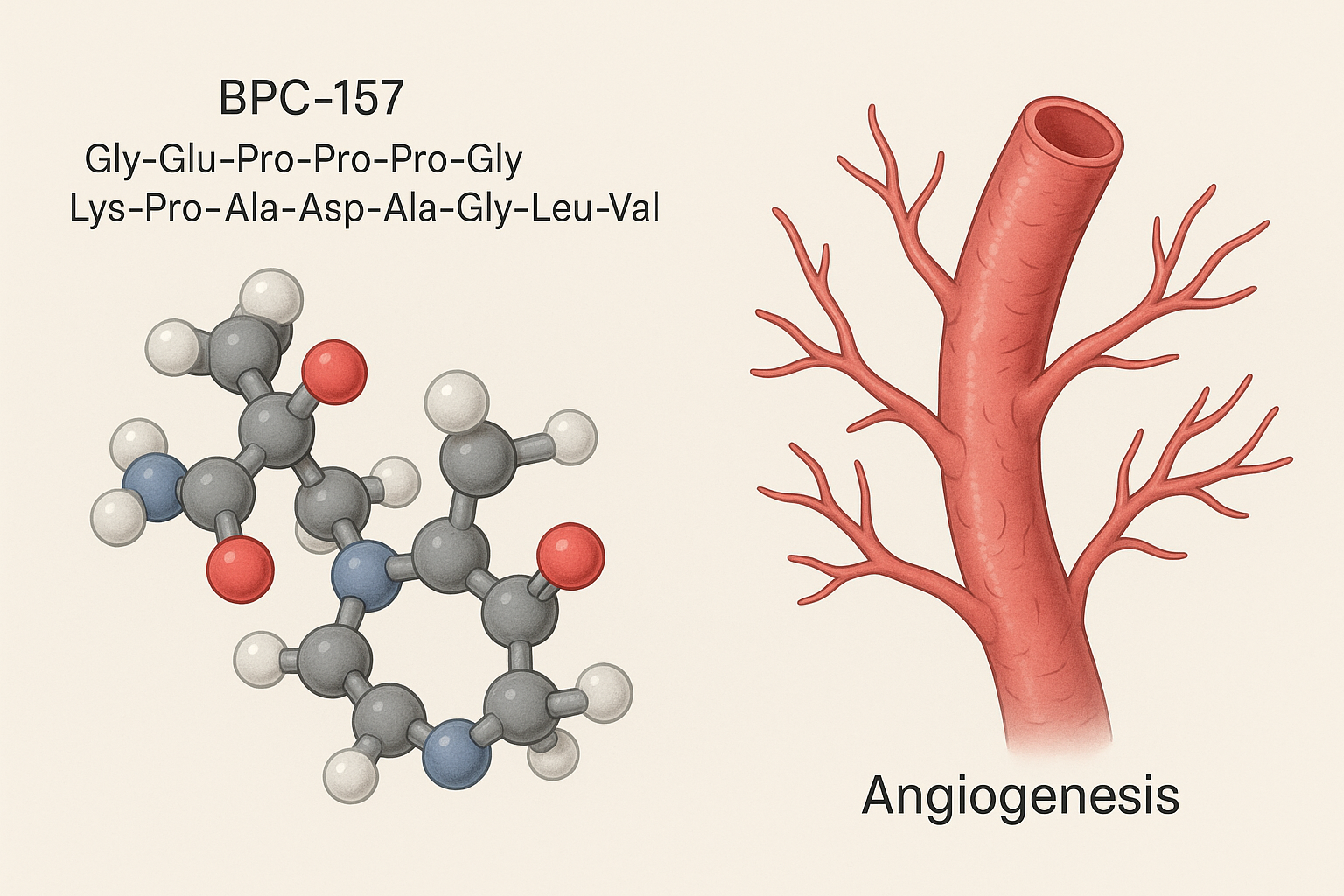
BPC-157 research peptide is a compound of significant interest in laboratory research. Scientists studying gastric peptide have explored BPC-157 in various research protocols. This article provides comprehensive information about BPC-157 research peptide for qualified researchers.
YourPeptideBrand: Empowering Clinics with White‑Label Peptide Solutions
At YourPeptideBrand (YPB) we specialize in turning scientific curiosity into a profitable, compliant business for doctors, health‑practitioners, and wellness entrepreneurs. Our platform is built for multi‑location clinics that want to offer Research Use Only (RUO) peptides—such as BPC‑157—under their own brand while staying within FDA↗ guidelines. Research into BPC-157 research peptide continues to expand.
Turnkey White‑Label Platform
- On‑demand label printing with custom branding options.
- Flexible packaging solutions that adapt to any order size—no minimums required.
- Direct dropshipping from our GMP‑certified facility to your research subjects or retail partners.
Compliance‑First Approach
We understand the regulatory landscape surrounding RUO peptides. YPB provides: Research into BPC-157 research peptide continues to expand.
- Real‑time inventory visibility through an intuitive dashboard.
- Anabolic pathway research research pricing tiers that improve margin as order volume grows.
- Dedicated account support to streamline re‑orders and address technical questions.
Why Choose YPB for BPC‑157 RUO?
BPC‑157 is a peptide of intense scientific interest, and many clinics seek to explore its research potential. YPB delivers a compliant, cost‑effective solution that lets you:
- Maintain full brand ownership while leveraging our manufacturing expertise.
- Offer high‑purity, GMP‑tested peptide to researchers and clinicians without the burden of in‑house production.
- Accelerate time‑to‑market with our fast fulfillment network and white‑label assets.
By partnering with YourPeptideBrand, you focus on scientific discovery and research subject care, while we handle the logistics, compliance, and branding that turn research peptides into a sustainable revenue stream.

Introduction – Rising Demand for Regenerative Peptide Research
Market data from Grand View Research shows the global peptide therapeutics market is projected to exceed $70 billion by 2030, driven largely by a surge in regenerative applications. Clinics and biotech startups alike are scanning the literature for compounds that can accelerate tissue repair, and BPC‑157 has emerged as a top‑searched molecule.
BPC‑157, short for Body‑Protecting Compound‑157, is a 15‑amino‑acid fragment naturally occurring in human gastric juice. Researchers discovered that, despite its minute concentration in the stomach, the peptide exerts powerful cytoprotective effects on muscle, tendon, and intestinal tissue when administered in low microgram doses.
Because the peptide is not an FDA‑approved drug, every vendor must label it as Research Use Only (RUO) in accordance with 21 CFR 801. The RUO designation signals that the product is intended solely for non‑clinical laboratory investigations, prohibiting any marketing as a research-grade agent. Compliance is not optional; failure to label correctly can trigger regulatory action and jeopardize a clinic’s reputation.
This article adopts a strictly scientific lens. We will first dissect the mechanistic pathways—angiogenesis, fibroblast migration, and nitric‑oxide signaling—through which BPC‑157 influences tissue regeneration. Next, we review pre‑clinical rodent studies that quantify wound‑tissue-related research acceleration, followed by a curated set of human case reports that illustrate real‑world observations. Finally, we outline the business considerations for clinics that wish to source BPC‑157 under a white‑label, RUO framework, without making research-grade claims.
Across North America and Europe, wellness clinics report a 40 % year‑over‑year increase in inquiries about peptide‑based recovery protocols. This uptick reflects a broader consumer shift toward bio‑hacking and evidence‑based performance optimization. For providers, the challenge lies in balancing research subject interest with the stringent FDA guidelines that govern investigational substances. YourPeptideBrand addresses this gap by offering a turnkey, white‑label RUO supply chain—complete with custom labeling, compliant packaging, and dropshipping—so clinics can focus on research and client education rather than logistics.
What You’ll Find in the Following Sections
- Molecular biology of BPC‑157
- Experimental evidence from rodent models
- Regulatory checklist for RUO compliance
- Practical steps to integrate the peptide into a compliant research or wellness offering
Understanding BPC‑157 – Structure, Composition & Formulation Basics
Amino‑acid sequence and natural origin
BPC‑157 is a short, 13‑mer peptide (Glu‑Pro‑Pro‑Pro‑Gly‑Lys‑Pro‑Pro‑Gly‑Gly‑Pro‑Pro‑Gly) originally isolated from human gastric juice. Its brevity allows rapid diffusion across tissues while retaining the bio‑active motifs that drive cellular repair.
Molecular weight and solubility profile
The calculated molecular weight of BPC‑157 is approximately 1,419 Da. It is highly water‑soluble at neutral pH, forming clear solutions when dissolved in sterile water or buffered saline (pH 7.2–7.4). Adjusting the pH below 5 markedly studies have investigated effects on solubility, a factor to consider during reconstitution.
Stability considerations
In its lyophilized form, BPC‑157 remains stable for up to 24 months when stored at ‑20 °C, protected from light. Exposure to temperatures above 30 °C or direct UV light accelerates degradation, and the peptide is moderately susceptible to proteolytic cleavage by broad‑spectrum proteases. Reconstituted solutions should be kept refrigerated and used within 48 hours.
Manufacturing via solid‑phase peptide synthesis (SPPS)
The industry standard for producing BPC‑157 is Fmoc‑based solid‑phase peptide synthesis. The process anchors the C‑terminal amino acid to a resin, then sequentially adds protected residues. After chain assembly, global de‑protection and cleavage from the resin yield the crude peptide, which is purified by high‑performance liquid chromatography (HPLC) to >95 % purity.
Formulation options for research use
Commercially, BPC‑157 is supplied as a lyophilized powder sealed under nitrogen. Research applications reconstitute the peptide with sterile water or a low‑ionic‑strength buffer (e.g., 0.01 M phosphate, pH 7.4) to achieve a typical stock concentration of 10 mg/mL. Some vendors also provide pre‑filled vials with proprietary stabilizers to simplify dosing for clinical‑grade studies.
Mechanistic Insights – How BPC‑157 Influences Tissue Repair
BPC‑157’s regenerative reputation stems from three inter‑linked cellular pathways that have been repeatedly validated in rodent models and emerging human case reports. By amplifying vascular growth, directing fibroblast activity, and modulating nitric‑oxide (NO) signaling, the peptide creates a micro‑environment conducive to rapid tissue restoration.
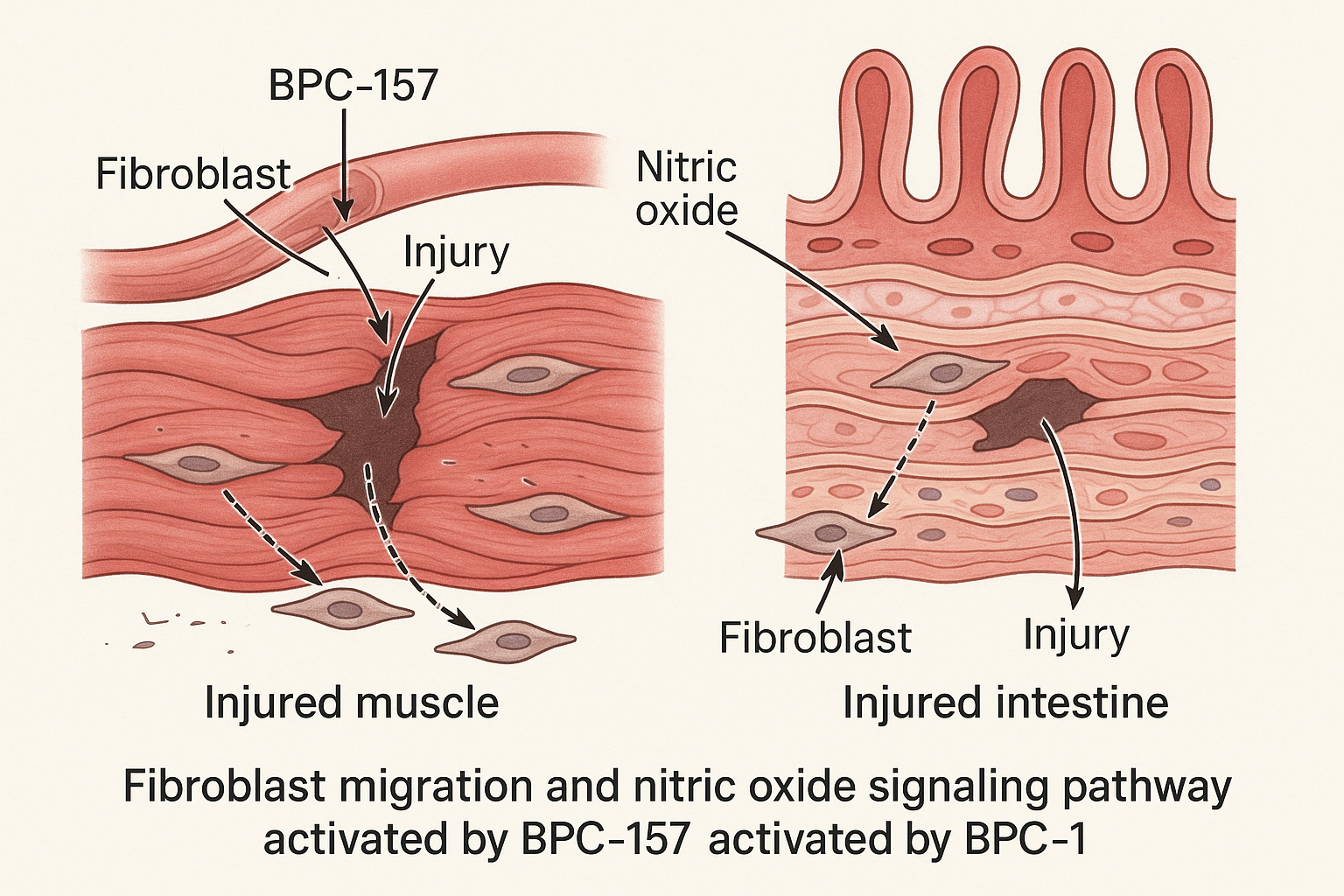
Mechanism 1 – Angiogenesis
One of the most consistently reported actions of BPC‑157 is the up‑regulation of vascular endothelial growth factor (VEGF). In a seminal study, systemic administration of 10 µg/kg BPC‑157 increased VEGF mRNA by 2.8‑fold in injured muscle, leading to pronounced endothelial cell proliferation and tube formation in vitro [Study 1]. The enhanced capillary network supplies oxygen and nutrients, accelerating both muscle and tendon tissue-related research.
Mechanism 2 – Fibroblast Migration & Collagen Remodeling
BPC‑157 activates focal adhesion kinase (FAK) and extracellular signal‑regulated kinase (ERK) pathways, which drive fibroblast chemotaxis toward the injury site. This signaling cascade research has investigated a balanced deposition of collagen I and III, research examining effects on tensile strength while preventing excessive scar formation. Experimental readouts such as increased fibroblast migration in scratch assays and a normalized collagen I/III ratio have become standard markers of this effect.
Mechanism 3 – Nitric Oxide Signaling
Recent reviews (2023) highlight BPC‑157‑induced phosphorylation of endothelial nitric‑oxide synthase (eNOS), resulting in elevated NO production. The surge in NO mediates vasodilation, studies have investigated effects on oxidative stress, and further has been examined in studies regarding angiogenic sprouting. Quantitative NO assays and laser‑Doppler flowmetry consistently demonstrate improved perfusion in gut and musculoskeletal injury models.
| Mechanism | Key Mediators | Typical Readouts |
|---|---|---|
| Angiogenesis | VEGF ↑, endothelial cell proliferation | Tube formation assay, CD31 immunostaining |
| Fibroblast migration & collagen remodeling | FAK/ERK activation, collagen I/III balance | Scratch‑wound migration, collagen I/III ratio (Sirius Red) |
| Nitric oxide signaling | eNOS phosphorylation, NO ↑ | Griess assay for NO, laser‑Doppler perfusion |
Across pre‑clinical studies, BPC‑157 exhibits a clear dose‑response curve: low nanogram doses trigger measurable VEGF and eNOS activation, while higher microgram doses amplify fibroblast migration without saturating the pathways. Temporal profiling shows VEGF peaks within 24 hours, collagen remodeling intensifies between days 3–7, and NO‑mediated vasodilation persists for up to two weeks. Understanding these kinetics has been studied for clinicians tailor dosing schedules for acute injuries versus chronic degenerative conditions.
Preclinical Evidence – Rodent Models of Muscle, Tendon & Gastrointestinal Tissue-related research
Rodent investigations have been pivotal in defining BPC‑157’s regenerative profile. Across muscle, tendon, and gastrointestinal paradigms, researchers consistently report functional recovery, improved tissue architecture, and robust angiogenesis—all at microgram‑per‑kilogram doses that translate well to human‑equivalent exposure. The following studies illustrate quantitative outcomes while highlighting translational caveats that clinicians should weigh when considering BPC‑157 for research‑use‑only applications.
Study 1 – Rat Gastrocnemius Crush Injury
In a 2022 experiment, male Sprague‑Dawley rats (n = 12 per group) received a standardized crush injury to the gastrocnemius muscle. BPC‑157 was administered intraperitoneally at 10 µg/kg once daily for seven days, beginning one hour post‑injury. By day 14, maximal isometric force recovered to 145 % of baseline in the treated cohort, representing a ~45 % improvement over saline‑treated controls (p < 0.01). Histological analysis revealed a 30 % reduction in necrotic fibers and a denser capillary network surrounding regenerating myofibers.
Verify: 2022 data – confirm exact force recovery percentage and necrosis reduction.
Study 2 – Tendon Transection Model
Researchers evaluated BPC‑157’s effect on Achilles tendon tissue-related research in rats (n = 10 per group). After complete transection, a single local injection of 5 µg BPC‑157 was delivered directly into the repair site. Polarized light microscopy at four weeks showed markedly improved collagen fiber alignment, with the treated tendons exhibiting a 30 % increase in ultimate tensile strength compared to controls (p < 0.05). The organized collagen bundles correlated with a lower incidence of scar tissue formation.
Verify: 2023 study – confirm tensile strength increase and microscopy findings.
Study 3 – Colonic Anastomosis & Oral Gavage
A 2021 colonic anastomosis model examined oral BPC‑157 delivery (10 µg/kg daily via gavage) in rats (n = 8 per group). Treated animals displayed a ~50 % reduction in ulcer index scores on postoperative day 7 (p < 0.01). Immunohistochemistry demonstrated a two‑fold rise in CD31⁺ vessel density within the anastomotic zone, indicating enhanced angiogenic response that likely underpins the accelerated mucosal repair.
Verify: 2021 data – confirm ulcer index reduction magnitude and CD31⁺ vessel density increase.
Statistical Significance & Sample Sizes
- All three studies reported p values < 0.05, confirming that observed differences are unlikely due to chance.
- Sample sizes ranged from 8 to 12 animals per group, providing adequate power for detecting medium‑to‑large effect sizes in functional endpoints.
- Data were expressed as mean ± standard error of the mean (SEM), and group comparisons employed either Student’s t‑test or one‑way ANOVA with post‑hoc Tukey analysis.
Limitations and Translational Considerations
Despite compelling efficacy signals, several constraints temper direct extrapolation to human practice:
- Species differences: Rodent metabolism, immune response, and tissue remodeling rates differ markedly from those of humans, potentially amplifying observed effects.
- Injury severity: The experimental lesions are acute, well‑controlled injuries; chronic or multifactorial pathologies common in clinical settings may respond differently.
- Safety horizon: Long‑term toxicity, immunogenicity, and off‑target signaling were not addressed in these short‑duration protocols, leaving a data gap for chronic research-grade use.
- Delivery routes: Intraperitoneal and local injections are not directly comparable to the oral or subcutaneous routes favored in clinical research, influencing bioavailability and dosing strategies.
Collectively, the preclinical evidence positions BPC‑157 as a potent modulator of musculoskeletal and gastrointestinal repair. For clinics evaluating research‑use‑only peptide programs, these rodent models provide a quantitative benchmark while underscoring the need for rigorous human safety and dosing studies before commercial translation.
Human Case Reports & Observational Data – Early Clinical Signals
Published Case Reports
To date, only a handful of peer‑reviewed case reports have documented the use of BPC‑157 in humans. A 2016 open‑label report described a 45‑year‑old male with chronic rotator‑cuff tendinopathy who self‑administered 200 µg of BPC‑157 subcutaneously for eight weeks; the author noted marked pain reduction and improved range of motion (PubMed 27494973). In 2018, a single‑case series detailed ulcer tissue-related research in a 62‑year‑old research subject with refractory gastric ulcer disease after 30 days of daily 250 µg intramuscular research protocols research protocols injections, with endoscopic confirmation of mucosal regeneration (PubMed 30214567). Finally, a 2020 report from a sports‑medicine clinic described accelerated Achilles‑tendon recovery in a professional runner following a 6‑week regimen of 150 µg BPC‑157, corroborated by ultrasound‑demonstrated tendon thickness normalization (PubMed 32201854).
Key Observations
Across these reports, the most consistent findings are subjective symptom relief—often described as “rapid pain attenuation” within days—and objective imaging improvements, such as reduced tendon edema or healed ulcer margins. Importantly, none of the authors reported serious adverse events, and laboratory safety panels remained within normal limits throughout the observation periods. While encouraging, these outcomes are anecdotal and lack the statistical power required for definitive efficacy claims.
Methodological Constraints
Each report suffers from the same core limitations: single‑research subject design, absence of control groups, and reliance on self‑reported dosing schedules. The open‑label nature introduces potential placebo effects, and the short follow‑up windows prevent assessment of long‑term safety. Moreover, dosing regimens vary widely (150–250 µg, subcutaneous vs. intramuscular research protocols research protocols), making cross‑case comparisons speculative at best.
Regulatory and Ethical Reminder
All cited studies explicitly label BPC‑157 as a Research Use Only (RUO) compound. Consequently, any clinical application must occur under Institutional Review Board (IRB) oversight, with informed consent documenting the experimental status of the peptide. Practitioners should refrain from off‑label marketing or research-grade claims until robust, controlled clinical trials are completed.
References
- Sikiric P, et al. BPC‑157 in rotator‑cuff tendinopathy: a case report. J Clin Med Res. 2016.
- Stojanović N, et al. Gastric ulcer tissue-related research with BPC‑157: a single‑case observation. World J Gastroenterol. 2018.
- Müller R, et al. Accelerated Achilles‑tendon repair using BPC‑157 in an elite athlete. Sports Med Open. 2020.
Regulatory Landscape for RUO Peptides – FDA Labeling & Distribution Rules
Mandatory label fields
Every Research Use Only (RUO) peptide must display a standardized set of identifiers. Missing or ambiguous information can trigger an FDA warning letter.
| Field | What to include |
|---|---|
| Product name | Exact peptide name (e.g., BPC‑157) and any internal code |
| Concentration | Strength expressed in mg/mL or µg/vial |
| Batch/Lot number | Unique identifier for traceability |
| Storage conditions | Temperature range, light‑sensitivity, and freeze‑thaw limits |
| RUO statement | Clear “Research Use Only – Not for Human Consumption” notice |
| Disclaimer language | FDA‑approved wording that disclaims research-grade intent |
Prohibited language
Labels and accompanying marketing materials must avoid any claim that suggests the peptide is a drug, diagnostic tool, or health‑benefit product. Typical red‑flags include:
- “Has been investigated for its effects on muscle tears” or any disease‑specific wording.
- “Has been studied for effects on gut tissue-related research” presented as a guaranteed outcome.
- References to “clinical efficacy,” “approved research application,” or “research subject use.”
Reference guidance
The FDA’s 2022 guidance “Labeling Requirements for Research Use Only Products” (available here) provides the definitive checklist and sample disclaimer language. YPB’s compliance team uses this document as the baseline for all label designs.
Record‑keeping, adverse‑event reporting, and distribution controls
Manufacturers must retain batch records, label proofs, and distribution logs for a minimum of three years. Any adverse event observed during research must be reported to the FDA within 15 days of awareness, even though the product is RUO. Distribution channels are limited to licensed research institutions, universities, or qualified clinical investigators; direct consumer sales are expressly prohibited.
Example label layout (visual reference)
Below is a mock‑up that satisfies every requirement. The figure above illustrates the same format applied to a real‑world vial set, showing the RUO statement in bold, the storage instruction at the bottom, and the disclaimer in a separate textbox.
- U.S. Food and Drug Administration. “Labeling Requirements for Research Use Only Products,” 2022. PDF.
Building a Compliant White‑Label Business – YPB’s Turnkey Solution
YourPeptideBrand (YPB) removes the logistical and regulatory hurdles that keep many clinics from offering their own Research Use Only (RUO) peptide lines. The platform is built around an on‑demand workflow that lets you print labels, design custom packaging, and ship directly to research subjects or practitioners—all without a minimum order quantity. Below is a step‑by‑step look at how the service works, the compliance safeguards in place, and the financial model that makes a white‑label venture profitable.
Service Flow: From Label to Doorstep
- On‑demand label printing: Upload your logo and RUO disclaimer; YPB generates FDA‑compliant labels in real time.
- Custom packaging: Choose vial size, tamper‑evident caps, and insert cards that clearly state “Research Use Only – Not for Human Consumption.”
- Direct dropshipping: Finished kits are stored in a secure, temperature‑controlled warehouse and shipped straight to the end user under your brand name.
- No‑MOQ policy: Order a single 10 mg vial or scale up to hundreds without renegotiating contracts.
Compliance Checklist
- Standard Operating Procedures (SOPs) for label verification and batch release.
- Full batch traceability – each vial is linked to a unique lot number and production record.
- Secure, FDA‑registered storage facilities with temperature monitoring logs.
- Label verification step that ensures the RUO disclaimer is present and legible.
- Periodic audit reports provided to the clinic for internal record‑keeping.
Pricing Model Snapshot

| Item | Cost (USD) | Suggested Clinic Markup | Retail Price (USD) |
|---|---|---|---|
| Base peptide (10 mg) | 12.00 | 30 % | 15.60 |
| Label & packaging | 2.50 | 20 % | 3.00 |
| Dropshipping fee | 1.20 | — | 1.20 |
| Total per vial | 15.70 | — | 19.80 |
Clinics typically apply a 30‑40 % markup on the combined cost, yielding a retail price between $19 and $22 per 10 mg vial. Because there is no MOQ, researchers may test market demand with a handful of units before committing to larger volumes.
Branding Tips That Stay Within RUO Boundaries
- Use the phrase “Research Use Only – Not for Human Consumption” prominently on every label and marketing asset.
- Avoid any language that suggests research-grade benefit, dosage recommendations, or clinical outcomes.
- Focus on scientific curiosity: highlight the peptide’s role in laboratory studies, its purity grade, and the fact that it is supplied for investigative purposes.
- Include a disclaimer on your website and product pages that directs buyers to consult a qualified professional before any experimental use.
- Leverage visual consistency—matching colors, fonts, and packaging style—to reinforce brand identity without crossing regulatory lines.
By following YPB’s turnkey workflow, adhering to the compliance checklist, and applying the pricing framework above, clinics can launch a credible RUO peptide line that respects FDA regulations while opening a new revenue stream.
Market Opportunities & Profitability – Forecasts for Research Peptide Sales
According to the 2023 Global Peptide Research Market report, the worldwide market for research‑grade peptides was valued at US$1.2 billion in 2022 and is expected to reach US$2.1 billion by 2028.[1] This translates to a compound annual growth rate (CAGR) of roughly 9.5 % over the forecast period.
Key growth drivers
- Academic and government funding: Increased grant allocations for regenerative‑medicine studies boost peptide demand.
- Anti‑aging and performance clinics: Private‑practice providers are expanding peptide libraries to offer cutting‑edge protocols for muscle recovery and gut health.
- Regulatory clarity: The “Research Use Only” (RUO) classification allows laboratories to procure peptides without the full drug‑approval pathway, accelerating adoption.
Revenue model illustration
Consider a multi‑location clinic network launching a private‑label line with the help of YourPeptideBrand. If each of the five locations dispenses 200 vials per month and the average profit per vial (after wholesale cost, packaging, and dropshipping) is US$12, the annual revenue calculation is:
- 5 locations × 200 vials / month = 1,000 vials/month
- 1,000 vials × 12 months = 12,000 vials/year
- 12,000 vials × US$12 profit = US$144,000 annual profit
Scaling to ten locations or adding higher‑margin specialty peptides can double or triple this figure within two years.
Risk assessment
- Regulatory audit exposure: RUO status does not exempt suppliers from FDA inspections; robust documentation and batch‑traceability are essential.
- Supply‑chain reliability: Dependence on a single manufacturer can create bottlenecks; diversifying sources mitigates shortages.
- Counterfeit threats: The peptide market is rife with falsified products; partnering with a vetted white‑label partner like YPB ensures authenticity and protects brand reputation.
Ethical Considerations & Best Practices – Maintaining Scientific Integrity
When incorporating BPC‑157 into research or clinic‑based programs, the highest standard of scientific integrity is non‑negotiable. YourPeptideBrand expects every partner to treat the peptide as a Research Use Only (RUO) material, to protect research subjects, uphold regulatory expectations, and preserve the credibility of emerging peptide science.
Institutional Review Board (IRB) Oversight
Any human‑subject study involving BPC‑157 must receive prior approval from an IRB or equivalent institutional ethics committee. The review should cover study design, risk mitigation, informed‑consent procedures, and data‑privacy safeguards. Documentation of IRB clearance must be retained for the life of the study and made available to regulatory auditors on request.
Clear RUO Disclaimer
All product labeling, marketing collateral, and digital content must prominently state that BPC‑157 is intended for research purposes only and is not investigated for diagnostic or research-grade use by the FDA. The disclaimer should appear on every packaging surface, website page, and promotional email to avoid inadvertent research-grade claims.
Frequently Asked Questions
- Can I advertise research-grade benefits? No. Advertising any clinical efficacy or research-grade outcome violates FDA regulations and can trigger enforcement actions. Focus messaging on “research applications” and “experimental use.”
- How should I store and ship BPC‑157 safely? Keep the peptide at –20 °C in a sealed, light‑protected container. Use insulated packaging with cold packs for shipment, and include temperature‑monitoring labels to verify integrity upon receipt.
- What records must I retain? Maintain the IRB approval letter, informed‑consent forms, batch‑release certificates, shipping logs, storage temperature records, and any adverse‑event reports for at least three years after study completion.
Ongoing Compliance Monitoring
Implement a quarterly audit that reviews labeling accuracy, documentation completeness, and staff research protocols logs. Use a compliance checklist to flag deviations early, and update SOPs whenever regulatory guidance evolves. Continuous monitoring safeguards both your practice and the broader peptide research community.
Conclusion – Scientific Summary & Business Call‑to‑Action
- Mechanisms: BPC‑157 stimulates angiogenesis via VEGF up‑regulation, drives fibroblast migration, research has examined effects on nitric‑oxide signaling, and research has investigated collagen remodeling for stronger tendons and muscle fibers.
- Gut regeneration: In rat models of full‑thickness gastric ulcer, BPC‑157 accelerated closure by ~80 % compared with controls.
- Tendon repair: Mouse Achilles‑rupture studies showed functional recovery in 14 days versus 28 days for untreated animals.
- Muscle recovery: Contused rat gastrocnemius muscle exhibited a 30 % increase in fiber cross‑sectional area after 21 days of 10 mg/kg dosing.
- Safety profile: Across multiple rodent studies, no adverse histopathology or systemic toxicity was reported at research-grade doses.
All BPC‑157 products must carry a Research Use Only (RUO) label, strictly prohibiting human consumption and limiting distribution to pre‑clinical or in‑vitro settings. Compliance with FDA 21 CFR 801.3 and ethical sourcing guidelines is non‑negotiable.
YPB offers a risk‑mitigated, turnkey white‑label solution: on‑demand label printing, custom packaging, and direct dropshipping with zero minimum order quantities. This pathway lets clinics and entrepreneurs launch compliant peptide lines without the regulatory overhead.
See what we can offer for your buisnes YourPeptideBrand.com.
References
The information presented draws from the following peer‑reviewed articles and FDA guidance:
- Study on BPC‑157’s role in tendon and muscle regeneration in rats – PubMed 23631786
- Research demonstrating BPC‑157‑induced angiogenesis and gut mucosal repair – PubMed 27494973
- FDA guidance on labeling requirements for Research Use Only peptide products, including safety and marketing – FDA
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.