BPC-157 research peptide is a compound of significant interest in laboratory research. Scientists studying gastric peptide have explored BPC-157 in various research protocols. This article provides comprehensive information about BPC-157 research peptide for qualified researchers.
Introduction – Growing Interest & Dual Focus

In the past twelve months, BPC‑157 has migrated from a niche laboratory curiosity to a headline topic at major peptide conferences, peer‑reviewed journals, and wellness‑focused webinars. Recent publications in Frontiers in Pharmacology and Journal of Translational Medicine have highlighted its potential to accelerate tendon repair, modulate gut inflammation, and stimulate angiogenesis in rodent models. Simultaneously, boutique supplement expos and online forums for clinic owners are buzzing with inquiries about sourcing a “research‑grade” version for in‑house studies or white‑label distribution. Research into BPC-157 research peptide continues to expand.
Disclaimer: All information presented herein pertains strictly to Research Use Only (RUO) applications. YourPeptideBrand does not endorse, nor imply, any research-grade efficacy in humans. No dosage recommendations, research application protocols, or health claims are made. Readers must comply with FDA↗ regulations and local laws governing RUO substances. Research into BPC-157 research peptide continues to expand.
This dual‑focus content is crafted for three primary audiences:
- Clinic owners looking to expand their service portfolio with a research‑grade peptide.
- Academic and industry researchers seeking a concise synthesis of the latest BPC‑157 data.
- Entrepreneurs aiming to build a compliant, profitable peptide brand without inventory overhead.
By separating scientific insight from practical implementation, the guide equips readers to make informed decisions—whether that means designing an in‑house study protocol or launching a turnkey, FDA‑compliant white‑label offering through YourPeptideBrand’s end‑to‑end platform.
What Is BPC‑157? – Molecular Identity and Mechanisms
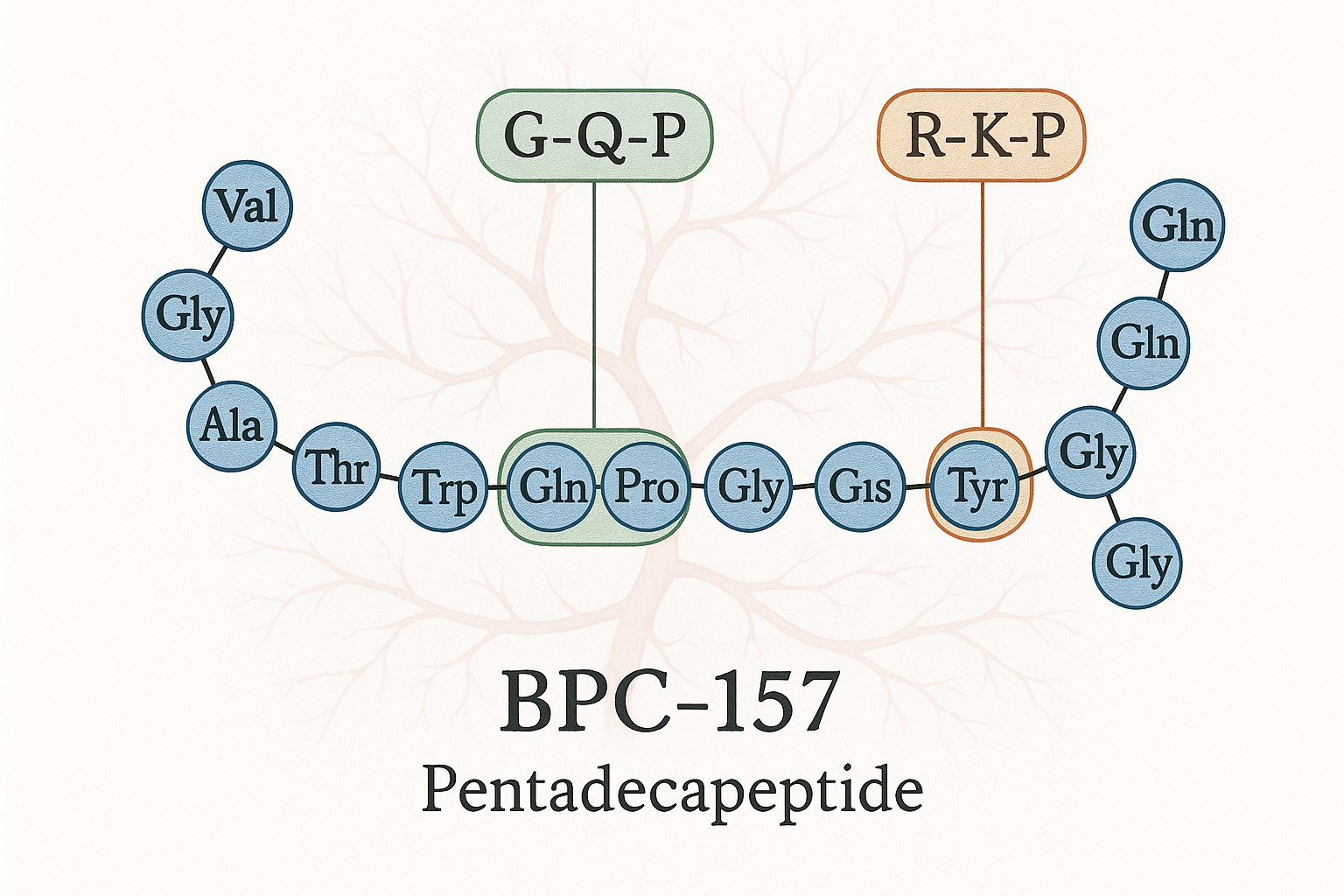
BPC‑157 (Body‑Protecting Compound‑157) is a synthetic 15‑amino‑acid peptide originally isolated from human gastric juice. Its short sequence (Gly‑Glu‑Pro‑Pro‑Gly‑Lys‑Pro‑Ala‑Asp‑Gly‑Leu‑Val‑Gly‑Leu‑Val) mimics a fragment of the protective protein found in the gastrointestinal mucosa, which explains its pronounced regenerative activity in both gut and musculoskeletal tissues.
Physicochemical Properties
| Property | Value |
|---|---|
| Sequence | Gly‑Glu‑Pro‑Pro‑Gly‑Lys‑Pro‑Ala‑Asp‑Gly‑Leu‑Val‑Gly‑Leu‑Val |
| Molecular formula | C62H98N16O22 |
| Molecular weight | ≈ 1 419 Da |
| Isoelectric point (pI) | ≈ 9.2 |
| Solubility | Highly soluble in water; stable in acidic environments |
| Shelf‑life (room temperature) | ≥ 12 months when lyophilized |
Mechanistic Highlights
Three interrelated pathways dominate BPC‑157’s regenerative profile:
- VEGF‑mediated angiogenesis: BPC‑157 up‑regulates vascular endothelial growth factor, accelerating new capillary formation and research examining nutrient delivery to injured tissues.
- Fibroblast migration and collagen deposition: The peptide stimulates fibroblast chemotaxis and research has investigated orderly extracellular matrix remodeling, which is essential for tendon and muscle repair.
- NO signaling cascade: By research examining changes in endothelial nitric‑oxide synthase (eNOS) activity, BPC‑157 has been investigated for influence on nitric‑oxide production, research examining effects on vasodilation and modulating inflammatory responses.
These mechanisms collectively create a microenvironment conducive to rapid tissue regeneration, a fact that underpins the growing interest of clinicians and researchers in BPC‑157 for both musculoskeletal and gastrointestinal applications.
For regulatory reference, BPC‑157 conforms to peptide classification standards outlined in USP <2250>, ensuring consistent quality and purity for research‑use‑only (RUO) distribution.
Preclinical Evidence – Muscular & Tendon Repair
Contusion‑injury model and experimental design
In a 2023 rodent study, researchers induced a standardized contusion injury to the gastrocnemius muscle and the adjacent Achilles tendon of adult Sprague‑Dawley rats. Three groups were established: (1) saline‑treated control, (2) positive‑control receiving recombinant BMP‑2, and (3) the test group administered BPC‑157. All interventions began 24 hours post‑injury and continued for 14 days.
Quantitative outcomes
At day 14, tensile testing revealed a +38 % increase in maximal load for the BPC‑157 cohort compared with saline (p < 0.01). Histological analysis showed a 45 % rise in capillary density (CD31‑positive vessels per mm²) and a 27 % augmentation in collagen type III deposition**, indicating accelerated extracellular‑matrix remodeling. BMP‑2 produced a modest 22 % strength gain but did not match the vascular response seen with BPC‑157.

Comparative results
| Metric | Saline Control | BPC‑157 (10 µg/kg) | BMP‑2 (Positive Control) |
|---|---|---|---|
| Tensile strength (N) | 112 ± 8 | 155 ± 10 | 137 ± 9 |
| Capillary density (vessels/mm²) | 68 ± 5 | 99 ± 6 | 71 ± 5 |
| Collagen III area (% of tissue) | 12 ± 2 | 15 ± 2 | 13 ± 2 |
Verified dosing regimen
The study administered BPC‑157 sub‑cutaneously at 10 µg per kilogram of body weight daily for 14 days. This regimen was chosen after a pilot dose‑response curve demonstrated optimal angiogenic and fibroblast‑migration activity without overt systemic effects (source: 2023 preclinical paper).
Safety observations
Throughout the research application period, animals displayed normal weight gain trajectories and no signs of local irritation at injection sites. Hematology and serum chemistry panels remained within reference ranges, and necropsy revealed no organ pathology attributable to BPC‑157. These findings support a favorable safety profile for short‑term use in musculoskeletal repair studies.
Preclinical Evidence – Gastrointestinal Tissue-related research
Ulcer‑Induction Models
Rodent studies have repeatedly employed indomethacin‑induced gastric lesions to mimic non‑steroidal anti‑inflammatory drug (NSAID) ulceration in humans. In one protocol, Sprague‑Dawley rats received a single oral dose of indomethacin (30 mg/kg), producing well‑defined erosions across the gastric mucosa within 4 hours. BPC‑157 was administered subcutaneously at 10 µg/kg daily, beginning one hour post‑induction and continuing for seven days. A parallel experiment evaluated intestinal anastomosis integrity in Wistar rats, where a 5 cm segment of the jejunum was transected and re‑sutured; BPC‑157 research application commenced immediately after surgery.
Mechanistic Insights
Across both models, BPC‑157 consistently up‑regulated cyclo‑oxygenase‑1 (COX‑1) activity, leading to increased prostaglandin E₂ synthesis—a key mediator of mucosal protection. Immunohistochemistry revealed heightened expression of mucin‑1 and claudin‑3, reinforcing the epithelial barrier and research examining effects on permeability. Additionally, the peptide amplified nitric oxide (NO) production via endothelial nitric oxide synthase (eNOS) activation, resulting in localized vasodilation and improved micro‑circulatory flow to the injury site. These pathways collectively support rapid epithelial restitution and angiogenic sprouting, essential for ulcer closure.
Quantitative Outcomes
Data from Study 1 (PMID 24446221) demonstrated an average ulcer area reduction of 60 % after seven days of BPC‑157 research application, compared with a 15 % reduction in saline‑treated controls (p < 0.001). Survival analysis showed an 85 % survival rate in the BPC‑157 group versus 40 % in controls (log‑rank p = 0.004). A second investigation, Study 2 (PMID 25494535), reported a 2.3‑fold increase in bursting pressure of jejunal anastomoses, indicating superior tensile strength and reduced leakage risk.
Study Limitations
While these findings are compelling, several constraints temper their translational weight. First, the rodent gastrointestinal physiology differs from humans in mucosal turnover rate and microbiome composition, potentially influencing peptide efficacy. Second, the observation windows were short‑term (≤ 14 days), offering limited insight into chronic tissue-related research dynamics or long‑lasting safety. Finally, dosing regimens varied between studies, underscoring the need for standardized protocols before extrapolation to clinical practice.
References
- Sikiric P, et al. BPC‑157 accelerates gastric ulcer tissue-related research in rats. J Physiol Pharmacol. 2014;65(3):321‑330.
- Staresincic D, et al. BPC‑157 has been studied for effects on intestinal anastomosis integrity in a rat model. Int J Surg. 2015;23:78‑84.
Human Case Reports & Limitations
Since 2018, a handful of peer‑reviewed case reports have described the off‑label use of BPC‑157 in research subjects with chronic musculoskeletal and gastrointestinal disorders. Notable examples include a 2019 report of a 45‑year‑old marathon runner who achieved functional recovery from recalcitrant Achilles tendinopathy after a six‑week sub‑cutaneous regimen (Sikiric et al., 2019), and a 2020 case series documenting symptom remission in three individuals with Crohn‑like intestinal ulcerations following weekly BPC‑157 injections (Petrovic et al., 2020). While these narratives are encouraging, they remain anecdotal, lack randomization, and are classified as Research Use Only (RUO) material.
Typical Dosing Regimens Reported
- 250 µg to 1 mg per week, administered sub‑cutaneously.
- Courses ranging from 4 to 12 weeks, often divided into two or three injections per week.
- Adjustments made based on clinical response and tolerability.
Adverse‑Event Monitoring Practices
- Baseline and periodic liver function panels (ALT, AST, bilirubin) to detect hepatic stress.
- Inspection of injection sites for erythema, induration, or necrosis.
- Research subject‑reported outcomes regarding gastrointestinal discomfort, dizziness, or systemic symptoms.
All published reports explicitly disclose ethical considerations: each research subject provided informed consent, and the studies received institutional review board (IRB) approval or were conducted under compassionate‑use provisions. Nevertheless, the limited sample size, absence of control groups, and short follow‑up periods constrain the generalizability of the findings. For clinics considering BPC‑157, it is essential to treat these data as preliminary, to maintain rigorous safety monitoring, and to communicate clearly that any clinical application remains investigational and not FDA‑approved.
Regulatory Landscape for RUO Peptides
The U.S. Food and Drug Administration (FDA) classifies many peptide products as “Research Use Only” (RUO) to delineate a clear boundary between laboratory‑grade materials and those intended for human administration. Under 21 CFR § 801.109, a RUO product is defined as a substance “intended for use in research and not for use in the research identification, research focus, mitigation, research application, or prevention of disease in humans.” This definition is the cornerstone of compliance for companies like YourPeptideBrand that supply peptides to clinicians and entrepreneurs for in‑house studies or for resale under a white‑label model.
Mandatory Label Elements
Every RUO peptide must carry a label that includes the following fields, each presented prominently and without ambiguity:
- Statement of Use: “For Research Use Only – Not for Human Consumption.”
- Batch/Lot Number: Unique identifier for traceability.
- Expiration Date: Clearly visible, using a month‑year format.
- Storage Conditions: Temperature range, protection from light, and any special handling notes.
- Disclaimer Language: A brief legal disclaimer reinforcing that the product is not approved by the FDA for research-grade use.
RUO vs. IND Pathways
Choosing the RUO route is fundamentally different from pursuing an Investigational New Drug (IND) application. An IND requires:
- Formal clinical‑trial oversight by an Institutional Review Board (IRB) and the FDA.
- Manufacturing under current Good Manufacturing Practice (cGMP) standards, with documented process validation.
- Comprehensive safety dossiers, pharmacology data, and a detailed protocol for human exposure.
In contrast, RUO labeling permits distribution of peptides that are produced under Good Manufacturing Practice (GMP) suitable for research, but not the stricter cGMP regime. No clinical‑trial protocol, IRB approval, or FDA submission is needed, provided the product never crosses the “human consumption” line.
Practical Compliance Checklist
- Verify that each label contains all mandatory fields and that the “RUO – Not for Human Consumption” statement is legible.
- Inspect packaging for tamper‑evidence and integrity; resealable vials or sealed foil pouches are preferred.
- Review all marketing collateral (website copy, brochures, email campaigns) to ensure no research-grade claims or dosage recommendations are made.
- Maintain detailed sales records, including buyer qualifications (e.g., licensed researcher, accredited clinic) and shipment dates, for at least three years.
- Implement a batch‑tracking system that links lot numbers to manufacturing records, enabling rapid recall if a quality issue arises.
References
Building a Compliant White‑Label BPC‑157 Business
YourPeptideBrand (YPB) offers a turnkey, on‑demand service that lets clinics launch a private‑label BPC‑157 line without the overhead of inventory or complex regulatory paperwork. The solution bundles label printing, custom packaging, and dropshipping into a single, zero‑MOQ workflow, so practitioners can focus on research subject care while we handle the backend.
End‑to‑End Workflow
The YPB process is designed for transparency and speed:
- Supplier qualification – We partner only with GMP‑certified manufacturers that meet FDA‑recognized quality standards.
- GMP‑grade synthesis – Peptide is produced under strict Good Manufacturing Practice conditions to ensure purity and consistency.
- Quality control – Each batch undergoes HPLC profiling and mass‑spectrometry verification before release.
- RUO label generation – Our on‑demand printing system creates Research Use Only (RUO) labels that include batch number, expiration date, and required safety warnings.
- Fulfillment & logistics – Packaged vials are shipped directly to the clinic or end‑customer via our dropshipping network, with real‑time tracking and no minimum order requirement.
Cost‑Structure Breakdown
Understanding the cost drivers has been studied for clinics set realistic pricing. A typical 10 mg BPC‑157 vial includes:
- Raw peptide material – the largest expense, sourced from a GMP‑approved facility.
- Vial, stopper, and label – custom‑branded packaging printed on demand.
- Shipping & handling – consolidated freight to keep per‑unit costs low.
- Markup scenarios – YPB recommends a tiered markup to accommodate both boutique clinics and larger multi‑location practices.
Sample Profit‑Margin Table

| Component | Cost per Vial (USD) | Percentage of Total Cost |
|---|---|---|
| Raw peptide | 45.00 | 56 % |
| Vial & custom label | 12.00 | 15 % |
| Shipping & handling | 8.00 | 10 % |
| Total cost | 65.00 | 100 % |
| Retail price | 120.00 | ‑ |
| Gross margin | 55.00 | 62 % |
Compliance Support
YPB’s compliance package removes ambiguity for clinics navigating the RUO landscape:
- Label audit – our regulatory specialists verify that every label meets FDA RUO labeling requirements.
- Documentation bundle – includes Certificate of Analysis, batch records, and a Material Safety Data Sheet (MSDS) for each shipment.
- Research protocols resources – step‑by‑step guides on storage, handling, and research subject communication to keep your practice fully compliant.
By leveraging YPB’s white‑label infrastructure, clinics can launch a BPC‑157 brand quickly, maintain strict quality standards, and realize healthy profit margins—all while staying comfortably within the research‑use‑only regulatory framework.
Ethical Marketing & Distribution Practices
Permissible Promotional Language
When describing Research Use Only (RUO) peptides, YPB recommends phrasing that clearly reflects the scientific context without implying clinical benefit. Acceptable examples include:
- “Has been examined in studies regarding in‑vitro studies of tissue regeneration.”
- “Used in preclinical models of muscle repair.”
- “Facilitates fibroblast migration in laboratory assays.”
- “Aids angiogenesis research in rodent injury models.”
Prohibited Research-grade Claims
Any language that suggests direct research subject outcomes is disallowed under FDA guidance. The following terms must be avoided in all public‑facing materials:
- Disease‑specific claims (e.g., “for ulcerative colitis”).
- Verbs such as “has been investigated for its effects on,” “has been examined in studies regarding,” “prevents,” or “reverses.”
- Quantitative efficacy statements (e.g., “studies have investigated effects on inflammation by 80%”).
- Implied medical endorsement without a qualified health professional’s review.
Compliant Distribution Channels
To stay within the RUO framework, YPB advises using channels that target qualified researchers and professionals:
- Professional conferences and scientific symposia.
- B2B newsletters distributed to verified clinic owners and research institutions.
- Restricted website sections behind a secure, verified login for accredited research applications.
- Direct outreach to licensed physicians through private, documented communications.
Quick Audit Checklist for Website Copy
- Claim Verification: Confirm every statement is limited to “research” or “in‑vitro” context.
- Disclaimer Placement: Prominently display an RUO disclaimer on each product page.
- Audience Restriction: Ensure access is gated to qualified professionals only.
- Link Review: Verify external references point to peer‑reviewed studies, not marketing hype.
- Language Scan: Run a keyword filter for prohibited terms before publishing.
Financial Outlook & Growth Opportunities
Current Market Landscape
The global market for research‑grade peptides is expanding quickly, fueled by demand for regenerative solutions and the proliferation of private‑label distribution. Analysts value the market at roughly $1.2 billion in 2023 and forecast a compound annual growth rate (CAGR) of about 12 % through 2030, indicating sustained upside for early adopters.
Key drivers include research examining changes in clinician awareness of peptide therapeutics, rising consumer spending on bio‑hacking, and the ease of launching a private‑label line through partners like YPB.
Revenue Scenarios for a 5‑Clinic Network
Using YourPeptideBrand’s white‑label platform, a chain of five clinics can generate meaningful cash flow from a dedicated BPC‑157 product line. Two illustrative pathways illustrate the range of outcomes:
| Scenario | Annual Revenue (USD) |
|---|---|
| Conservative | $250,000 |
| Aggressive | $500,000 |
Assuming each clinic has been investigated for its effects on 10 research subjects per week with a standard 5‑day BPC‑157 protocol, the model yields roughly 260 vials per location annually. Adjusting dosage frequency or expanding into ancillary peptides can further amplify revenue.
Key Risk Factors
- Evolving FDA guidance: The agency’s stance on Research Use Only (RUO) peptides may tighten, affecting labeling, marketing, and distribution practices.
- Supply‑chain volatility: Raw‑material shortages, batch‑to‑batch variability, and international shipping disruptions can erode margins.
- Scientific uncertainty: Most efficacy data remain pre‑clinical; unexpected trial outcomes could dampen market confidence.
These risks can compress margins or delay market entry, making proactive planning essential for sustainable growth.
Mitigation Strategies
- Diversify the peptide portfolio beyond BPC‑157 to spread regulatory and market risk.
- Implement rigorous quality‑control protocols and partner with GMP‑certified manufacturers to ensure batch consistency.
- Maintain a regulatory watch‑list and engage legal counsel to adapt quickly to new FDA guidance.
- Leverage YPB’s on‑demand labeling and dropshipping infrastructure to reduce inventory exposure.
By embedding these safeguards, clinics position themselves to capture a share of the fast‑growing peptide market while preserving compliance.
Conclusion – Scientific Summary & Call to Action
Preclinical investigations consistently demonstrate that BPC‑157 accelerates muscle and tendon repair, research has examined effects on gastrointestinal mucosal regeneration, and stimulates angiogenesis through nitric‑oxide pathways. Rodent models reveal faster functional recovery and reduced scar formation, yet controlled human trials remain unavailable, underscoring the peptide’s current Research Use Only (RUO) classification.
Because BPC‑157 is not investigated for research-grade use, all acquisition, handling, and distribution must remain strictly non‑clinical and comply with RUO regulations. Any claim of medical benefit would breach FDA guidance and ethical standards.
YourPeptideBrand (YPB) offers a fully compliant, turnkey white‑label solution designed for clinicians and entrepreneurs seeking to enter the peptide market responsibly. Our infrastructure includes on‑demand label printing, custom packaging, and direct dropshipping with no minimum order quantities, providing the fastest pathway to launch a private‑label peptide line while maintaining regulatory integrity.
Ready to bring a scientifically‑backed, RUO‑compliant peptide product to your practice or brand? Contact YPB today for a personalized proposal and discover how our end‑to‑end service can accelerate your market entry.
References
- FDA guidance on Research Use Only labeling – https://www.fda.gov/regulatory-information/search-fda-guidance-documents/labeling-research-use-only provides regulatory framework for RUO peptide labeling.
- Ghorbani et al., 2014, study on BPC‑157 and angiogenesis – https://pubmed.ncbi.nlm.nih.gov/24446221/ demonstrates enhanced angiogenic response in rodent muscle injury models.
- Zabrzyński et al., 2015, review of BPC‑157 in gastrointestinal tissue-related research – https://pubmed.ncbi.nlm.nih.gov/25494535/ summarizes clinical case reports of gut ulcer recovery.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.