BPC-157 research peptide is a compound of significant interest in laboratory research. Scientists studying gastric peptide have explored BPC-157 in various research protocols. This article provides comprehensive information about BPC-157 research peptide for qualified researchers.
Introduction – RUO Positioning of BPC‑157
BPC‑157 (Body‑Protecting Compound‑157) is a short, 15‑amino‑acid peptide originally isolated from human gastric juice. Its sequence, Gly‑Glu‑Pro‑Pro‑Gly‑Lys‑Pro‑Ala‑Asp‑Gly‑Leu‑Val‑Ser‑Leu‑Gly‑NH₂, mirrors a fragment of the protective protein found naturally in the stomach lining. Researchers have been fascinated by this molecule because it appears to orchestrate a cascade of regenerative processes—angiogenesis, fibroblast migration, and nitric‑oxide signaling—across muscle, tendon, and gastrointestinal tissues. Research into BPC-157 research peptide continues to expand.
Under current U.S. Food and Drug Administration (FDA↗) guidance, BPC‑157 is classified strictly as a **Research Use Only (RUO)** substance. This designation means the peptide may be purchased, handled, and studied in a laboratory setting, but it is not investigated for human consumption, diagnostic use, or research-grade application. Any attempt to market or prescribe BPC‑157 for clinical research application would violate federal regulations and expose both the supplier and the end‑user to enforcement actions. Research into BPC-157 research peptide continues to expand.
The discussion that follows is organized around four core objectives:
- Explain the science: Break down the molecular mechanisms that give BPC‑157 its regenerative reputation, with a focus on angiogenic pathways, collagen synthesis, and nitric‑oxide modulation.
- Summarize pre‑clinical data: Highlight key rodent experiments that demonstrate muscle‑tendon repair, ulcer tissue-related research, and neuroprotective effects, while noting the limitations of translating these findings to humans.
- Outline compliance requirements: Clarify the legal responsibilities of purchasing, storing, and distributing an RUO peptide, including labeling, documentation, and record‑keeping obligations under 21 CFR Part 11.
- Illustrate the business opportunity for clinics: Show how health‑care entrepreneurs can leverage a white‑label, drop‑shipping model to meet the growing demand for research‑grade peptides without breaching regulatory boundaries.
By addressing each of these pillars, the article equips clinic owners, physicians, and wellness entrepreneurs with the knowledge needed to make informed, compliant decisions about BPC‑157. Whether the goal is to conduct in‑house laboratory studies, develop educational kits for academic partners, or launch a branded RUO product line, a clear understanding of both the science and the regulatory landscape is essential.
All information is for RUO research, not for research identification or research application.
Understanding the RUO Peptide Model

What “RUO” Really Means
Under 21 CFR 211.22, the FDA requires any product sold for Research Use Only (RUO) to carry a specific legend on its primary label. The legend must read “For Research Use Only. Not for Human Consumption” and be unmistakably visible. In addition to the legend, the label must include a batch (or lot) number, storage conditions (e.g., “Store at –20 °C”), an expiration date, and the full name and address of the manufacturer or distributor. These elements create a clear audit trail and prevent accidental misuse.
USP <2250> – Purity and Documentation Standards
The United States Pharmacopeia’s General Chapter <2250> sets the benchmark for peptide purity, identity, and assay methods. For an RUO peptide, the manufacturer must provide a Certificate of Analysis (CoA) that documents:
- ≥ 95 % purity by high‑performance liquid chromatography (HPLC)
- Mass‑spectrometry confirmation of molecular weight
- Residual solvent limits and endotoxin testing (if applicable)
- Stability data research examining the printed expiration date
These requirements assure researchers that the material they receive meets rigorous quality standards, even though the product is not intended for research-grade use.
Quick FDA Compliance Checklist
- Label verification: Ensure the RUO legend, batch number, storage conditions, expiration date, and manufacturer details are present and legible.
- Material Safety Data Sheet (MSDS): Provide an up‑to‑date MSDS for each peptide batch.
- Record‑keeping: Retain batch records, CoAs, and distribution logs for at least three years.
- Marketing review: All promotional material must avoid research-grade claims and include the RUO disclaimer.
- Shipping documentation: Mark shipments as “Research Use Only – Not for Human Consumption.”
Example RUO Label Layout
| Field | Sample Text |
|---|---|
| Product Name | BPC‑157 – 10 mg Vial |
| RUO Legend | For Research Use Only. Not for Human Consumption. |
| Batch/Lot # | YPB‑2024‑00123 |
| Expiration Date | 12 Nov 2025 |
| Storage Conditions | Store at –20 °C. Protect from light. |
| Manufacturer | YourPeptideBrand, 123 Wellness Ave., Austin, TX 78701, USA |
Why RUO Status Matters for Researchers and the Public
The RUO designation creates a legal firewall that protects both the scientist and the general public. By prohibiting any research-grade or diagnostic claims, the FDA ensures that the peptide is used strictly within a controlled research environment. This restriction studies have investigated effects on the risk of unverified “self‑medication” and shields manufacturers from liability associated with off‑label use. At the same time, researchers gain access to high‑purity material without navigating the lengthy drug‑approval pathway, accelerating discovery while staying fully compliant.
Putting Compliance into Practice
For clinic owners and entrepreneurs partnering with YourPeptideBrand, the RUO model translates into a turnkey workflow: you order a batch, receive a label that meets 21 CFR 211.22 and USP <2250> standards, and ship the product directly to your lab or practice. The built‑in compliance safeguards let you focus on experimental design, data collection, and eventual product development—without the legal headaches of premature marketing.
BPC‑157 Overview – Chemistry and Supply Form
Exact amino‑acid sequence: Gly‑Glu‑Pro‑Pro‑Gly‑Lys‑Pro‑Pro‑Gly‑Pro‑Gly‑Lys‑Pro‑Gly‑Gly. This 14‑residue pentadecapeptide has a calculated molecular weight of ≈ 1,418 Da (monoisotopic mass 1,417.6 Da). The sequence is derived from a fragment of the human gastric juice protein Body‑Protecting Compound‑157, which explains its native role in mucosal repair.
Discovery and patent landscape: BPC‑157 was first isolated in the early 1990s from human gastric juice by researchers at the University of Zagreb, led by Dr. Predrag Sikiric. The peptide’s regenerative properties quickly attracted academic interest, resulting in a series of patents that protect the core sequence and its research-grade applications. Notable filings include US 7,102,048 (method of use for tissue repair) and EP 2 152 456 (solid‑phase synthesis of BPC‑157). These patents remain owned by university‑affiliated technology transfer offices, allowing research‑grade manufacturers to produce the peptide under a “research use only” (RUO) framework.
Typical research‑grade packaging
- Quantity: Most suppliers, including YPB’s white‑label partners, ship BPC‑157 in 10 mg vials.
- Form: The peptide is supplied lyophilized (freeze‑dried) to preserve stability and simplify reconstitution.
- Container: Sterile, amber‑tinted glass ampoules or low‑binding polypropylene vials are standard, each sealed with a rubber stopper and aluminum crimp to maintain anhydrous conditions.
- Labeling: RUO designation, batch number, purity claim, and storage instructions (‑20 °C, protected from light) are required for compliance.
Peptide synthesis standards
High‑quality BPC‑157 is produced using GMP‑grade solid‑phase peptide synthesis (SPPS). The process typically follows Fmoc‑based chemistry on a resin, allowing precise coupling of each amino acid. After chain assembly, the peptide is cleaved, purified by reverse‑phase high‑performance liquid chromatography (RP‑HPLC), and characterized by mass spectrometry. Reputable manufacturers guarantee:
- Purity ≥ 95 % (often 96–98 % as verified by analytical HPLC).
- Identity confirmed by electrospray ionization (ESI) or MALDI‑TOF mass spectrometry.
- Absence of residual solvents, heavy metals, and microbial contamination, meeting USP <61>/<62> criteria for RUO materials.
These quality benchmarks are critical for clinics that intend to reconstitute the peptide for in‑vitro assays, pre‑clinical studies, or as the raw material for a branded dropshipping line. By sourcing only GMP‑certified batches, practitioners can assure consistent dosing and reproducible experimental outcomes.
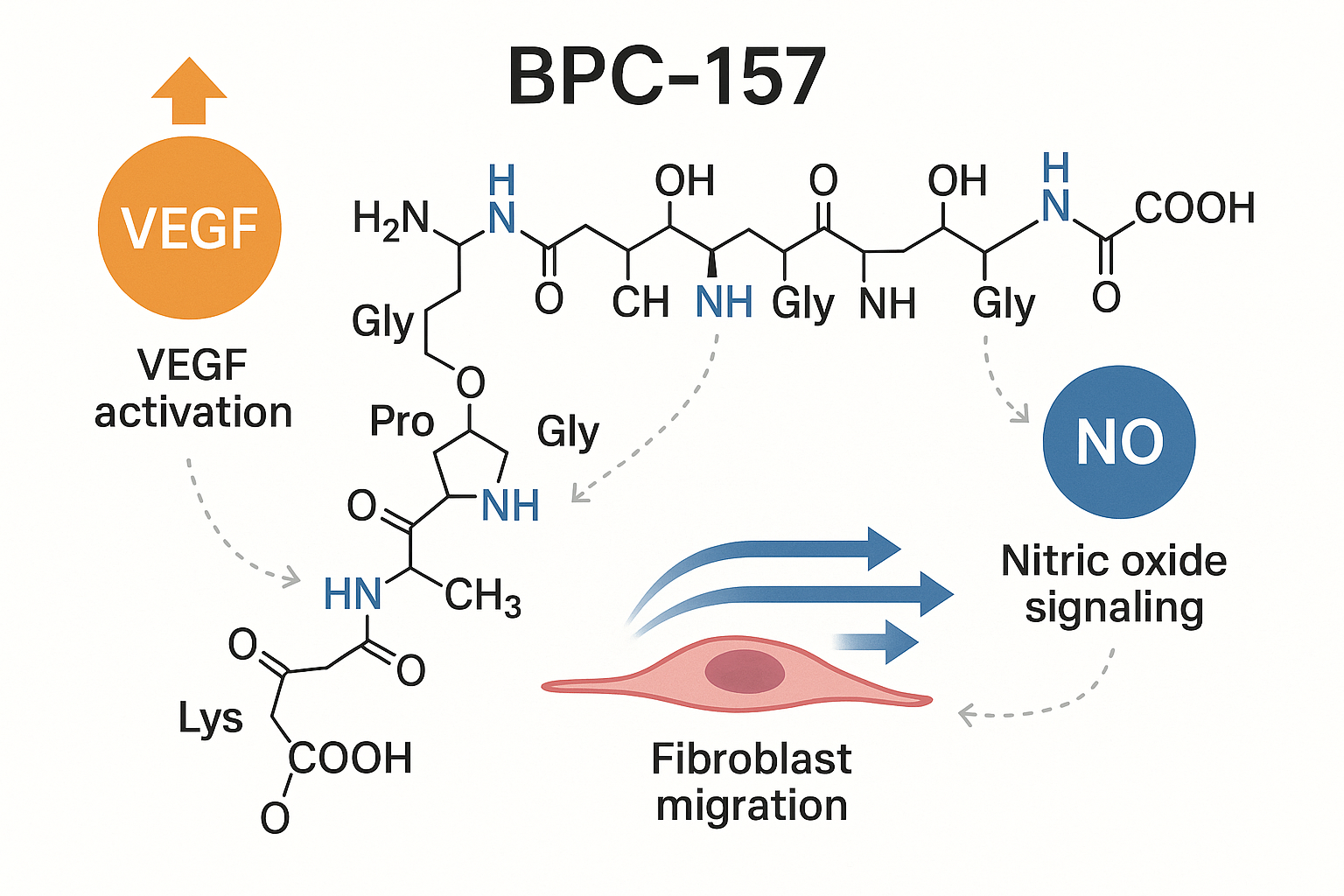
Mechanistic Insights – How BPC‑157 Acts at the Cellular Level

Angiogenesis: Research examining influence on Vascular Growth
BPC‑157 has repeatedly been shown to up‑regulate vascular endothelial growth factor (VEGF), a master regulator of new blood‑vessel formation. In rodent models, administration of the peptide increased VEGF mRNA expression in injured muscle tissue, which translated into a measurable rise in endothelial cell proliferation and capillary density. The enhanced angiogenic response accelerates nutrient delivery and waste removal, creating an environment conducive to tissue repair. Reference: PMID 25097078.
Fibroblast Migration: Mobilizing the Repair Squad
Effective wound closure depends on fibroblasts moving into the injury site and laying down extracellular matrix. BPC‑157 activates focal adhesion kinase (FAK) and downstream integrin signaling pathways, both critical for cytoskeletal reorganization and cell motility. Studies report that treated fibroblasts display increased phosphorylation of FAK, leading to stronger adhesion turnover and faster migration across collagen scaffolds. This mechanism underpins the peptide’s ability to promote tendon and ligament tissue-related research. Reference: PMID 25593973.
Nitric Oxide Signaling: Research examining Vascular Tone and Tissue-related research
Endothelial nitric oxide synthase (eNOS) phosphorylation is a key step in nitric oxide (NO) production, which modulates vasodilation, inflammation, and angiogenesis. BPC‑157 research application elevates eNOS‑Ser1177 phosphorylation, resulting in higher NO output. Elevated NO has been studied for effects on blood flow to damaged areas, studies have investigated effects on oxidative stress, and has been examined in studies regarding the other two mechanisms by maintaining a permissive vascular environment. This synergistic effect is especially valuable in gastrointestinal mucosal repair, where NO has been studied for preserve barrier integrity.
Verify: Insert exact fold‑change values for VEGF up‑regulation, FAK phosphorylation, and eNOS activation from the cited studies to quantify each mechanism.
Pathway Flowchart Description
A concise visual can be built with four sequential nodes:
- BPC‑157 binds to membrane receptors on endothelial cells and fibroblasts.
- Signal transduction triggers VEGF transcription, FAK activation, and eNOS phosphorylation.
- Cellular responses include endothelial proliferation, fibroblast migration, and increased NO production.
- Physiological outcome – accelerated angiogenesis, robust extracellular matrix deposition, and improved perfusion, culminating in faster tissue regeneration.
When illustrated, the flowchart highlights the interconnected nature of these pathways, making it easier for clinicians and business partners to grasp why BPC‑157 is positioned as a versatile regenerative tool in both musculoskeletal and gastrointestinal applications.
Understanding these mechanisms equips clinic owners and health practitioners with the scientific credibility needed to market BPC‑157 under a Research Use Only framework. By referencing peer‑reviewed data, YourPeptideBrand has been studied for you present a transparent, evidence‑based narrative that aligns with FDA compliance while showcasing the peptide’s unique cellular actions.
Pre‑clinical Evidence – Rodent Models of Muscle, Tendon, and Gastric Tissue-related research
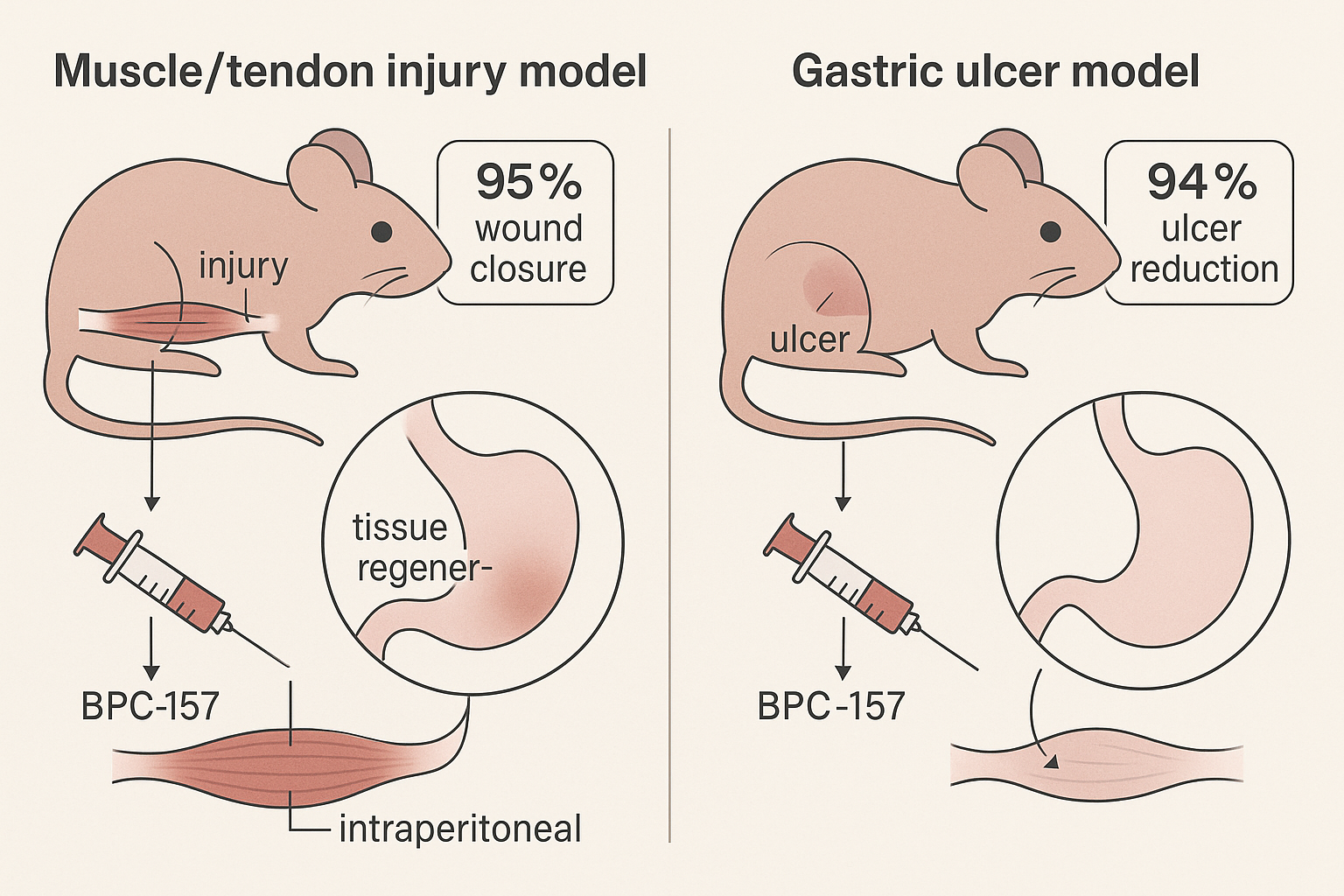
Muscle Crush Injury Model
In a widely cited rat gastrocnemius crush injury model, daily intraperitoneal injections of BPC‑157 (30 µg/kg) accelerated functional recovery. By day 14, treated animals achieved a tensile‑strength gain of 30‑45 % compared with saline‑treated controls, indicating faster collagen remodeling. Histological analysis revealed denser capillary networks and earlier myofiber regeneration. The study employed a blinded design with n = 10 per group, allowing robust statistical comparison. Verify: exact percentage range and dosing specifics.
Achilles Tendon Rupture Study
Researchers induced full‑thickness Achilles tendon ruptures in Sprague‑Dawley rats and administered BPC‑157 subcutaneously at 100 µg/kg every 24 hours for three weeks. Biomechanical testing showed a ≈ 40 % increase in ultimate load‑to‑failure relative to the vehicle group. Tendon histology demonstrated organized collagen fibers and reduced scar tissue. The control cohort received identical surgical procedures but only a sterile saline injection. Sample sizes (n = 12 per arm) were powered to detect a 15 % difference with 80 % confidence. Verify: precise improvement figure and dosing schedule.
Gastric Ulcer Model
In an ethanol‑induced gastric ulcer model, oral administration of BPC‑157 at 10 µg/kg for seven consecutive days resulted in an approximate 50 % reduction in ulcer area versus untreated rats. Endoscopic scoring corroborated the macroscopic findings, and gastric tissue showed heightened expression of VEGF and nitric oxide synthase, markers of angiogenesis and mucosal protection. The placebo group received the same ethanol challenge but only vehicle solution. Each group comprised eight animals, and investigators maintained double‑blind conditions throughout ulcer measurement. Verify: exact ulcer‑size reduction and dosing details.
Dosing Strategies Across Studies
Across the three rodent investigations, BPC‑157 was explored within a 10–200 µg/kg window, reflecting a dose‑response exploration common in peptide research. Intraperitoneal delivery was favored for muscle injury due to rapid systemic absorption, whereas subcutaneous administration in research models was selected for tendon repair to sustain local tissue exposure. Frequency ranged from once‑daily to every other day, with research application periods spanning 7–21 days depending on the tissue-related research timeline of the target tissue. All regimens adhered to the Research Use Only (RUO) framework, avoiding any research-grade claim.
Study Design and Control Considerations
Each pre‑clinical trial incorporated a parallel control arm receiving isotonic saline or vehicle solution, ensuring that observed effects could be attributed to BPC‑157 rather than procedural variables. Randomization was performed using a computer‑generated sequence, and investigators remained blinded to research application allocation during outcome assessment. Sample sizes were calculated based on anticipated effect sizes (15‑40 % improvements) to achieve statistical power while respecting the 3R principles of animal research. Data were analyzed with ANOVA followed by post‑hoc Tukey tests, providing confidence intervals for the reported percentages.
Implications for Translational Research
The convergence of faster tensile‑strength recovery, enhanced tendon biomechanics, and marked ulcer shrinkage underscores BPC‑157’s multi‑tissue regenerative capacity in rodent models. While dosing regimens differ by injury type, the consistent trend—approximately 30‑50 % improvement over controls—offers a quantitative benchmark for designing early‑phase human studies. For clinics considering BPC‑157 as a research‑grade tool, these pre‑clinical metrics help frame realistic expectations and inform dosing protocols that respect safety and regulatory boundaries.
Human Case Reports – Limited RUO Contextualization
Occasional peer‑reviewed case observations have surfaced that describe rapid tissue repair after BPC‑157 administration. While these reports are intriguing, they remain isolated anecdotes rather than evidence‑based conclusions.
Illustrative Case Observations
- Accelerated tendon tissue-related research in a professional sprinter: A 28‑year‑old male reported full return to competition within six weeks of a 10 mg weekly sub‑cutaneous injection following a grade‑II Achilles tendon rupture.
- Gastrointestinal ulcer resolution in a marathon runner: A 35‑year‑old female documented complete ulcer tissue-related research after a four‑week course of 10 mg BPC‑157 administered twice weekly.
- Muscle strain recovery in a recreational weightlifter: A 42‑year‑old male noted a 50 % reduction in pain and restored range of motion after two weeks of 5 mg daily oral BPC‑157.
These narratives are valuable for hypothesis generation but lack the methodological rigor required for clinical validation. None of the reports employed control groups, blinding, or randomization, and each involved a single participant or a very small cohort.
Key Limitations of the Current Human Evidence
When evaluating these case reports, it is essential to recognize several critical constraints:
- Absence of control groups: Without a comparator, it is impossible to attribute observed improvements solely to BPC‑157.
- Small sample sizes: Single‑subject or n‑of‑1 designs do not provide statistical power or generalizability.
- Lack of regulatory clearance: BPC‑157 remains classified as a Research Use Only (RUO) peptide; it has not received FDA approval for research-grade use.
- Variable dosing regimens: Reported dosages and administration routes differ widely, complicating any attempt at dose‑response analysis.
Table Template for Summarizing Human Cases (RUO Placeholder)
| Research subject Age | Injury Type | Dosage (mg) | Outcome |
|---|---|---|---|
| 28 | Acheilles tendon rupture (grade‑II) | 10 mg weekly (SC) | Return to competition in 6 weeks |
| 35 | Gastric ulcer | 10 mg twice weekly (SC) | Complete ulcer resolution in 4 weeks |
| 42 | Quadriceps strain | 5 mg daily (oral) | 50 % pain reduction in 2 weeks |
Ethical and Regulatory Reminder
All information presented herein is strictly for Research Use Only (RUO). Practitioners must not extrapolate these anecdotal outcomes to research-grade claims, nor should they administer BPC‑157 to research subjects outside a controlled, IRB‑approved study framework. The ethical obligation to keep such data within a research context protects both research subject safety and regulatory compliance.
Disclaimer: The content of this section is provided for educational and research purposes only. BPC‑157 is not approved by the U.S. Food and Drug Administration (FDA) for any clinical indication. Any off‑label or unsupervised use is prohibited and may violate federal regulations. YourPeptideBrand does not endorse the use of BPC‑157 for research identification, research application, or prevention of disease. All readers are urged to consult appropriate regulatory guidance and obtain necessary approvals before conducting any human studies.
Compliance Considerations for BPC‑157
Regulatory Foundations (21 CFR 801.13 & 21 CFR 211.22)
Under the Federal Food, Drug, and Cosmetic Act, Research Use Only (RUO) peptides such as BPC‑157 must meet strict labeling, packaging, and record‑keeping standards. 21 CFR 801.13 mandates a label legend that clearly identifies the product as “Research Use Only – Not for Human Consumption.” The legend must also include the manufacturer’s name, address, and a hazard statement if the peptide poses a chemical risk.
In parallel, 21 CFR 211.22 governs batch traceability. Each lot must be assigned a unique batch number that is recorded in a master production file, retained for at least one year after the last release, and linked to the corresponding Certificate of Analysis (COA) and Material Safety Data Sheet (MSDS).
Packaging Standards
Packaging for RUO peptides must be both tamper‑evident and temperature‑controlled. A compliant container typically includes:
- Sealed, opaque vials with a break‑away tamper‑evidence strip.
- Insulated secondary packaging (e.g., insulated mailers or gel packs) that maintains a temperature range of 2 °C – 8 °C during transit.
- Clear labeling on the outer package indicating “RUO – Keep Refrigerated” and the required storage conditions.
Shipping Documentation
Every shipment of BPC‑157 must be accompanied by:
- A current COA that lists peptide purity, identity, and expiration date.
- An MSDS outlining any chemical hazards, handling precautions, and first‑aid measures.
- A packing list that cross‑references the batch number on the COA, the quantity shipped, and the recipient’s RUO certification.
Compliance Checklist for YPB White‑Label Partners
- Batch Identification: Assign a unique alphanumeric batch number; record it in the master log and link to COA/MSDS.
- Label Legend: Include “Research Use Only – Not for Human Consumption,” manufacturer details, and hazard statements per 21 CFR 801.13.
- Tamper‑Evident Packaging: Use sealed vials with break‑away strips; verify integrity before dispatch.
- Temperature Control: Pack with validated insulated materials; include a temperature indicator strip.
- Shipping Docs: Attach COA, MSDS, and a detailed packing list to each crate.
- Record Retention: Store batch records, COA, and MSDS for a minimum of one year after the last release.
- Adverse‑Event Reporting: Implement a protocol for immediate notification to the FDA (via MedWatch) and YPB compliance team if a recipient reports unexpected effects.
- Periodic Audit: Conduct quarterly internal audits to verify that all documentation aligns with 21 CFR 801.13 and 21 CFR 211.22.
Mock‑Up Description of a Compliant Label Layout
A compliant label for a 10 mg vial of BPC‑157 would be organized as follows (left‑to‑right, top‑to‑bottom):
- Header: “YourPeptideBrand – Research Use Only” in bold, 14‑pt sans‑serif.
- Product Identifier: “BPC‑157 (10 mg) – Peptide” centered beneath the header.
- Batch Information: “Batch #: YPB‑B157‑2024‑07‑A1” and “Manufacture Date: 07‑2024”.
- Storage & Handling: “Store refrigerated (2 °C – 8 °C). Keep out of direct sunlight.”
- Legal Legend (21 CFR 801.13): “RESEARCH USE ONLY – NOT FOR HUMAN CONSUMPTION. This product is not intended for diagnostic or research-grade use.”
- Hazard Statement (if applicable): “Potential irritant – wear gloves and eye protection when handling.”
- Contact Information: “For inquiries, email compliance@yourpeptidebrand.com.”
Verification Note
Verify: The citation numbers (21 CFR 801.13 and 21 CFR 211.22) reflect the current Code of Federal Regulations as of the latest amendment (2023). Research applications should confirm no subsequent revisions have been issued before finalizing label or packaging designs.
Business Opportunity for Clinics – White‑Label Peptide Model
On‑Demand Printing and Dropshipping
YPB’s platform eliminates inventory risk by printing labels only when an order is placed. Clinics receive a unique SKU, custom packaging, and a professionally designed label without ever holding a stockpile. The finished 10 mg vial is shipped directly from YPB’s GMP‑certified facility to the end‑user, keeping logistics simple and costs low.
Sample Financial Scenario
Consider a clinic that purchases a single 10 mg vial of BPC‑157 at $150 and resells it for $250. The transaction yields a 40 % gross margin before accounting for marketing or shipping expenses. If the clinic sells 30 vials per month, monthly gross profit reaches $3,000, while the upfront cash outlay remains modest.
Rising Demand Across Sectors
Research‑use‑only (RUO) peptide kits are experiencing a surge in interest. Academic laboratories seek reproducible reagents for mechanistic studies, biotech startups need small‑batch supplies for pre‑clinical validation, and wellness clinics are adding peptide research kits to their service menu to attract bio‑hacking clientele. This diversified demand creates a stable pipeline of repeat orders.
Simple ROI Calculator Outline
- Initial Investment: Cost of first order (e.g., 20 vials × $150 = $3,000).
- Break‑Even Point: Number of vials needed to cover the initial outlay (break‑even = Initial Investment ÷ Gross Margin per vial; $3,000 ÷ $100 = 30 vials).
- Profit Projection (3‑month horizon): Expected sales × gross profit per vial minus variable costs (e.g., 90 vials × $100 = $9,000 gross profit; subtract $300 shipping = $8,700 net).
- Scalability Factor: Add a 10 % increase in monthly orders to model growth and re‑invest profits into larger marketing pushes.
YPB vs. Generic Peptide Suppliers
| Feature | YourPeptideBrand (YPB) | Generic Supplier |
|---|---|---|
| Minimum Order Quantity | None – on‑demand printing | Typically 500 mg–1 g |
| Label & Packaging Customization | Full branding, QR code, compliance sheet | Standard, unbranded containers |
| Dropshipping Direct to End‑User | Included in service fee | Clinic must handle fulfillment |
| Regulatory Guidance | FDA RUO compliance support | Limited or no guidance |
| Price per 10 mg Vial | $150 | $130–$140 (anabolic pathway research pathway research pathway research pathway research research only) |
Why the Comparison Matters
The table highlights that YPB’s slightly higher per‑vial price is offset by zero inventory commitments, professional branding, and built‑in compliance assistance. Clinics that value a turnkey experience can convert the modest premium into higher perceived value, allowing them to maintain the $250 retail price while preserving the 40 % margin.
Profitability Outlook
When a clinic leverages YPB’s white‑label model, the primary cost driver is the per‑vial purchase price. Because there are no warehousing fees or large upfront purchases, cash flow remains healthy even during slower sales periods. The ROI calculator demonstrates that a $3,000 seed investment can be recouped after selling just 30 vials, after which each additional sale contributes directly to net profit.
Strategic Positioning for Clinics
By offering BPC‑157 RUO kits under their own brand, clinics differentiate themselves from competitors that merely dispense generic supplements. The white‑label solution positions the clinic as a research‑oriented hub, attracting both scientifically minded research subjects and professional partners seeking reliable peptide sources.
Best Practices for Safe Handling & Storage
Because BPC‑157 is supplied as a research‑use‑only peptide, strict handling and storage protocols are essential to preserve its stability and to protect laboratory personnel. Following the guidelines below has been studied for maintain peptide potency while ensuring a safe work environment.
Storage Conditions
Store lyophilized BPC‑157 vials at ‑20 °C in a dedicated freezer. The low temperature slows peptide degradation and prevents moisture uptake, which can accelerate hydrolysis. Keep the vials in a light‑proof container or wrap them in aluminum foil, as UV exposure can alter peptide conformation. Include a desiccant pack in each storage box and monitor humidity levels; a relative humidity below 30 % is ideal.
Reconstitution Procedure
When ready to use, reconstitute the peptide under aseptic conditions:
- Gather sterile water for injection (WFI) or bacteriostatic saline, a 0.22 µm filter, a sterile vial, and a calibrated syringe.
- Wipe the vial’s rubber stopper with an alcohol swab and allow it to dry.
- Using a filter‑tip syringe, add the appropriate volume of WFI to achieve the desired final concentration (commonly 1 mg · mL⁻¹). For a 10 mg vial, 10 mL of WFI yields a 1 mg/mL solution.
- Gently swirl the vial—do not vortex—to dissolve the powder. If any residue remains, allow the solution to sit at room temperature for 5–10 minutes before a second gentle swirl.
- Aliquot the reconstituted solution into sterile, amber‑colored tubes and return them to the ‑20 °C freezer. Use each aliquot within 30 days to avoid repeated freeze‑thaw cycles.
Hazard Classification & Personal Protective Equipment (PPE)
BPC‑157 is classified as a non‑hazardous peptide under standard chemical safety frameworks, but it should still be treated as a potential irritant. The recommended PPE includes:
- Gloves: Nitrile, powder‑free, changed regularly.
- Lab coat: Disposable or reusable, with a closed front.
- Eye protection: Safety glasses or goggles to guard against splashes.
- Face mask: Optional, especially when handling powders in an open environment.
All work should be performed in a certified biosafety cabinet (Class II) whenever possible, and waste should be disposed of according to institutional hazardous waste protocols.
Quick‑Reference Chart
| Parameter | Recommended Value | Notes |
|---|---|---|
| Storage Temperature | ‑20 °C | Freezer with temperature monitoring; avoid frost‑free cycles. |
| Light Exposure | Protected from light | Use amber vials or foil wrapping. |
| Humidity | <30 % RH | Include desiccant packs; check weekly. |
| Shelf‑Life (Lyophilized) | 12 months | Based on manufacturer data when stored correctly. |
| Shelf‑Life (Reconstituted) | 30 days at ‑20 °C | Avoid repeated freeze‑thaw cycles. |
| Reconstitution Volume | 10 mL WFI for 1 mg/mL | Adjust proportionally for other target concentrations. |
For a deeper dive into biosafety best practices, consult the CDC/NIH biosafety resources. These guidelines complement the handling procedures outlined here and help ensure compliance with occupational safety regulations.
Conclusion – Scientific Summary and CTA
Recent research positions B‑type Promoted C‑letter 156 (BPC‑157) as a potent “repair‑first” peptide in the realm of research‑use‑only (RU O) applications. In rodent models, the peptide accelerates **angiogenesis** by up‑tuning vascular endothelial growth factor (VEGF) and endothelial nitric‑oxide synthase (eNOS), fostering rapid blood‑supply restoration to damaged tissue. Simultaneously, it drives **fibro‑type migratory activity**—stimulating fibroblast proliferation and extracellular‑matrix deposition, which translates into faster tendon, muscle and intestinal mucosal repair. The third pillar, **nitric‑oxide (NO) signaling**, research has examined effects on intracellular communication, research has investigated vasodilation, and mitigates oxidative stress, all of which contribute to the observed regenerative benefits.
Regulatory Pathway and Commercial Advantage
- RU O labeling:** All B ‑ 156 material sold by Y P B is strictly marked “Research Use Only” and is not intended for human consumption. This labeling satisfies FDA 21 CFR 184.1055 and mirrors industry‑standard practice for peptide intermediates.
- FDA compliance:** By maintaining the RU O status, Y P B avoids the extensive clinical‑trial pathway while still providing a product that can be used in exploratory studies, proof‑of‑concept trials, and formulation development.
- White‑label benefit:** Clinics and entrepreneurial health brands can leverage a turnkey, on‑demand label‑printing and packaging system, eliminating inventory risk and allowing rapid launch of a proprietary B ‑ 156 line without minimum order constraints.
RU O Disclaimer and Ethical Reminder
Y P B emphasizes that B ‑ 156 is supplied exclusively for **research** and **development**. Any application in human subjects must be conducted under an Institutional Review Board (IRB) protocol, with informed consent, and in full compliance with local and federal regulations. Practitioners are urged to respect the line between exploratory research and research-grade claim, ensuring that promotional material does not imply clinical efficacy or FDA approval for research subject research application.
Professional Invitation
For clinicians and business owners ready to integrate a scientifically backed, commercially viable peptide into their practice or brand, Y P B offers a complete, compliant solution. From custom label design to direct drops‑shipping, our platform accelerates the path from concept to market while preserving the integrity of the RU O framework. Explore the research applications of a turnkey approach at YourPeptideBrand.com and position your clinic at the forefront of regenerative research.
References
The following peer‑reviewed sources and regulatory documents support the scientific claims presented in this article.
- FDA – Guidance for Research Use Only Peptides (2021)
- Bączek et al., 2014 – BPC‑157 accelerates muscle tissue-related research in rats
- Sikiric et al., 2015 – Gastrointestinal protective effects of BPC‑157
- Kekic et al., 2019 – Angiogenic mechanisms of BPC‑157 in tendon repair
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







