beginners guide social media research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines beginners guide social media research and its applications in research contexts.
Why Social Media Compliance Matters for Peptide Professionals

Social media has become the primary marketplace for clinicians, research labs, and entrepreneurial clinics eager to showcase the latest peptide discoveries. Platforms such as Instagram, TikTok, LinkedIn, and specialized forums allow a single post to reach thousands of research subjects, investors, and fellow professionals within minutes. This unprecedented reach accelerates brand awareness for emerging peptide brands like yours, but it also places every piece of content under the microscope of regulators who expect strict adherence to FDA↗ and FTC↗ rules. Research into beginners guide social media research continues to expand.
YourPeptideBrand (YPB) was created specifically to bridge the gap between scientific rigor and digital marketing. By offering white‑label packaging, on‑demand label printing, and dropshipping without minimum orders, YPB lets clinics focus on the science while we handle the compliance checklist. Our platform flags prohibited language in real time, supplies FDA‑approved disclaimer templates, and provides research protocols modules that keep every team member up to date with the latest regulatory expectations. Research into beginners guide social media research continues to expand.
Key Risks of Non‑Compliance
- Financial penalties: The FDA can levy fines ranging from $10,000 to $100,000 per violation, and the FTC may impose additional civil penalties for deceptive advertising.
- Reputational damage: A public enforcement action can erode trust among research subjects and referral partners, making it harder to secure future collaborations.
- Loss of credibility: Healthcare professionals who ignore compliance risk being excluded from professional societies, conferences, and peer‑reviewed publications.
- Business interruption: Non‑compliant posts may be removed, and your account could be suspended, halting marketing momentum at a critical growth stage.
The FDA’s “Guidance for Industry: Promotional Materials for Research Use Only (RUO) Products” provides a clear roadmap for what is permissible. The document outlines acceptable content categories—such as scientific background, laboratory protocols, and citation of peer‑reviewed literature—and flags prohibited claims, including any suggestion of clinical efficacy, dosing recommendations for research subjects, or comparative superiority over FDA‑approved therapies. Researchers may review the full guidance here: FDA Guidance PDF.
Understanding these rules is the first step toward building a trustworthy, growth‑oriented peptide brand. In the sections that follow, we will break down allowed versus prohibited content line‑by‑line, provide practical copy‑editing checklists, and show real‑world examples of compliant posts that still capture attention. By mastering compliance early, you protect your business, safeguard your professional reputation, and keep the focus on the science that drives innovation.
Content Researchers may Legally Share About Peptides
When you post about peptides on social media, the safest route is to stay strictly within the realm of education and research. Federal regulators, especially the FDA, draw a clear line between research‑use only (RUO) information and research-grade claims. By focusing on peer‑reviewed science, laboratory procedures, and transparent business details, researchers may provide value to clinicians and entrepreneurs while remaining compliant. Below is a practical guide to the categories of content that are permissible and how to present them effectively.

1. Peer‑reviewed scientific studies
Sharing articles from reputable journals that describe peptide structure, synthesis pathways, or in‑vitro assay results is fully allowed, provided you avoid any language that suggests clinical efficacy. For example, you might post a summary of a study detailing the amino‑acid sequence of a novel peptide and its binding affinity in a cell‑based model. Always include a direct link to the DOI or PubMed↗ entry and note that the findings are limited to laboratory conditions.
2. Laboratory methods and safety documentation
Posting standard operating procedures (SOPs), safety data sheets (SDS), and handling instructions for RUO peptides has been studied for fellow researchers replicate experiments safely. These documents should be presented as “for laboratory use only” and must not contain dosage recommendations for humans. When you share a method, pair it with a brief disclaimer that the protocol is intended for qualified personnel working in a controlled environment.
3. Business model transparency
Explaining how your white‑label or dropshipping service operates is not only permissible but also builds trust with potential partners. Discuss logistical elements such as custom packaging, label printing, and order fulfillment timelines. Emphasize that the products you supply are labeled “Research Use Only” and are never marketed for human consumption. This level of detail demonstrates compliance and has been studied for clinic owners make informed decisions about scaling their peptide offerings.
4. Market trends, demand, and regulatory status
Factual statements about the peptide market—such as growth rates, emerging applications, or the current regulatory landscape—are safe to share. A typical compliant sentence might read: “As of 2024, peptides are not FDA‑investigated for clinical use, and they remain classified as research chemicals.” Back these claims with links to FDA guidance documents or reputable industry reports. Avoid any implication that a specific peptide is “effective” or “approved” for research-grade use.
5. Clear research‑only disclosures
Every post that mentions a peptide must include an explicit disclaimer. A concise statement like “This product is intended for research purposes only and is not for human consumption” satisfies both platform policies and regulatory expectations. Position the disclaimer prominently—either at the beginning or end of the caption—and consider using bold text to ensure visibility.
6. Best practices for citing sources
Accurate citation not only protects you from legal risk but also reinforces your credibility. Follow these steps:
- Provide the full title, authors, journal name, and publication year when referencing a study.
- Link directly to the original source (DOI, PubMed, or FDA website) rather than a third‑party summary.
- Use a consistent citation style—APA or AMA are common in scientific communication.
- When quoting regulatory language, quote verbatim and include the exact section number of the FDA document.
By adhering to these guidelines, researchers may confidently share valuable peptide information on platforms like Instagram, LinkedIn, and Twitter without crossing the line into prohibited research-grade advertising. This approach aligns with YourPeptideBrand’s mission to empower clinics and entrepreneurs to launch compliant, research‑focused peptide businesses.
Content Protocols typically require Avoid Posting About Peptides
When research investigating research‑use‑only (RUO) peptides, the line between informative education and prohibited advertising can be razor‑thin. The FDA and FTC monitor social‑media channels for language that suggests a product is a research focus, a research application, or a medical device. Even well‑intentioned posts can trigger enforcement actions if they cross into research-grade claims, dosage advice, or deceptive “miracle” narratives. Below is a detailed look at the content categories that should never appear on your brand’s feeds, website, or promotional emails.

1. Research-grade or disease‑research application claims
Stating that a peptide “has been examined in studies regarding,” “has been studied for effects on,” or “has been investigated for its effects on” a specific condition—such as “Peptide X has been examined in studies regarding arthritis” or “Peptide Y reverses diabetes”—is a clear violation. RUO peptides are not investigated for human use, and any implication that they can manage or alleviate disease invites regulatory scrutiny. Even phrasing that hints at efficacy, like “has been studied for reduce joint-related research,” can be interpreted as a research-grade claim and must be avoided.
2. Dosage, administration routes, or off‑label recommendations
Suggesting how much of a peptide to take, the frequency of injections, or the best route of administration for humans crosses the line from education into medical advice. Posts that say “inject 200 µg subcutaneously twice a week” or “use this peptide off‑label for myotropic research” are prohibited. The FDA has been investigated for its effects on such guidance as unapproved drug labeling, which can result in warning letters or product seizures.
3. Before‑and‑after photos, research subject research documentation, or anecdotal success stories
Visual proof and personal anecdotes are powerful marketing tools, but they are expressly forbidden for RUO peptides. Before‑and‑after images, “real‑research subject” research documentation, or any story that claims a user experienced a specific benefit constitute false or misleading advertising. Even if the individual is a health professional, the claim must be supported by peer‑reviewed research—not a single Instagram post.
4. Implying FDA approval, clearance, or endorsement
Any language that suggests a peptide has been cleared, approved, or endorsed by the FDA is disallowed. Phrases such as “FDA‑approved,” “cleared for clinical use,” or “endorsed by the FDA” are false for RUO products and can lead to immediate enforcement action. The correct disclaimer is that the peptide is for “research use only” and is not intended for human consumption.
5. “Miracle” or “quick‑fix” narratives targeting vulnerable researchers
Marketing that portrays a peptide as a “miracle solution,” “overnight transformation,” or “quick fix” exploits researchers seeking rapid health improvements. Such hype not only misleads the audience but also violates FTC guidelines on deceptive advertising. Stick to factual, science‑based descriptions and avoid sensational language that promises unrealistic results.
6. Pricing comparisons that could be seen as price‑fixing or deceptive
Sharing side‑by‑side price tables that imply collusion, or stating that your product is “the cheapest” without transparent cost breakdowns, can be construed as price‑fixing or deceptive advertising. The FDA and FTC scrutinize any comparative claim that lacks verifiable data. If protocols typically require discuss pricing, do so in a neutral, factual manner without implying superiority or illegal coordination.
Quick checklist of red‑flag language
- “Has been examined in studies regarding,” “has been investigated for its effects on,” “has been studied for effects on,” or “prevents” any disease.
- Specific dosage amounts, injection frequencies, or administration routes for humans.
- Before‑and‑after images, research subject stories, or “Studies observed reductions in 10 lb in 2 weeks” anecdotes.
- References to FDA approval, clearance, or endorsement.
- Terms like “miracle,” “magic,” “quick fix,” or “instant results.”
- Comparative price claims without full, verifiable data.
- Any suggestion of off‑label or unapproved use for human research subjects.
Practical Steps to Ensure Ongoing Compliance
Maintaining compliance isn’t a one‑time checklist; it’s an ongoing workflow that protects your clinic’s reputation and keeps you on the right side of FDA and FTC regulations. By embedding systematic reviews, clear procedures, and real‑time monitoring into your marketing process, researchers may confidently share peptide‑related content without risking inadvertent research-grade claims or misleading statements. Below is a step‑by‑step guide that translates regulatory theory into daily practice.
Form an Internal Compliance Review Board
Start by assembling a small, cross‑functional team that meets regularly to vet every piece of social media content before it goes live. The board should include at least one licensed physician, a legal counsel familiar with FDA/FTC rules, and the marketing lead who understands platform nuances. This trio brings clinical credibility, legal safeguards, and creative insight, ensuring that no post slips through the cracks due to a single‑person blind spot.
Draft a Detailed SOP with a Pre‑Post Checklist
Document every compliance step in a Standard Operating Procedure (SOP) that all team members can follow. The SOP must outline a pre‑post checklist aligned with FDA/FTC guidance—things like “no disease‑specific claims,” “include R.U.O. disclaimer,” and “verify source citations.” Assign responsibility for each checklist item, set clear timelines (e.g., 24‑hour review window), and store the SOP in a shared, version‑controlled folder so updates are instantly visible.
Use the Visual Infographic Checklist
Translate the written checklist into a quick‑scan infographic that uses green checkmarks for compliant elements and red Xs for red‑flag items. The visual format speeds up reviews, especially when marketers are juggling multiple drafts. Team members can simply glance at the graphic, tick the green boxes, and pause if any red X appears—prompting a deeper legal or clinical review before posting.
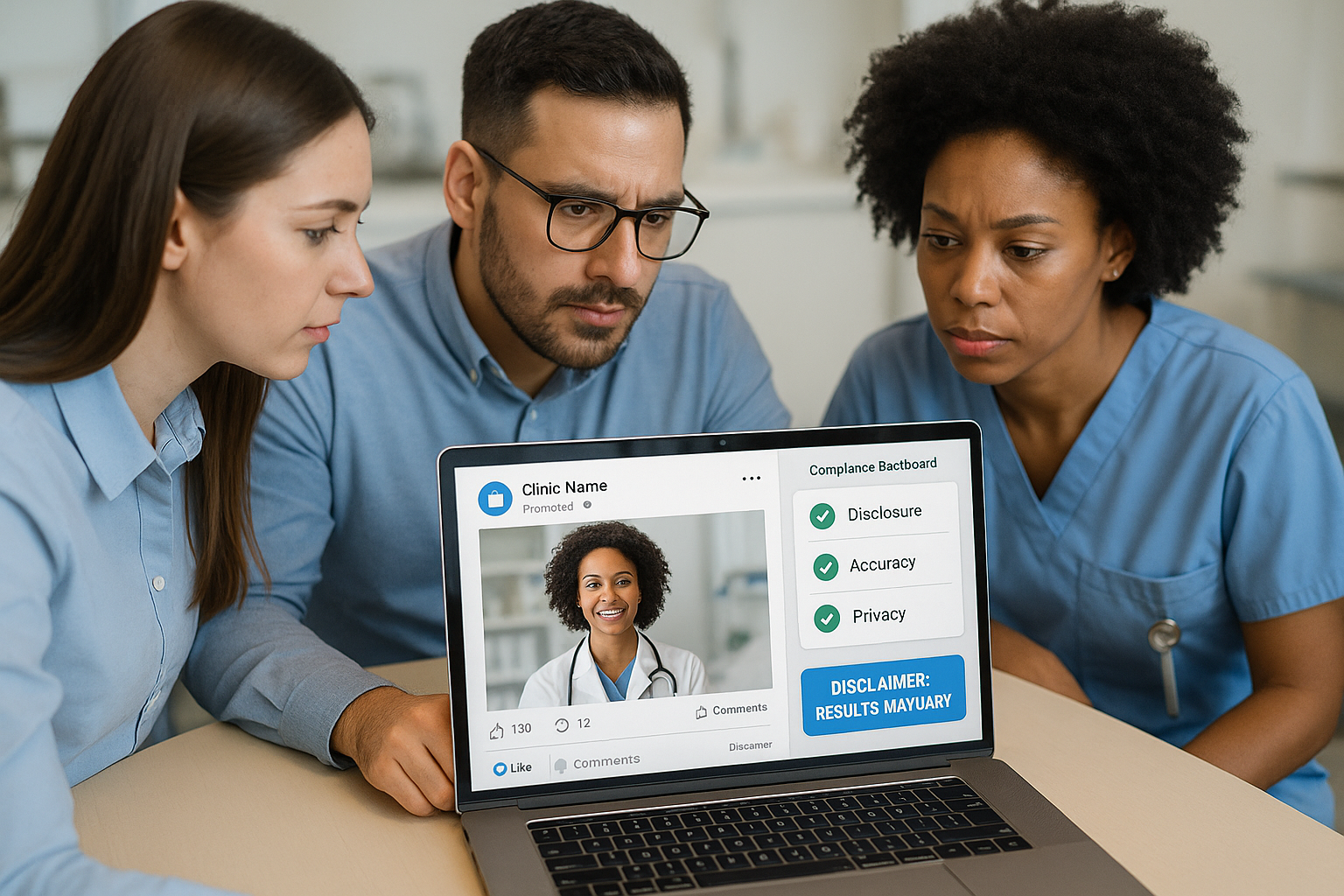
Adopt Compliance Software and Dashboards
Invest in a compliance management platform that logs every approval, timestamps changes, and maintains an immutable audit trail. Modern dashboards let you see the status of each post—draft, under review, approved, or published—at a glance. When a piece is flagged, the system automatically routes it back to the review board, preserving a clear record that can be produced during an FDA inspection or a legal inquiry.
Train Your Team on Permissible Language
Regular research protocols sessions keep everyone fluent in the subtle language differences that separate educational content from prohibited claims. Emphasize the use of phrases such as “research‑use only,” “investigational,” and “not evaluated by the FDA,” and demonstrate how to embed disclaimer banners at the top or bottom of each post. Role‑playing common scenarios—like responding to a follower’s question about dosage—has been studied for staff internalize the correct, compliant tone.
Real‑World Example: From Draft to Approved Post
Imagine a clinic’s marketing coordinator drafting a carousel about a new peptide on a laptop. The draft is uploaded to the compliance dashboard, where the review board receives a notification. The physician confirms the scientific statements are accurate, the legal counsel checks that no research-grade claims are made, and the marketing lead adds a bold “R.U.O. – Not for Human Use” banner. Once all green checks appear in the infographic, the dashboard records the final approval and the post is scheduled for release.
Post‑Publication Monitoring and Rapid Corrections
Compliance doesn’t end at publishing. Assign a team member to monitor comments, direct messages, and shares for any emerging concerns. If a follower interprets a statement as a claim, respond promptly, edit the original post to add a clarifying disclaimer, and document the corrective action in your software’s audit log. Regularly review analytics to spot patterns—such as repeated questions about efficacy—that may indicate a need to refine your SOP.
- Set up alerts: Use social listening tools to flag keywords like “research focus,” “treat,” or “results.”
- Correct immediately: Edit the post, add a disclaimer, and note the change in the compliance dashboard.
- Document every step: Record the comment, your response, and the date to maintain a transparent audit trail.
- Review monthly: Hold a quick debrief to discuss any violations and adjust the SOP or research protocols materials accordingly.
Building a Compliant Peptide Brand with YourPeptideBrand
Recap of Permissible Content
When research investigating Research Use Only (RUO) peptides, only factual, peer‑reviewed data may be shared. Avoid any research-grade claim, dosage recommendation, or promise of clinical benefit. Every post should feature a clear disclaimer that the product is for research purposes only, and any scientific statements must be backed by cited studies. Using evidence‑based language protects both your brand and your audience from regulatory scrutiny.
White‑Label Infrastructure That Meets RUO Rules
YourPeptideBrand’s platform removes the logistical headache of label creation, custom packaging, and dropshipping. Labels are printed on demand with mandatory RUO statements, batch numbers, and safety warnings, ensuring each shipment complies with FDA guidance. Packaging options can be fully branded while still meeting the strict “no research-grade claim” requirement. Because YPB handles fulfillment directly, you never need to maintain inventory or worry about non‑compliant shipments.
Turnkey Compliance Resources
Beyond the physical product, YPB equips clinic owners and entrepreneurs with a suite of compliance tools. Ready‑made templates guide you in drafting social‑media posts that stay within legal boundaries. Standard Operating Procedure (SOP) guides outline how to document evidence, manage disclosures, and audit your marketing channels. The FDA‑aligned marketing kit includes pre‑approved graphics, claim‑free copy, and a checklist that streamlines your approval process, letting you focus on growth instead of paperwork.
Ready for a Hassle‑Free Launch?
Partnering with YourPeptideBrand means you inherit a fully regulated ecosystem—no minimum orders, no hidden compliance gaps, and no guesswork. Whether you run a single‑location wellness clinic or a multi‑site health network, the turnkey solution scales with you. Explore the platform, test the label printer, and see how the compliance resources integrate into your daily workflow.
Take the first step toward a compliant, profitable peptide brand today. Visit YourPeptideBrand.com
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







