best practices maintaining peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines best practices maintaining peptide and its applications in research contexts.
Why Peptide Integrity Matters in Research
What is peptide integrity?
Peptide integrity refers to the ability of a peptide to retain its defined physicochemical and biological characteristics from synthesis through to the point of use. The most critical quality attributes are purity (the proportion of the target sequence relative to impurities), biological activity (the capacity to engage its intended target or elicit a measurable response), and solubility (the ability to remain dissolved under assay conditions). When these attributes are preserved, researchers can trust that observed effects stem from the peptide itself rather than from degradation products or contaminants. Research into best practices maintaining peptide continues to expand.
How instability undermines experimental outcomes
Peptides are inherently prone to several degradation pathways. Aggregation can mask active sites and reduce apparent potency; oxidation of methionine, cysteine, or tryptophan residues can alter conformation and receptor binding; and hydrolysis of peptide bonds, especially at labile termini, can truncate the sequence, rendering it biologically inactive. Even subtle changes can shift dose‑response curves, generate spurious signaling events, or produce inconsistent results across replicates. In practice, a single batch that has partially oxidized will often yield data that appear contradictory to a freshly prepared control, leading investigators to chase false leads or publish misleading conclusions. Research into best practices maintaining peptide continues to expand.
Regulatory expectations for research‑use‑only materials
Regulators such as the U.S. Food and Drug Administration (FDA↗) expect that any research‑use‑only (RUO) peptide employed in pre‑clinical studies be handled with documented rigor. The FDA’s guidance on RUO materials explicitly states that manufacturers and research applications must maintain traceability, demonstrate consistent quality, and mitigate risks associated with degradation [FDA guidance]. Failure to uphold these standards can result in non‑compliant data packages, jeopardizing downstream IND submissions or FDA inspections. Maintaining integrity, therefore, is not merely a best practice—it is a compliance requirement that protects both scientific credibility and regulatory standing.
Setting the stage: the three pillars of peptide stewardship
Understanding why peptide integrity matters prepares the ground for actionable solutions. The remainder of this guide is organized around three interconnected pillars: handling (how you thaw, aliquot, and work with peptides in the lab), storage (temperature, container choice, and atmosphere control), and documentation (record‑keeping, batch tracking, and deviation reporting). Mastering each pillar ensures that every peptide you purchase or produce remains fit for purpose, safeguarding experimental validity, cost efficiency, and regulatory compliance.
Handling Peptides to Preserve Stability
Peptides are inherently prone to adsorption, aggregation, and oxidation. Even brief mishandling can shift a batch from “research‑grade” to compromised, jeopardizing downstream assays and regulatory compliance. The following protocol translates best‑practice guidelines—mirrored in FDA recommendations for biologic handling—into everyday laboratory actions that protect peptide integrity without adding unnecessary complexity.

1. Choose Low‑Binding Consumables
Standard polypropylene tubes and tips can bind up to 30 % of a peptide’s mass, especially for hydrophobic sequences. Low‑binding microcentrifuge tubes (often marketed as “siliconized” or “polypropylene‑treated”) and low‑retention pipette tips dramatically reduce surface adsorption. This simple switch preserves both concentration and activity, ensuring that the amount you dispense matches the amount you intended to deliver.
2. Maintain Cold Chain During Manipulation
Most peptides remain stable at 4 °C for short periods but begin to degrade rapidly at room temperature, particularly in the presence of light or moisture. Keep all working solutions on ice or a refrigerated block, and limit exposure to ambient conditions to no more than 15 minutes per handling step. For high‑throughput workflows, pre‑chill pipette tips, tube racks, and vortex adapters to maintain a consistently low temperature throughout the process.
3. Prevent Freeze‑Thaw Stress
Repeated freezing and thawing creates ice crystal formation that can denature peptide structures and promote aggregation. Aliquot anabolic pathway research pathway research pathway research research peptide stocks into single‑use volumes (typically 10–20 µL depending on downstream needs) before the initial freeze. Store each aliquot at –80 °C, and thaw only the exact volume required for the experiment. This practice eliminates the need for refreezing and preserves the peptide’s original conformation.
4. Use Protective PPE and Controlled Lighting
Gloves and a lab coat are mandatory not only for personal safety but also to prevent contaminant transfer. Many peptides contain oxidation‑sensitive residues (e.g., methionine, cysteine). Working in a low‑light environment—or using amber‑colored tubes—minimizes photo‑oxidation. If possible, perform manipulations under a nitrogen‑purged hood to further reduce exposure to atmospheric oxygen.
5. Apply Gentle Mixing Techniques
Vigorous vortexing can introduce shear forces that promote peptide aggregation, while prolonged sonication may generate localized heating. Use a brief, low‑speed vortex (≤ 500 rpm for 2–3 seconds) or a short pulse of a probe sonicator (≤ 10 seconds at low amplitude) only when complete dissolution is essential. Follow mixing with an immediate return to cold conditions to arrest any temperature‑induced degradation.
6. Quick‑Reference Daily Handling Checklist
- ✅ Verify that all tubes and tips are low‑binding.
- ✅ Keep reagents on ice; limit ambient exposure to ≤ 15 minutes.
- ✅ Use pre‑aliquoted, single‑use volumes; never refreeze thawed aliquots.
- ✅ Wear gloves and a lab coat; work under dim lighting or amber containers.
- ✅ Mix gently—short vortex or brief sonication only when necessary.
- ✅ Document any deviations from the protocol in the lab notebook.
This checklist, often reproduced as an infographic near the bench, serves as a visual cue that reinforces consistent handling habits across multiple research applications and shifts.
“For biologic products, including peptide therapeutics, manufacturers should implement handling procedures that minimize exposure to temperature fluctuations, light, and mechanical stress to preserve product potency and safety.” – FDA Guidance for Industry, 2023.
Optimizing Storage Conditions for Long‑Term Quality
Peptides are highly susceptible to temperature, light, and moisture. Even brief exposure to sub‑optimal conditions can trigger oxidation, aggregation, or hydrolysis, jeopardizing experimental reproducibility and regulatory compliance. For labs that maintain anabolic pathway research pathway research pathway research research peptide inventories—whether for internal research or white‑label distribution—establishing a clear storage hierarchy is essential.
Temperature hierarchy: matching stability to usage timeline
Align storage temperature with the intended shelf life. -80 °C cryogenic freezers are the gold standard for long‑term stocks lasting months to years. -20 °C freezers suit short‑term supplies accessed weekly to monthly, provided they stay within ±1 °C. For aliquots used within days, a dedicated 4 °C refrigerator is acceptable, but only when the peptide is protected from light and humidity.
Sealed, low‑oxygen containers to block light and moisture
Amber‑tinted vials or screw‑cap tubes with PTFE liners block ultraviolet photons and limit oxygen diffusion. For hygroscopic peptides, include a desiccant packet or purge the headspace with an inert gas (argon or nitrogen) before sealing, creating a dry micro‑environment that preserves the peptide’s anhydrous form.

Choosing the right freezer and monitoring temperature
Mechanical (compressor‑based) freezers are cost‑effective for -20 °C and -80 °C storage if they feature dual compressors that reduce temperature swings. Cryogenic units, though pricier, eliminate compressor‑induced spikes and are frequently researched for high‑value peptide libraries that cannot tolerate thaw‑refreeze cycles.
Continuous temperature logging is non‑negotiable. Data loggers with alarm thresholds should be calibrated quarterly and linked to your LIMS. Set alerts for deviations >2 °C (‑20 °C units) or >5 °C (‑80 °C units) to intervene before irreversible damage occurs.
Desiccants and inert gas blankets for hygroscopic peptides
Silica‑gel packets inside sealed vials keep relative humidity below 30 %, preventing hydrolysis. For especially moisture‑sensitive sequences, purge the vial with argon or nitrogen and seal with a 0.2 µm‑filtered port to maintain an oxygen‑free environment that also curbs oxidative side reactions.
Labeling conventions that prevent mix‑ups
Every vial should display batch number, peptide name, exact concentration, and an expiration date calculated from the first freeze. Adding a QR code linked to the LIMS entry enables rapid verification of purity certificates and storage history. Color‑coded stickers (blue for ‑80 °C, green for ‑20 °C, yellow for 4 °C) provide an immediate visual cue, research examining effects on retrieval errors.
Managing rapid temperature fluctuations
Frequent door openings, power outages, or faulty defrost cycles can cause sudden temperature swings, leading to precipitation and aggregation. Invest in alarm‑enabled storage units with backup power (UPS or generator) and “soft‑start” compressors that minimize rapid changes. Document any excursion in the SOP log; if a peptide experiences >5 °C above its set point for more than 30 minutes, re‑evaluate its integrity by HPLC or mass spectrometry before reuse.
Documentation and Tracking Systems for Peptide Management
Why a Digital Lab Notebook or LIMS Is Non‑Negotiable
In peptide research, a single vial can represent weeks of synthesis, costly raw material, and critical experimental data. When that vial is misplaced, mislabeled, or used past its shelf‑life, the downstream results become unreliable and regulatory audits turn into firefighting sessions. A digital laboratory notebook (ELN) or a full‑featured Laboratory Information Management System (LIMS) creates a single source of truth that links every physical sample to its electronic record. This traceability eliminates guesswork, studies have investigated effects on manual transcription errors, and provides instant visibility for every team member—from the bench scientist to the compliance officer.
Core Data Fields Every Entry Must Capture
Regardless of the software you choose, the system should enforce a standardized set of fields. Missing or inconsistent information is the fastest route to data drift. Below is a concise list of the minimum metadata required for each peptide batch:
| Field | Purpose |
|---|---|
| Peptide Name | Unique identifier used in protocols and publications |
| Source / Vendor | Enables verification of purity certificates and batch provenance |
| Lot Number | Critical for batch‑specific quality assessments and recalls |
| Concentration (mg/mL) | Ensures dosing calculations are reproducible |
| Storage Location | Freezer, fridge, or nitrogen tank coordinates for rapid retrieval |
| Aliquot Dates | Tracks when sub‑samples were prepared, research examining freeze‑thaw accounting |
| Freeze‑Thaw Count | Prevents over‑cycling, which accelerates peptide degradation |
| Expiration Date | Triggers alerts before the material is used out of spec |
Automated Expiration Alerts: Guarding Against Degradation
Modern LIMS platforms can calculate an expiration window based on the peptide’s declared stability and the number of freeze‑thaw cycles logged. When a vial approaches its “use‑by” date, the system pushes a notification to the responsible scientist’s dashboard, email, or mobile device. This proactive reminder stops the accidental use of compromised material, preserves experimental reproducibility, and protects the lab from costly repeat assays.
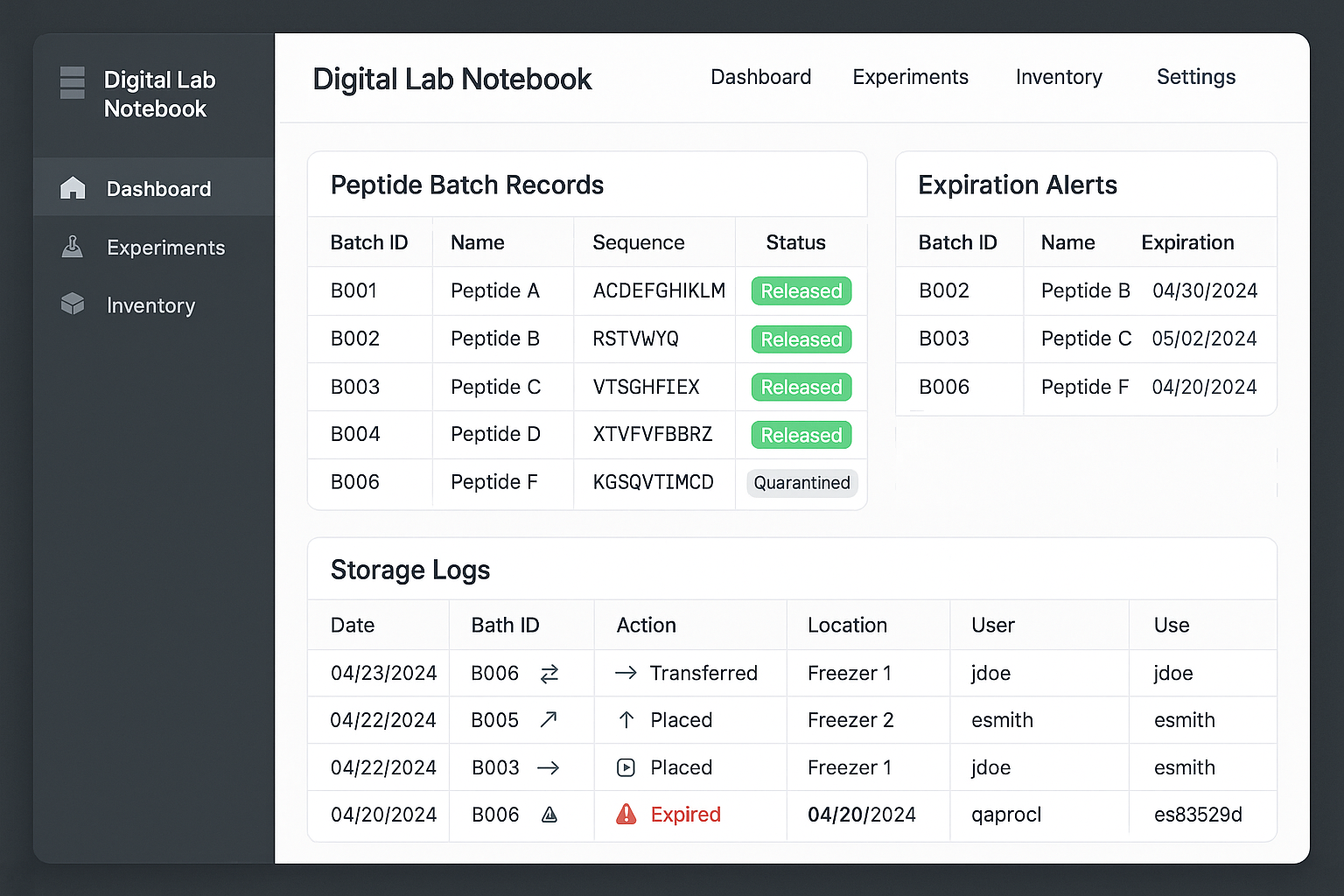
Walking Through a Mock LIMS Dashboard
The screenshot below illustrates a typical LIMS home screen. Notice the top‑right “Alerts” pane, which aggregates upcoming expirations, temperature excursions, and pending SOP acknowledgments. The central “Peptide Inventory” table can be filtered by storage location, lot number, or freeze‑thaw count, allowing a quick audit of any subset of samples. Clicking a row opens a detailed view where researchers may add aliquot events, upload analytical certificates, and attach the relevant SOP version.
Standard Operating Procedure (SOP) Templates
To keep documentation consistent, adopt ready‑made SOP templates that map directly onto your LIMS fields. A well‑structured SOP should include:
- Receiving and Verification: Log vendor certificates, confirm lot numbers, and assign a unique barcode.
- Aliquoting Procedure: Document date, volume removed, and update the freeze‑thaw counter.
- Storage Guidelines: Specify temperature ranges, recommended container types, and location labeling conventions.
- Disposal Protocol: Record date, method (e.g., incineration, chemical neutralization), and sign‑off by a compliance officer.
Embedding these SOPs in the LIMS as attachable PDFs ensures that every user can access the latest version with a single click, and the system can enforce acknowledgment before any critical action is performed.
Audit‑Ready Documentation for FDA Compliance
The FDA expects a complete, immutable trail from peptide acquisition to final experimental use. By capturing every transaction in a searchable, time‑stamped digital log, you satisfy 21 CFR 11 requirements for electronic records. During an inspection, auditors can generate a “batch history report” that lists every aliquot, temperature log, and SOP version associated with a specific lot. This transparency not only streamlines compliance but also builds confidence with partners who rely on your data integrity.
Cheat Sheet: Quick‑Reference Best‑Practice Steps
- Enter every new peptide into the LIMS within 15 minutes of receipt.
- Assign a barcode and scan it for every aliquot or movement.
- Record freeze‑thaw events immediately; do not estimate later.
- Set automated alerts for 30‑day, 7‑day, and 1‑day before expiration.
- Link the current SOP version to each record; require electronic acknowledgment.
- Generate a monthly “Inventory Health” report and review it with the compliance team.
- Archive all certificates of analysis and disposal records in the LIMS for the required retention period.
Ensuring Compliance and Growing Your Peptide Business
Recap of the Three Pillars
Successful peptide work rests on three non‑negotiable pillars: careful handling, controlled storage, and meticulous documentation. Each step— from thawing a vial on ice to recording batch numbers in a secure log— safeguards the molecule’s structural integrity. When these practices become routine, the risk of degradation, contamination, or data inconsistency drops dramatically.
Why Integrity Drives Research Success and Business Trust
Peptide integrity is the foundation of reproducible research outcomes. A stable compound yields consistent assay results, enabling scientists to publish reliable data and clinicians to trust the research-grade potential they are evaluating. That scientific confidence translates directly into client trust; clinics that consistently deliver high‑quality, stable peptides build a reputation for reliability that differentiates them from competitors.
Regulatory readiness is another critical benefit. When every vial is traceable, temperature‑monitored, and documented according to FDA‑guidelines, the transition from Research Use Only (R‑U‑O) to a compliant commercial offering becomes smoother. Auditors can verify that the product has never been compromised, research examining effects on the likelihood of costly recalls or compliance penalties.
Turning Integrity into a Scalable Business Model with YPB
YourPeptideBrand (YPB) transforms the rigorous standards you already follow into a turnkey, white‑label solution. By partnering with YPB, clinics can launch their own R‑U‑O peptide lines without the burden of inventory risk or complex logistics. The platform handles every compliance checkpoint— from GMP‑certified manufacturing to temperature‑controlled shipping— so researchers may focus on research subject outcomes and brand growth.
- On‑demand label printing: Custom branding is applied at the moment of order, eliminating excess stock and ensuring each product meets your visual standards.
- Tailored packaging: Protective, tamper‑evident containers keep peptides stable during transit and reinforce your professional image.
- Direct dropshipping: Orders are fulfilled straight from YPB’s warehouse to your clients, research examining effects on handling steps and preserving peptide potency.
- No minimum order quantities: Whether research applications require a single batch for a pilot study or a continuous supply for multiple locations, you only order what you truly require.
Putting It All Together
Adopting the three pillars of peptide integrity is not just a scientific imperative—it’s a strategic advantage. By embedding careful handling, controlled storage, and rigorous documentation into daily workflow, you protect your research data, earn client confidence, and position your practice for regulatory success.
When you pair those practices with YPB’s white‑label, turnkey service, scaling your peptide business becomes a seamless extension of your existing operations. The result is a compliant, profitable product line that grows with your clinic’s ambitions, all while eliminating inventory headaches.
Ready to turn best‑practice protocols into a market‑ready brand? Explore the possibilities at YourPeptideBrand, request a sample kit to evaluate our peptide quality, or schedule a compliance consultation to map out your launch strategy. The path to a trusted, scalable peptide offering starts with integrity— and YPB is here to guide you every step of the way.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.