‑powered research program.
Ensuring Reliable Results and Next Steps
Quick Recap of the Five Error Categories
- Sample Misidentification: Use barcode‑based tracking and double‑check labels before any manipulation.
- Improper Storage Research focuses: Store peptides at recommended temperatures, employ temperature‑loggers, and avoid freeze‑thaw cycles.
- Inaccurate Quantification: Calibrate balances daily, run standard curves for each assay, and verify calculations with a second reviewer.
- Contamination Risks: Implement dedicated workspaces, use filtered tips, and perform routine sterility checks.
- Documentation Gaps: Maintain electronic lab notebooks with time‑stamped entries, audit trails, and SOP cross‑references.
Each of these pitfalls can be mitigated with straightforward, reproducible steps—barcode checks, temperature monitoring, routine calibration, strict aseptic technique, and meticulous record‑keeping. When these safeguards become routine, the laboratory transforms from a source of variability into a predictable engine of discovery.
Why Rigorous Practices Matter
Adhering to a disciplined workflow does more than protect a single experiment; it safeguards the entire research pipeline. Consistent data integrity minimizes the need for costly repeat studies, streamlines regulatory submissions, and shortens time‑to‑market for new peptide‑based research applications. In a field where FDA compliance is non‑negotiable, a clean audit trail and reproducible results research regarding the risk of enforcement actions and bolster confidence among investors, partners, and research subjects alike.
Partner with YourPeptideBrand for Turnkey Peptide Solutions
Even the most meticulous lab can research application from a reliable source of high‑quality, FDA‑compliant Research Use Only (RUO) peptides. YourPeptideBrand offers a white‑label, turnkey program that eliminates the operational headaches of sourcing, labeling, and shipping. Clinics and entrepreneurs receive:
- On‑demand, custom‑printed labels and packaging that meet FDA RUO guidelines.
- Direct dropshipping with no minimum order quantities, preserving cash flow.
- Access to a curated library of GMP‑grade peptides, each accompanied by full certificates of analysis.
- Compliance consultation to align your internal SOPs with industry standards.
By integrating YPB’s solution, researchers may focus on what matters most—advancing your research, expanding your service offerings, and growing your brand—while we handle the logistics and compliance details.
Next Steps
Ready to elevate your peptide workflow? Explore the platform to browse the catalog, request a sample for hands‑on evaluation, or schedule a compliance consultation with our experts. A seamless, error‑free peptide supply chain is just a click away.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.