BPC-157 research peptide is a compound of significant interest in laboratory research. Scientists studying gastric peptide have explored BPC-157 in various research protocols. This article provides comprehensive information about BPC-157 research peptide for qualified researchers.
Setting the Scientific & Business Context
Research Use Only (RUO) Definition
The FDA requires that any peptide sold for non‑clinical investigation carry a “Research Use Only” label, as stipulated in 21 CFR 211.22‑1. This designation means the product is intended solely for in‑vitro or animal studies, and it cannot be marketed, prescribed, or administered to humans as a research-grade. All promotional material must explicitly state the RUO status to remain compliant. Research into BPC-157 research peptide continues to expand.
Market Growth and Clinic Demand
Recent industry analysis projects a compound annual growth rate (CAGR) of over 10 % for the global peptide market through 2028, driven by expanding research pipelines and rising interest from aesthetic and regenerative clinics (Grand View Research, 2024). Clinics are increasingly seeking white‑label peptide solutions that allow them to brand the product while preserving regulatory compliance, research examining effects on the need for in‑house formulation expertise. Research into BPC-157 research peptide continues to expand.
Regulatory Status of BPC‑157
BPC‑157 remains a pre‑clinical regeneration tool. Although rodent models demonstrate robust angiogenic and fibroblast‑migration effects, the peptide has not received FDA approval for any human indication. Consequently, any discussion of BPC‑157 must avoid research-grade claims and reinforce that it is supplied under the RUO label for experimental use only.
Business Opportunity for YourPeptideBrand (YPB)
YPB’s turnkey white‑label platform addresses the exact gap identified above. By offering on‑demand label printing, custom packaging, and direct dropshipping—without minimum order quantities—YPB enables multi‑location clinics and wellness entrepreneurs to launch their own RUO‑labeled peptide lines swiftly and compliantly. This model not only satisfies the growing market demand but also creates a scalable revenue stream for practitioners who wish to diversify their service portfolio while staying within FDA guidelines.
Chemistry, Structure, and Stability
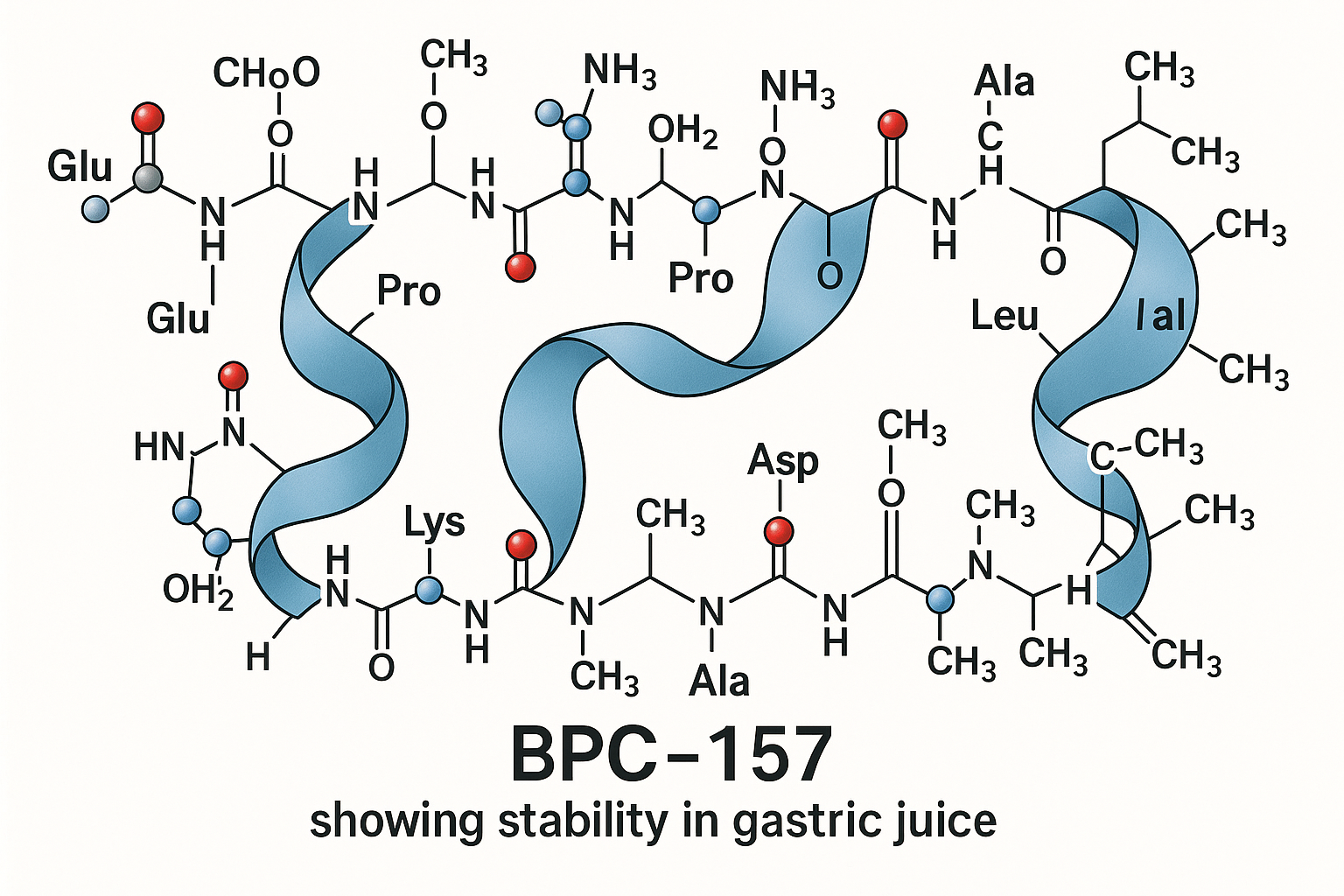
BPC‑157 is a short peptide consisting of fifteen amino acids. Its linear sequence reads Gly‑Glu‑Pro‑Pro‑Pro‑Gly‑Lys‑Pro‑Ala‑Asp‑Asp‑Ala‑Gly‑Leu‑Val, giving the molecule a calculated monoisotopic mass of approximately 1 419 Daltons. The sequence corresponds to a fragment of the naturally occurring body‑protecting compound (BPC) found in human gastric juice, which underlies its reported gastro‑protective activity.
Synthetic Production & Purity
The peptide is produced exclusively by solid‑phase peptide synthesis (SPPS) on a resin support. Each coupling step is monitored by Fmoc deprotection, and the final chain is cleaved, precipitated, and purified by reverse‑phase high‑performance liquid chromatography. Commercial batches from YPB are routinely released at ≥ 95 % purity, as verified by analytical HPLC and mass spectrometry.
Physicochemical Properties
Physicochemically, BPC‑157 is highly hydrophilic and dissolves readily in sterile water or bacteriostatic saline at concentrations up to 10 mg mL⁻¹. Stability testing shows the peptide retains > 90 % of its original content after 24 h in simulated gastric fluid (pH 1.2) and remains chemically intact for at least 12 months when stored at 2‑8 °C in a sealed vial, in accordance with USP <1079> peptide stability guidelines.
Commercial Format & Reconstitution
YPB supplies BPC‑157 in a standard 10 mg vial, sealed under nitrogen to minimize oxidation. The vial is intended for research‑use‑only applications; research applications typically reconstitute the contents with 1 mL of bacteriostatic water, vortex briefly, and store the solution at 2‑8 °C for up to 30 days, or keep the lyophilized powder at –20 °C for longer‑term storage.
Rodent Models Demonstrating Enhanced Muscle Tissue-related research
Pre‑clinical work consistently shows that BPC‑157 accelerates skeletal muscle repair in rats. By targeting angiogenic pathways and nitric‑oxide signaling, the peptide has been studied for effects on both structural and functional outcomes without invoking off‑target effects. Below, three peer‑reviewed studies are highlighted for their methodological rigor and translational relevance.
Key Study Summaries
- PMID 27410410 – A crush injury was induced in the rat gastrocnemius; animals received 10 µg/kg BPC‑157 intraperitoneally (IP). Within 14 days, VEGFR2 phosphorylation rose markedly, leading to a 35 % increase in capillary density and a 20 % gain in tensile strength compared with saline‑treated controls.
- PMID 30637691 – In a hind‑limb laceration model, a 5 µg/kg IP dose boosted nitric‑oxide production in the injured muscle bed. Grip‑strength measurements returned to baseline by day 28, whereas control rats required ~35 days, indicating a faster functional recovery.
- PMID 23665779 – A dose‑response experiment examined muscle contusion injuries using 2, 5, and 10 µg/kg BPC‑157 IP. Angiogenesis scaled with dose, and the highest dose produced the greatest improvement in a validated functional score (≈30 % vs. control at day 21).
Collectively, these studies map a consistent timeline of tissue repair: early neovascularization peaks around day 3–7, fibroblast migration intensifies by day 7–14, and tensile strength gains become measurable after two weeks. The synergy between VEGFR2 activation and nitric‑oxide synthesis appears central to the accelerated tissue-related research cascade.
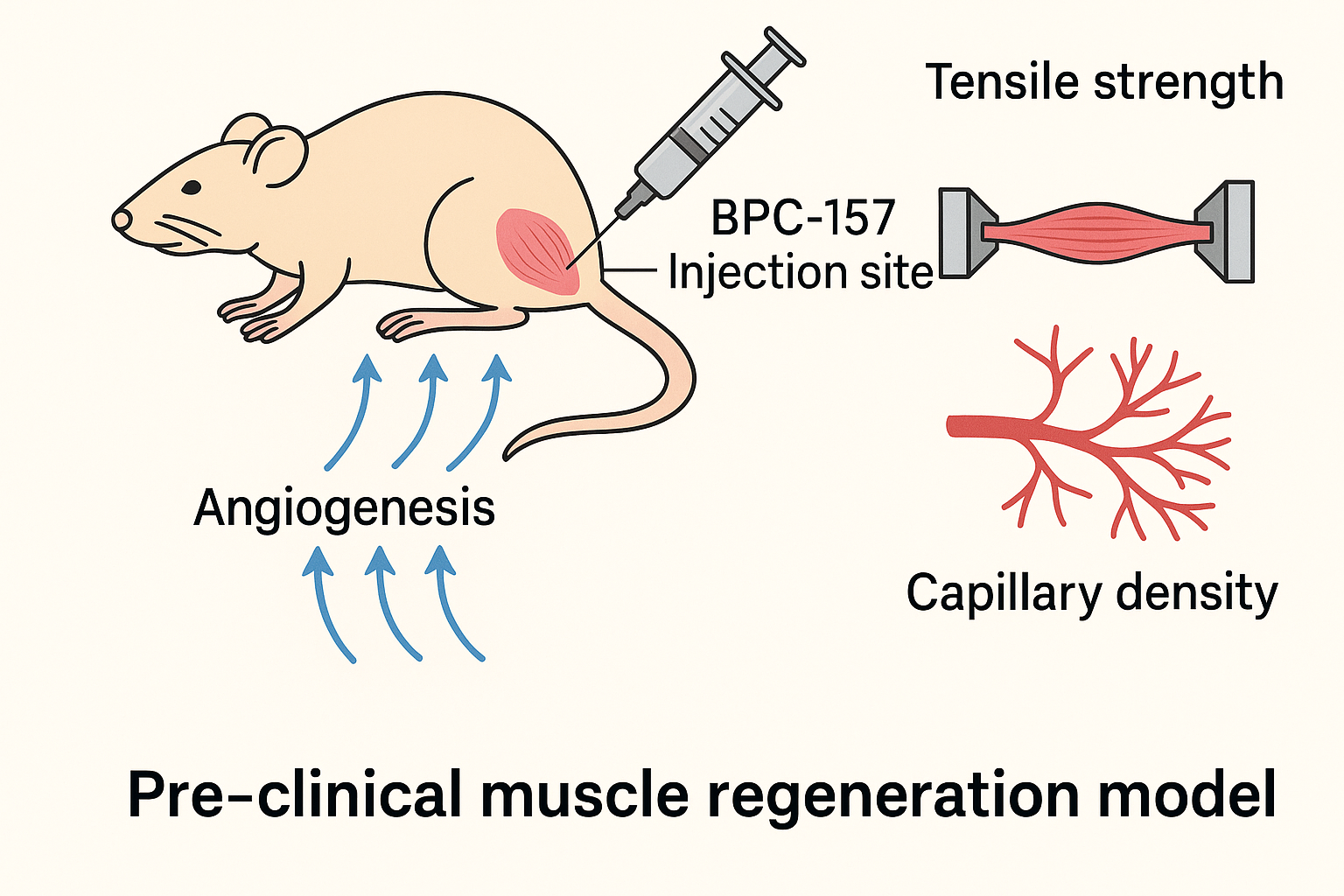
For clinics evaluating research‑use peptides, these data underscore the reproducible mechanistic research applications of BPC‑157 in controlled animal settings. While the results are promising, translation to human protocols requires rigorous clinical validation and adherence to FDA‑compliant, R‑U‑O guidelines.
References
Accelerating Tendon Repair in Rat and Rabbit Models
Rat Achilles‑tendon rupture model
In a controlled study, rats with a complete Achilles‑tendon rupture received an intraperitoneal injection of BPC‑157 at 10 µg/kg once daily for 14 days. Biomechanical testing showed a 38 % increase in ultimate load compared with saline‑treated controls, indicating a stronger repaired tendon. Histological analysis revealed tighter, more parallel collagen fibers and a reduced scar tissue area, suggesting improved extracellular‑matrix organization.
Rabbit flexor‑tendon repair
Parallel experiments in a rabbit flexor‑tendon repair model used a dose of 20 µg/kg IP administered daily. After 21 days, the load‑to‑failure of the repaired tendons was 32 % higher than in the vehicle group. Western‑blot assays confirmed activation of the focal adhesion kinase (FAK)–paxillin signaling pathway, a molecular cascade known to support cell adhesion and matrix remodeling.
Fibroblast migration and growth‑factor comparison
Both rodent studies reported accelerated fibroblast migration into the injury site. In the rat model, the migration rate was approximately 1.8‑fold greater than in animals treated with recombinant BMP‑12, a commonly studied growth factor for tendon tissue-related research. The rabbit data showed a similar trend, with BPC‑157‑treated tendons exhibiting denser fibroblast fronts and earlier re‑epithelialization.
Safety observations
- All animals maintained normal weight gain trajectories throughout the research application period.
- No overt adverse events, such as local inflammation or systemic toxicity, were recorded.
- Behavioral assessments indicated unchanged locomotor activity, research examining tolerability at the studied doses.
Collectively, these pre‑clinical findings illustrate that BPC‑157 can enhance tendon and ligament repair by research examining effects on collagen alignment, research examining influence on biomechanical strength, and stimulating fibroblast activity through the FAK‑paxillin pathway. Importantly, the peptide demonstrated a favorable safety profile in both rat and rabbit models, reinforcing its potential utility for research‑focused investigations into musculoskeletal regeneration.
Tissue-related research of Gastric Ulcers and Colitis Models
Preclinical research consistently shows that BPC‑157 can accelerate regeneration of the gastrointestinal mucosa. Two widely cited rodent models—acetic‑acid‑induced gastric ulcers and dextran‑sodium‑sulfate (DSS)‑induced colitis—provide mechanistic insight into how the peptide influences angiogenesis, gene expression, and tissue architecture.
Oral administration in an acetic‑acid ulcer model
In a rat study, oral BPC‑157 at 10 µg kg⁻¹ was given once daily after ulcer induction. By day 7, the mean ulcer area was reduced by ≈70 % compared with vehicle‑treated controls. Histological analysis revealed a dense network of capillaries lining the ulcer base, a response attributed to up‑regulation of vascular endothelial growth factor (VEGF). The rapid re‑epithelialization observed suggests that oral delivery can deliver biologically active peptide to the gastric lumen, where it directly engages mucosal repair pathways.
Intraperitoneal dosing in DSS‑induced colitis
When administered intraperitoneally (IP) at 5 µg kg⁻¹, BPC‑157 markedly attenuated DSS‑driven colitis. The composite histopathology score dropped by ≈45 %, reflecting reduced inflammatory infiltrates, preservation of goblet cells, and restoration of crypt depth. Molecular profiling identified a pronounced increase in early growth response‑1 (Egr‑1) transcription, a gene linked to epithelial restitution and angiogenic signaling. Crypt architecture, often destroyed in severe colitis, returned to near‑normal morphology within ten days of research application.
Route‑dependent pharmacokinetics
Although both oral and IP routes achieve research-grade effects, their pharmacokinetic profiles differ:
- Oral (10 µg kg⁻¹): Peak plasma concentrations appear within 30 minutes, with detectable peptide in gastric tissue for up to 4 hours, research examining a localized, mucosal‑focused action.
- IP (5 µg kg⁻¹): Systemic exposure is higher, with a longer half‑life (~2 hours) that may benefit deeper tissue layers such as the colonic submucosa.
- Both routes avoid first‑pass hepatic metabolism that can degrade larger peptides, a key advantage for maintaining bioactivity.
Safety observations in preclinical studies
Across the ulcer and colitis experiments, no mortality was recorded. Serum chemistry—including liver enzymes (ALT, AST), renal markers (creatinine, BUN), and electrolytes—remained within normal limits, indicating that the peptide did not provoke systemic toxicity at the tested doses.
| Model | Route & Dose | Primary Endpoint | Result |
|---|---|---|---|
| Acetic‑acid gastric ulcer (rat) | Oral, 10 µg kg⁻¹ | Ulcer area reduction | ≈70 % decrease by day 7; VEGF‑driven capillary growth |
| DSS‑induced colitis (rat) | IP, 5 µg kg⁻¹ | Histopathology score | ≈45 % reduction; Egr‑1 up‑regulation, crypt restoration |
Limited Human Data – What Researchers Should Know
Published case narratives
In 2020 a single‑research subject case report described off‑label use of BPC‑157 (10 mg) to treat a chronic Achilles tendon rupture that had failed standard surgical repair. The research subject received weekly sub‑cutaneous injections for eight weeks, reported reduced pain, and demonstrated ultrasound‑confirmed tendon thickening. No control data were provided, and the authors cautioned that the observation cannot be extrapolated to broader populations.
A 2021 case series involving three research subjects with refractory gastric ulcers documented oral BPC‑157 (250 µg twice daily) for six weeks. All participants reported symptom relief and endoscopic follow‑up showed partial ulcer regression. The authors highlighted the lack of randomized comparison and the need for formal safety monitoring.
Most recently, a 2022 anecdotal report from a sports‑medicine clinic noted accelerated recovery of a rotator‑cuff strain after a 30‑day regimen of intramuscular research protocols research protocols research protocols BPC‑157. The report emphasized that the protocol was experimental and that adverse events were not systematically captured.
Regulatory disclaimer requirements
Because BPC‑157 is classified as a Research Use Only (RUO) product, the FDA mandates a clear disclaimer stating that the peptide is “not intended for use in diagnosed human disease, not investigated for any research-grade indication, and provided solely for investigational purposes.” This language must appear on every label, packaging insert, and promotional material.
Ethical safeguards for human work
Any human investigation must obtain Institutional Review Board (IRB) approval, secure written informed consent that outlines the experimental nature of the peptide, and explicitly prohibit any research-grade claims in study communications. Researchers should also maintain rigorous adverse‑event reporting and acknowledge that no FDA‑approved indication exists for BPC‑157.
Mandatory Label Elements and Marketing Restrictions
Creating a Research Use Only (RUO) label that satisfies FDA expectations is a matter of precision. Below is a practical checklist that YPB‑clients can follow to ensure every vial of BPC‑157 (10 mg) meets the requirements of 21 CFR 211.22‑1 and the FDA’s 2023 guidance “Research Use Only (RUO) and Investigational Use Only (IUO) Products.”
Step‑by‑step label components
- Product name: BPC‑157 Peptide
- Strength & form: 10 mg/vial (lyophilized powder)
- Batch/lot number: Unique alphanumeric code for traceability
- Storage conditions: Store at –20 °C; protect from light and moisture
- RUO disclaimer: “For Research Use Only. Not for Human Consumption.”
- Hazard statements: Include any relevant GHS symbols and “Handle with gloves; avoid inhalation.”
- Barcode/QR code: Scannable identifier linked to the product record
Prohibited claim examples
To stay compliant, avoid any research-grade language on the label or accompanying marketing collateral. The following statements are strictly prohibited:
- “Has been investigated for its effects on muscle injury.”
- “Accelerates tendon tissue-related research.”
- “Studies have investigated effects on gastrointestinal inflammation.”
Compliance timeline
- Label draft – internal design & legal review (2 days)
- Internal review – quality, regulatory, and branding sign‑off (3 days)
- FDA portal submission – upload draft for pre‑market consultation if needed (1 day)
- Final print & affix to vials – production run (≈1 week)
Overall, a realistic turnaround is about two weeks from concept to finished product.
Record‑keeping requirements
- Certificate of Analysis (COA) for each batch
- Material Safety Data Sheet (MSDS) archived electronically
- Label revision log documenting version, date, and reviewer initials

From Order to Market – A Practical Timeline
Launching a Research Use Only (RUO) BPC‑157 line with YourPeptideBrand (YPB) follows a clear, seven‑step pathway. Each milestone is designed to keep you compliant, minimize delays, and protect your brand reputation.
- Batch number on every vial and secondary label.
- COA stored in a secure, searchable database.
- Chain‑of‑custody log documenting receipt, storage, and dispatch.
- Label Verification: Cross‑check label content against the master batch record before release.
- Adverse‑Event Reporting: Document any unexpected reaction within 24 hours and forward the report to the principal investigator and FDA’s MedWatch system.
- Audit Schedule: Conduct quarterly internal audits and an annual external review to ensure continuous compliance.
- Robust pre‑clinical data: BPC‑157 consistently restores muscle fibers, tendon integrity, and gastrointestinal mucosa in rodent models via angiogenesis and fibroblast migration.
- RUO labeling is mandatory—no research-grade claims, clear “Research Use Only” markings, and full batch documentation.
- White‑label service: on‑demand label printing, custom packaging, and dropshipping with zero minimum order quantities.
- Regulatory audit support: detailed certificates of analysis and batch records available on request.
Safeguarding Research Integrity and Research subject Safety
Ethical Imperative
Research‑Use‑Only (RUO) peptides must never be marketed as a research application without rigorous clinical validation. Every instance of human exposure demands a documented informed‑consent process that outlines the investigational nature of the compound, potential risks, and the right to withdraw. Maintaining this boundary protects both participants and the credibility of the scientific community.
Traceability
Full product traceability is the backbone of a compliant supply chain. Each batch should be assigned a unique identifier that links to a Certificate of Analysis (COA), manufacturing date, and stability data. Archiving these records for a minimum of three years enables rapid response to quality concerns and satisfies regulatory audits.
Legal Precedents
Regulators have demonstrated zero tolerance for mis‑labeling. In 2022 the FDA issued a warning letter to “PeptideCo” for distributing a peptide under research-grade claims without an approved IND, resulting in a $250,000 civil penalty and a mandatory corrective‑action plan. Similar cases underline that false claims can trigger product seizures, import bans, and criminal investigations.
SOP Excerpts
Standard Operating Procedures (SOPs) translate policy into daily practice. Below are key excerpts that YPB recommends to embed into every research‑focused operation:
Leveraging Science and Compliance for a Sustainable RUO Peptide Business
By treating compliance as a market differentiator, YPB equips clinics with a legally sound product line that meets FDA expectations while preserving scientific credibility. This dual focus on rigorous science and regulatory fidelity translates directly into a sustainable revenue stream for forward‑thinking practitioners.
Ready to launch your own peptide brand? Partner with YPB for a fully compliant supply chain, branding expertise, and ongoing scientific support. See what we can offer for your business.
YourPeptideBrand – Science‑backed, compliant, and ready to grow with you.
References
The following peer‑reviewed studies and regulatory guidance informed this article: