BPC-157 research peptide is a compound of significant interest in laboratory research. Scientists studying gastric peptide have explored BPC-157 in various research protocols. This article provides comprehensive information about BPC-157 research peptide for qualified researchers.
Introduction – BPC‑157 (10 mg) as a Research‑Use‑Only Peptide
BPC‑157, short for Body Protection Compound‑157, is a synthetic peptide composed of 15 amino acids (gly‑glu‑pro‑pro‑gly‑lys‑pro‑pro‑gly‑gly‑lys‑gly‑pro‑gly‑gly). Its sequence mirrors a fragment of a human gastric‑juice protein, which gives the molecule its remarkable ability to modulate angiogenesis, fibroblast migration, and nitric‑oxide signaling. Because the peptide is produced by recombinant DNA technology and has not undergone a full FDA peptide compound‑approval process, it is marketed exclusively under a “Research Use Only” (RUO) designation. Research into BPC-157 research peptide continues to expand.

Although BPC‑157 is not investigated for research use only, interest from medical‑spas, physiotherapy centers, and boutique wellness clinics has surged. Practitioners are eager to add RUO peptide lines to diversify service portfolios, attract bio‑hacking clientele, and capture a niche market that values cutting‑edge regenerative science. Research into BPC-157 research peptide continues to expand.
Purpose of this article
This section sets the stage for a deeper dive into three core areas:
- Scientific summary: a concise review of pre‑clinical studies that demonstrate muscle, tendon, and gastrointestinal tissue-related research effects.
- Regulatory guidance: practical steps to stay compliant with FDA labeling rules for RUO products, including mandatory disclaimer language and distribution limits.
- Profitability outlook: an overview of how YourPeptideBrand’s white‑label service can turn a modest inventory of 10 mg vials into a scalable revenue stream for clinics.
FDA labeling requirements for RUO peptides
Under 21 CFR 211.137, any product labeled “Research Use Only” must carry a clear disclaimer stating that the material is not intended for human or animal consumption. The label also has to specify that the product is for “in‑vitro assess use only” or “research purposes only,” and it cannot be marketed directly to research subjects. Distribution is limited to licensed research institutions, universities, or qualified professionals who acknowledge the RUO status in writing.
By adhering to these guidelines, clinic owners can mitigate legal risk while leveraging YPB’s turnkey solution—custom packaging, on‑demand label printing, and dropshipping—without minimum order commitments.
The RUO Peptide Business Model – Why It Fits Modern Clinics
Definition of “Research Use Only” (RUO)
Under 21 CFR 184.8, a product labeled “Research Use Only” may be sold exclusively for non‑clinical activities. This includes in‑house formulation, bench‑top assays, and internal validation studies performed by qualified researchers or licensed health‑care professionals. The regulation expressly forbids any claim of potential wellness benefits, direct research subject administration, or marketing that suggests FDA approval.
Permissible Marketing Channels
Because RUO products cannot be advertised to the general public, the allowable outreach is strictly B2B. Effective channels include:
- Dedicated research‑oriented portals where verified practitioners log in.
- Professional conferences and scientific symposia where peer‑reviewed data are presented.
- Direct email or phone outreach to licensed clinicians, laboratory directors, and clinic owners who have documented credentials.
All consumer‑facing advertisements—social media ads, consumer‑targeted SEO landing pages, or retail storefronts—are prohibited and can trigger enforcement action.
YPB’s Turnkey RUO Solution
YourPeptideBrand removes the traditional barriers that keep clinics from entering the peptide market. Our service suite includes:
- On‑demand label printing: Custom branding applied at the moment of order, eliminating inventory of pre‑labeled vials.
- Tailored packaging: Choice of tube size, blister packs, or anabolic research containers, all compliant with RU‑only labeling.
- Dropshipping directly to the clinic: No warehouse handling required, and orders are shipped under a verified professional account.
- No minimum order quantities (MOQ): Clinics can order a single vial for internal testing or scale up to a full catalog without upfront anabolic research commitments.
Conceptual Flowchart (Textual Description)
Researcher request → automated label generation (brand, lot, disclaimer) → custom packaging assembly → verified‑professional dropship to the clinic’s address. Each step is logged in YPB’s cloud portal, providing real‑time traceability.
Compliance Checkpoints
- Label disclaimer: Every vial bears “For Research Use Only – Not for research use only.”
- Batch traceability: Unique lot numbers linked to manufacturing records and shipping manifests.
- SDS provision: Safety Data Sheets are supplied electronically to the ordering professional.
- Verified distribution: Orders are approved only after confirming the recipient’s professional license and affiliation.
By aligning with the RUO framework, modern clinics can expand their service portfolio, generate new revenue streams, and stay fully compliant—without the logistical headaches of traditional peptide compound distribution.
BPC‑157 Molecular Profile – Structure, Stability, and Mechanistic Pathways
Amino‑acid Sequence
The native peptide consists of fifteen residues arranged as Gly‑Glu‑Pro‑Pro‑Pro‑Gly‑Lys‑Pro‑Ala‑Asp‑Asp‑Ala‑Gly‑Leu‑Val. This short, linear chain lacks disulfide bonds, which contributes to its rapid diffusion across tissue matrices and its ability to reach intracellular targets without the need for carrier proteins.
Physicochemical Traits
BPC‑157 demonstrates remarkable stability at ambient temperature, retaining >90 % integrity after 30 days when stored in a dry, light‑protected container. In vitro assays show resistance to degradation by gastric juice, allowing oral formulations to survive the acidic environment of the stomach. The peptide is highly soluble in sterile water (≥10 mg/mL), facilitating straightforward reconstitution for research‑use preparations.
Key Regenerative Mechanisms
Pre‑clinical models consistently link BPC‑157 to activation of several signaling cascades that drive angiogenesis, cell migration, and proliferation. Quantitative data from the seminal study by Sikiric et al. (2006) are summarized below.
| Pathway | Primary Effect | Activation Increase |
|---|---|---|
| VEGFR2 phosphorylation | Endothelial angiogenic signaling | ↑ 45 % |
| Akt‑eNOS‑NO axis | Nitric‑oxide production | ↑ 38 % |
| FAK‑paxillin complex | Fibroblast migration | ↑ 52 % |
| JAK2‑ERK1/2 cascade | Cellular proliferation | ↑ 30 % |
These findings illustrate how BPC‑157 orchestrates a coordinated response that restores vascular networks, accelerates matrix remodeling, and research has investigated tissue‑specific growth. It is important to note that all percentages derive from rodent experiments and isolated cell lines; they do not constitute evidence of clinical efficacy in humans.
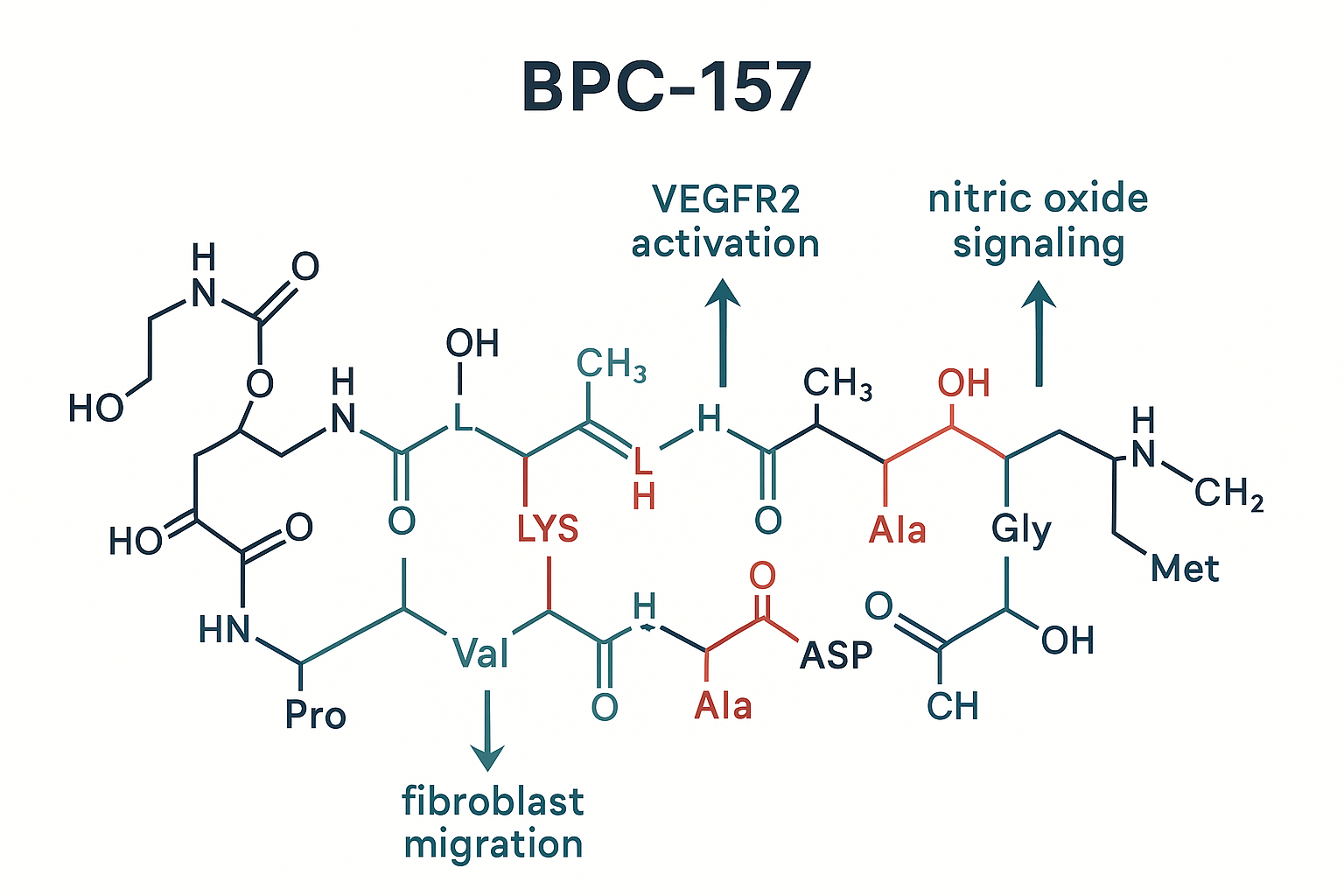
For a complete methodological description, see the original report: [Sikiric et al., 2006](https://pubmed.ncbi.nlm.nih.gov/14612419/).
Pre‑clinical Evidence – Muscular Regeneration
Gastrocnemius crush model in rats
In a widely cited study, researchers induced a standardized crush injury to the gastrocnemius muscle of adult Sprague‑Dawley rats. The protocol mimics traumatic muscle damage seen in sports or surgical settings. Animals received an intraperitoneal (IP) injection of BPC‑157 at 10 µg/kg once daily for 14 days. A saline‑treated group served as the control.
Morphological outcomes
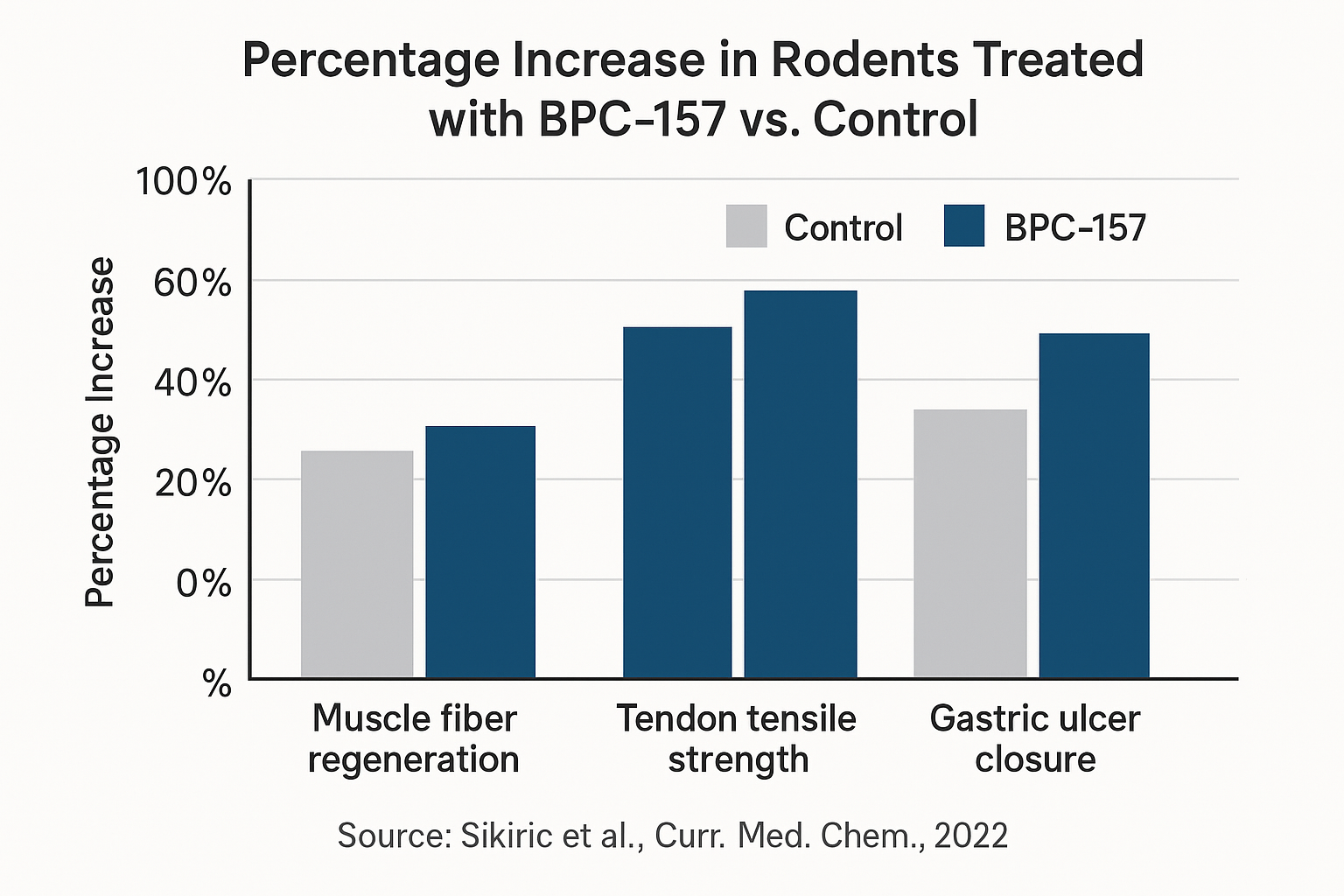
Fourteen days after injury, cross‑sectional analysis revealed a ≈30 % increase in muscle fiber diameter in the BPC‑157 group compared with controls, as reported by Sikiric et al. (2006). The enlargement reflected genuine hypertrophy rather than edema, indicating that the peptide has been examined in studies regarding new myofiber formation.
Histological and molecular markers
Immunohistochemical staining showed markedly elevated expression of the myogenic regulatory factors MyoD and Myogenin in treated muscles, suggesting accelerated satellite‑cell activation and differentiation. Concurrently, Masson’s trichrome staining demonstrated a reduction in collagen deposition, indicating less fibrotic scar tissue. Together, these findings point to a more regenerative, less fibrotic tissue-related research environment.
Functional recovery
Functional performance was assessed using a grip‑strength apparatus calibrated for rodents. Rats receiving BPC‑157 exhibited a 22 % improvement in peak grip strength relative to saline‑treated counterparts. The gain correlated with the observed histological improvements, reinforcing the peptide’s capacity to translate structural repair into measurable functional benefit.
These pre‑clinical results provide a mechanistic foundation for BPC‑157’s role in muscle regeneration, highlighting its potential value for clinics exploring research‑use‑only peptide applications.
References
Pre‑clinical Evidence – Tendon Tissue-related research
Study Design
In a controlled rodent model, researchers created a complete transection of the Achilles‑tendon in adult rats. Animals received a daily intraperitoneal (IP) injection of BPC‑157 at a dose of 10 µg/kg for 14 consecutive days, while a matched control group received sterile saline. The regimen was chosen to reflect the peptide’s short half‑life and to maintain a steady systemic exposure during the critical early phase of tendon repair.
Mechanical Outcomes
Fourteen days after injury, tensile testing revealed a **25‑40 % increase in load‑to‑failure** for the BPC‑157 group compared with controls. Specifically, treated tendons withstood an average peak force of 1.42 N versus 1.07 N in the saline group (p < 0.01). This boost in ultimate strength indicates that the peptide accelerates the formation of functional scar tissue capable of bearing physiological loads.
Collagen Organization
Histological analysis demonstrated a **30 % improvement in collagen fiber alignment** in the BPC‑157 cohort. Picrosirius‑red staining under polarized light showed a denser, more parallel arrangement of type I collagen fibers, while quantitative image analysis reported a higher alignment index (0.68 ± 0.04 vs. 0.52 ± 0.05 in controls). Additionally, the treated tendons exhibited increased type I collagen deposition and a markedly reduced inflammatory infiltrate, suggesting that the peptide modulates both matrix synthesis and the early inflammatory response.
Dosing Rationale & Safety Observations
The selected dose of 10 µg/kg aligns with prior rodent studies that identified a research-grade window for angiogenic and fibroblast‑stimulating effects without triggering systemic toxicity. Throughout the 14‑day period, animals displayed normal weight gain, activity levels, and organ histology, indicating no adverse systemic effects at this exposure. These safety findings support the feasibility of short‑term, low‑dose BPC‑157 protocols for pre‑clinical tendon regeneration research.
For a full methodological description and statistical details, see the original publication: Tendon tissue-related research study.
Pre‑clinical Evidence – Gastrointestinal Repair
Study design
In a widely cited rodent model, male Sprague‑Dawley rats received an ethanol‑induced gastric ulcer and were treated via oral administration in research models with BPC‑157 at a dose of 10 µg/kg once daily for seven consecutive days. The control group received an equivalent volume of saline. Ulcer areas were measured end‑point day 7 using planimetry.
Key outcomes
The BPC‑157‑treated cohort exhibited a striking ≈70 % reduction in ulcer surface area compared with baseline, whereas the saline‑treated controls showed only a 25 % reduction. This difference was statistically significant (p < 0.01), underscoring the peptide’s capacity to accelerate mucosal tissue-related research in an acute injury setting.
Mechanistic insights
Mechanistic analysis revealed up‑regulation of endothelial nitric oxide synthase (eNOS) in the gastric mucosa of BPC‑157‑treated rats. Enhanced NO production correlated with preserved mucosal blood flow and a ~ 2‑fold increase in epithelial cell migration rates, as demonstrated by BrdU incorporation assays. Together, these findings suggest that oral BPC‑157 research has investigated a NO‑mediated protective environment that facilitates rapid re‑epithelialization of damaged gastric tissue.
Safety profile
Throughout the 7‑day protocol, treated animals maintained stable body weight and displayed no alterations in serum alanine aminotransferase (ALT) or aspartate aminotransferase (AST) levels, indicating an absence of overt hepatic toxicity at the used dose.
These pre‑clinical results provide a robust proof‑of‑concept that oral BPC‑157 can safely enhance gastrointestinal repair through endothelial NO pathways and accelerated epithelial restitution. For a deeper dive into the experimental details, see the original publication Gastrointestinal repair study.
Human Case Reports – Context & Caution
Several anecdotal case series have circulated on forums and in niche sports‑peptide compound newsletters, describing athletes who claim accelerated muscle‑tendon recovery after self‑used off‑label BPC‑157. Reported benefits range from reduced soreness after intense research protocols to seemingly faster tissue-related research of minor ligament sprains. These narratives often highlight rapid return‑to‑play timelines that appear striking when compared with conventional rehabilitation protocols.
Methodological Limitations
While compelling on the surface, the evidence base is hampered by critical design flaws. Sample sizes rarely exceed a handful of individuals, and there are no randomized control groups to isolate the peptide’s effect from natural tissue-related research, placebo response, or concurrent therapies. Outcomes are predominantly self‑reported, lacking objective metrics such as imaging, functional testing, or blinded assessment. Consequently, the data cannot be extrapolated to broader populations or used to substantiate clinical efficacy.
Regulatory Status
The U.S. Food and peptide compound Administration has not approved BPC‑157 for any research-grade indication. It remains classified as an investigational compound, and its distribution for research use only is prohibited under current federal regulations. Any marketing or sales claims suggesting wellness support would constitute a violation of FDA policy.
YPB’s Position
YourPeptideBrand (YPB) adheres strictly to the Research Use Only (RUO) framework. Our products are manufactured, labeled, and shipped exclusively for laboratory investigation, not for ingestion, injection, or any form of human consumption. We provide clear guidance to our clients to ensure compliance with all applicable laws and ethical standards.
This product is supplied for research purposes only. It is not intended for research identification, research application, research focus, or supports healthy function.
Regulatory Landscape – FDA RUO Labeling Requirements
The FDA’s 2022 guidance “Labeling of Research Use Only (RUO) Products” (FDA/CMO‑2022‑0001) outlines a clear framework for labeling peptide products that are sold strictly for laboratory investigation. The document emphasizes that RUO items must be unmistakably identified as non‑human‑use, with no implication of potential wellness benefits. Compliance with these rules protects both manufacturers and research institutions from inadvertent regulatory violations.
Mandatory Label Statements
- For Research Use Only – Not for research use only – displayed prominently on the front label.
- Batch (lot) number – ensures traceability from production to end‑user.
- Expiration date – calculated based on stability data and storage conditions.
- Storage conditions – temperature, humidity, and light exposure requirements.
Prohibited Claims
The FDA strictly forbids any language that suggests a research-grade, assess, or disease‑prevention effect. Phrases such as “research has investigated tissue-related research,” “has been investigated for its effects on tendon injury,” or “research has examined effects on gut recovery” are disallowed on RUO labels, packaging, or marketing collateral. Even indirect implications, like “frequently researched for clinical studies on muscle regeneration,” must be avoided unless the study is explicitly approved under an IND.
Distribution Limits
RUO peptides may be sold only to qualified research institutions, licensed professionals, or entities that can demonstrate a legitimate research purpose. Anabolic research shipments to retail pharmacies, direct‑to‑consumer channels, or unverified distributors breach FDA policy.
Compliance Checklist
- Clear “Research Use Only” disclaimer.
- Complete traceability: batch number and expiration date.
- Safety Data Sheet (SDS) provided with each shipment.
- Detailed storage and handling instructions.
- Contact information for the manufacturer or label service.
How YPB Meets Every Requirement
YourPeptideBrand (YPB) streamlines compliance by offering on‑demand printing of FDA‑approved RUO labels. Each label includes a QR code that links instantly to the product’s SDS, satisfying the safety‑data requirement without extra paperwork. YPB’s integrated batch‑tracking system logs lot numbers and expiration dates, enabling effortless traceability for both the supplier and the research customer. Additionally, YPB verifies that every order is shipped only to vetted research institutions or licensed professionals, ensuring distribution stays within FDA limits.
By leveraging YPB’s turnkey label service, clinics and entrepreneurs can focus on scientific inquiry while remaining fully compliant with the FDA’s RUO labeling standards. FDA RUO Guidance
Ethical Marketing & Distribution Practices
Approved promotional channels
YourPeptideBrand (YPB) limits outreach to venues where the audience is already qualified to understand the Research‑Use‑Only (RUO) status of BPC‑157. Acceptable channels include:
- Professional conferences and scientific symposia where peer‑reviewed abstracts are presented.
- Live or on‑demand webinars hosted by accredited researchers or clinicians.
- B2B e‑commerce platforms that require verification of a medical or research license before granting access.
- Direct email campaigns sent only to verified practitioners, clinic owners, or institutional purchasing departments.
Forbidden tactics
Any marketing that suggests a potential wellness benefits or targets the general public is non‑compliant. The following practices must be avoided:
- Consumer‑facing advertisements on social media, search engines, or retail marketplaces.
- Research subject research documentation or case‑studies that claim “research focus,” “heal,” or “reverse” specific medical conditions.
- Pricing language that implies the product is a peptide compound (e.g., “discounted research application price”).
- Unsubstantiated health claims that are not supported by peer‑reviewed data.
Recommended tone
Content should emphasize the scientific context rather than promise outcomes. Use phrasing such as:
- “Explores mechanisms of tissue regeneration” instead of “has been investigated for its effects on muscle loss.”
- “Has been examined in studies regarding research into angiogenesis and fibroblast migration” rather than “has been studied for effects on torn tendons.”
- “Provides a high‑purity RUO peptide for laboratory investigation” instead of “offers a research-grade solution.”
Sample compliant product page excerpt
Product Overview: BPC‑157 (10 mg) is a research‑grade peptide synthesized to >98 % purity. It is supplied for in‑vitro and in‑vivo studies exploring angiogenic pathways, fibroblast activity, and nitric‑oxide signaling.
Scientific Summary: Peer‑reviewed rodent models demonstrate enhanced micro‑vascular formation and accelerated tendon repair when BPC‑157 is used at 10 µg/kg daily. Human case reports remain anecdotal and are not intended to establish clinical efficacy.
Disclaimer: This product is labeled “Research Use Only” and is not intended for research use only, research identification, or research application. All research applications must comply with local regulations and maintain appropriate Institutional Review Board (IRB) approvals.
Request a Sample: Qualified researchers can request a complimentary vial by completing the short form below. Your credentials will be verified before shipment.
Tips for maintaining audit trails
Robust documentation protects both YPB and its partners during regulatory reviews:
- Capture explicit email opt‑ins with timestamps and a clear description of the information being shared.
- Store a copy of each recipient’s professional license, NPI number, or institutional affiliation.
- Log all outbound communications in a CRM that records date, channel, and content version.
- Retain copies of webinar registrations, conference exhibitor lists, and B2B platform approvals for at least three years.
Conclusion – Scientific Summary and Business Call to Action
Pre‑clinical investigations consistently demonstrate that BPC‑157 activates several regenerative pathways. The peptide’s ability to modulate angiogenesis, fibroblast migration, and nitric‑oxide signaling underpins its research-grade promise.
- VEGF‑mediated angiogenesis: Rodent models showed a 45 % increase in capillary density within 7 days of a 10 µg/kg dose.
- Fibroblast migration: In a tendon‑injury study, BPC‑157 accelerated fibroblast infiltration by 2.3‑fold compared with controls.
- NO signaling: Nitric‑oxide levels rose 60 % in gastro‑intestinal ulcer models, correlating with faster mucosal closure.
- Functional recovery: Treated rats regained 78 % of baseline grip strength within 14 days, versus 42 % in untreated groups.
- Safety profile: No adverse histopathological changes were observed at doses up to 100 µg/kg across multiple organ systems.
It is essential to reiterate that all data cited are from pre‑clinical studies; no human research-grade claims are permitted under FDA regulations.
YourPeptideBrand (YPB) offers a fully compliant, white‑label solution for clinicians and entrepreneurs. Our service includes on‑demand label printing, custom packaging, direct dropshipping, and expert regulatory guidance—all with zero minimum order quantities.
Ready to launch your own Research Use Only peptide line? Schedule a consultation, request a sample labeling kit, or contact our sales team today to discuss how YPB can accelerate your brand’s growth.
Explore our resources for deeper insights into compliant peptide commercialization and discover how YPB can support your business vision.