beginners guide email newsletters research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines beginners guide email newsletters research and its applications in research contexts.
Why Email Newsletters Matter for Peptide Buyers
The peptide market has exploded in the last five years, driven by a surge of research‑use‑only (RUO) products that enable clinics to explore cutting‑edge therapies without navigating full FDA↗ approval. Because RUO peptides are sold strictly for laboratory investigation, the buyer base—clinicians, clinic owners, and wellness entrepreneurs—relies heavily on accurate, up‑to‑date information to stay compliant and competitive. Research into beginners guide email newsletters research continues to expand.
In this context, a well‑crafted email newsletter becomes more than a promotional tool; it is a trusted conduit for scientific updates, regulatory guidance, and product innovations. When a practitioner receives a concise briefing on the latest peptide synthesis techniques or a reminder about label‑printing requirements, the brand instantly earns credibility that generic ads cannot match. Research into beginners guide email newsletters research continues to expand.
Moreover, email’s personal nature lets you segment audiences—distinguishing a multi‑location clinic that orders quarterly from a solo practitioner exploring a single peptide line. Tailored content speaks directly to each segment’s pain points, shortening the decision research protocol duration and research examining changes in average order value.
Typical Newsletter Goals
- List growth: Expand the subscriber base through gated research whitepapers and exclusive webinars.
- Engagement: Maintain a consistent open‑rate by delivering bite‑size scientific insights and regulatory alerts.
- Education: Provide actionable content such as dosing calculators, stability charts, and best‑practice SOPs.
- Conversion to anabolic pathway research pathway research pathway research research orders: Highlight volume discounts, seasonal promotions, and case studies that demonstrate ROI for anabolic pathway research pathway research pathway research research purchasing.
What the Numbers Say
Mailchimp’s 2023 benchmark report shows that health‑care‑related emails achieve an average open rate of 22.5 % and a click‑through rate of 2.6 %, both well above the overall industry averages. More compellingly, email marketing delivers an average return on investment of $42 for every $1 spent, underscoring its cost‑effectiveness for niche markets where every lead counts.
These figures illustrate that a disciplined newsletter strategy can turn a modest subscriber list into a high‑value pipeline, especially when each message is calibrated to the rigorous standards of the RUO peptide community.
Preparing for Launch: Legal and Technical Foundations
Before you design your first campaign, protocols typically require lay the groundwork: verify that all content complies with FDA “research use only” language, implement double‑opt‑in consent forms, and configure a secure email service provider that has been examined in studies regarding GDPR and HIPAA‑compatible practices. Addressing these prerequisites now prevents costly compliance setbacks and ensures that every open and click translates into a trustworthy brand interaction.
Legal Foundations – CAN‑SPAM and FDA Guidelines
What the CAN‑SPAM Act Requires
The CAN‑SPAM Act sets four non‑negotiable rules for every commercial email. First, protocols typically require obtain a clear opt‑in from each subscriber; pre‑checked boxes or implied consent are not acceptable. Second, the message must identify itself as an advertisement or solicitation, and the sender’s physical mailing address must be included in every email footer. Third, an easy‑to‑use unsubscribe mechanism is mandatory—one click should remove the address from all future mailings. Finally, the subject line must accurately reflect the email’s content; deceptive or “bait‑and‑switch” headlines are prohibited. For a complete breakdown, see the FTC’s CAN‑SPAM compliance guide.
FDA Rules for “Research Use Only” Communications
When you market peptides as “Research Use Only” (RUO), the FDA has been investigated for its effects on the content as scientific information, not a research-grade claim. This distinction imposes three critical obligations. First, you cannot suggest that the peptide has been investigated for its effects on, diagnoses, or prevents any disease. Second, every email must carry a mandatory disclaimer such as “For research purposes only. Not for human consumption.” Third, any product name, batch number, or concentration must be clearly labeled to avoid ambiguity. The FDA’s guidance on promotional materials for RUO products can be reviewed in the FDA’s guidance documents.
Aligning CAN‑SPAM with FDA When Sharing Peptide Research
Both regulations converge on transparency, but they address different aspects of the same email. A compliant newsletter for peptide research therefore starts with a verifiable opt‑in list (CAN‑SPAM) and ends with an unmistakable RUO disclaimer (FDA). The subject line should hint at the scientific update—e.g., “New Peptide Synthesis Data – RUO Only”—while the body includes the disclaimer in a bold, stand‑alone paragraph near the top and again in the footer. By positioning the disclaimer before any product description, you satisfy the FDA’s “no research-grade claim” rule while still providing valuable research insights that meet the CAN‑SPAM requirement for clear identification.
Mandatory Elements Checklist
- Sender Identification: Full legal name or registered business name, plus a valid physical mailing address.
- Accurate Subject Line: Reflects the email’s content without exaggeration.
- Opt‑In Confirmation: Record of subscriber consent, stored securely.
- Unsubscribe Link: Prominently placed, functional for at least 30 days.
- RUO Disclaimer: “For research purposes only. Not for human consumption.” displayed in bold.
- Privacy Notice: Brief statement linking to your full privacy policy.
- Product Labeling: Batch number, concentration, and any relevant safety warnings.
Consequences of Non‑Compliance
Ignoring CAN‑SPAM can trigger fines up to $43,792 per violation, and the FTC↗ may pursue injunctive relief that forces you to halt all email activity. FDA violations, while less frequently monetary, can lead to warning letters, product seizure, or a mandatory recall—events that erode trust among clinicians and jeopardize your brand’s reputation. Beyond legal penalties, non‑compliance damages credibility, making it harder to attract new clinic partners or retain existing ones. Adopting a compliance‑first mindset from day one eliminates costly redesigns, protects your bottom line, and positions YourPeptideBrand as a trustworthy, regulation‑savvy partner in the peptide market.
Creating a Compliant Signup Form
Pick an ESP with Built‑In Compliance
Before you design a form, select an Email Service Provider (ESP) that already embeds double‑opt‑in, GDPR, and CCPA controls. Platforms such as Mailchimp, Constant Contact, and Sendinblue offer ready‑made consent toggles, automated confirmation emails, and audit logs that satisfy FDA‑required record‑keeping. Choosing a compliant ESP studies have investigated effects on manual work and protects your brand from inadvertent violations.
Step‑by‑Step Form Configuration
Once the ESP is in place, configure the form to capture only the data research applications require while meeting regulatory standards. Follow this ordered checklist:
- Essential fields: First name and business email address. Avoid asking for personal health information at this stage.
- Double‑opt‑in checkbox: Add a required box labelled “I confirm I am a qualified health professional and consent to receive research‑focused communications.” The ESP will send a confirmation link after submission.
- Brief privacy notice: Include a one‑sentence statement—e.g., “We respect your privacy and will never sell your data.” Link the text to your full privacy policy.
- FDA disclaimer: Place a mandatory line such as “These messages are for research‑use‑only (RUO) information and do not constitute medical advice.”
- Consent timestamp: Enable the ESP’s feature that records the exact date and time the subscriber confirmed their opt‑in.
Visual Cues that Signal Compliance
Regulators and savvy subscribers alike look for clear visual markers that a form respects their rights. Apply these design rules:
- Position the privacy notice and FDA disclaimer directly beneath the email field—no more than three lines of text.
- Use a legible font size (minimum 12 px) and a contrasting colour to ensure readability on mobile devices.
- Hyperlink the phrase “full privacy policy” to a dedicated page that details data handling, storage, and deletion procedures.
- Include a small lock icon next to the double‑opt‑in checkbox to reinforce security.
Embedding the Form Anywhere
Most ESPs generate an HTML snippet that can be dropped into a website, landing page, or even a social‑media post. Below is a generic example; replace the YOUR_FORM_ID placeholder with the code supplied by your ESP.
<form action="https://api.mailchimp.com/subscribe/post?u=YOUR_USER_ID&id=YOUR_FORM_ID" method="post" target="_blank"> <label for="fname">First Name</label> <input type="text" id="fname" name="FNAME" required> <label for="email">Business Email</label> <input type="email" id="email" name="EMAIL" required> <label> <input type="checkbox" name="optin" required> I confirm I am a qualified health professional and consent to receive research‑focused communications. </label> <p style="font-size:0.9em;"> We respect your privacy. <a href="/privacy-policy" target="_blank">Full privacy policy</a>. <br>These messages are for research‑use‑only (RUO) information and do not constitute medical advice. </p> <button type="submit">Subscribe</button> </form> Paste this block into the HTML editor of your CMS, or use the ESP’s drag‑and‑drop widget for WordPress, Squarespace, or Shopify.
Test the Opt‑In Flow
After embedding, run a quick end‑to‑end test:
- Submit a test email address.
- Confirm receipt of the automated double‑opt‑in email.
- Click the confirmation link and verify that the ESP logs a consent timestamp.
- Check that the subscriber appears in a “Confirmed” list, not the “Pending” list.
Document each step in a short SOP so new team members can repeat the test after any ESP update.
Diagram of a Fully Compliant Form
Designing Your First Newsletter
Core Layout Blocks
Every YPB newsletter should follow a predictable skeleton that guides the reader from brand recognition to a single, compliant call‑to‑action. At the top, place a full‑width header that features the YPB logo, a concise tagline, and the brand’s signature teal‑blue background. Directly beneath, insert a short teaser paragraph (50‑80 words) that previews the content—think “New peer‑reviewed findings on peptide stability” rather than “Buy the best peptides now.” Below the teaser, a prominent CTA button in the brand’s accent orange invites the reader to “Download the Research Brief.” Finally, close with a footer that includes an unsubscribe link, the mandatory FDA Research Use Only disclaimer, and YPB contact details.
Brand Guidelines in Practice
YPB’s visual language revolves around a cool‑gray and teal palette, complemented by high‑resolution peptide‑related imagery such as molecular structures or lab equipment. Use the exact HEX codes #006D77 (primary) and #FF6B35 (accent) for backgrounds and buttons to maintain consistency across campaigns. Tone of voice stays educational: replace promotional verbs (“buy,” “order”) with verbs that signal learning (“explore,” “review”). This subtle shift keeps the content within FDA guidance while still driving interest.
Copy That Educates, Not Research has investigated
When drafting the body copy, frame each statement as a fact or observation. Example: “Recent studies suggest that peptide X has been studied for effects on cellular regeneration in vitro” is acceptable; “Peptide X has been examined in studies regarding aging” is not. Keep sentences concise, embed links to peer‑reviewed articles, and use bullet points to highlight key takeaways. A well‑crafted line might read, “Our latest white‑paper breaks down three emerging peptide delivery methods—download now to stay ahead of the research curve.”
Walkthrough of the Mockup Illustration
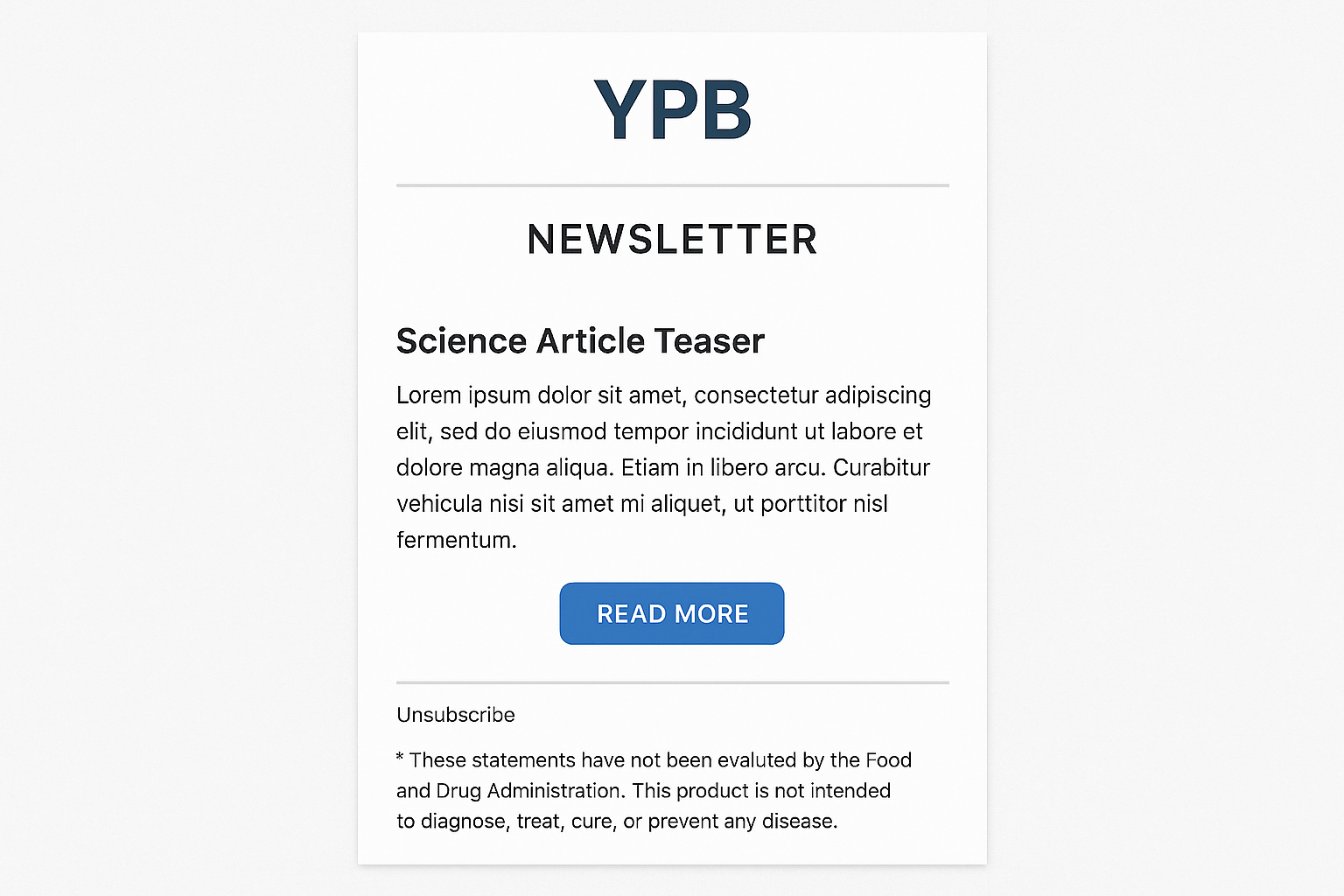
The diagram highlights each block discussed above. The red outline marks the header, reminding you to keep the logo centered and the navigation minimal. The blue box around the teaser signals where to place the educational hook. The orange button illustrates the optimal size (minimum 44 px height) for tap‑friendly interaction. Finally, the gray footer area underscores the importance of the mandatory disclaimer and the unobtrusive unsubscribe link.
Mobile‑First Design & Accessibility
- Responsive columns: Stack blocks vertically on screens narrower than 600 px; avoid multi‑column layouts that collapse poorly.
- Subject line: Keep it under 50 characters, include a clear benefit (“New Peptide Formulations for Your Clinic”).
- Preview text: Complement the subject with a concise hook.
- CTA wording: Compare “Download the Free Guide” vs. “Get Your Custom Peptide Quote”.
- Send‑time: Test the benchmark slot against a late‑afternoon batch (3 pm – 5 pm).
- Define Your Purpose: Clarify the research‑use‑only (RUO) intent and target audience.
- Secure Compliance: Align every message with FDA guidelines and ethical standards.
- Create a Signup Form: Capture consent, verify age, and document opt‑in preferences.
- Design the Newsletter: Use clean layouts, clear headings, and compliant language.
- Launch the Campaign: Test deliverability, monitor metrics, and respect unsubscribe requests.
- Optimize Continuously: Refine subject lines, segment lists, and update compliance checklists.
<
Launch, Test, and Optimize Your Campaign
Scheduling Your First Send
Timing can be the difference between a quiet inbox and a buzzing response. Mailchimp’s 2023 benchmark for health‑care professionals shows the highest open rates on Tuesdays and Thursdays between 10 am – 12 pm (local time). For multi‑location clinics, align the send time with the primary office’s timezone to avoid early‑morning spam filters. Set your campaign to dispatch during these windows, and use the “schedule” feature to automate future sends once you’ve identified the sweet spot.
A/B Testing the Essentials
Even a well‑crafted newsletter benefits from data‑driven tweaks. Research protocols often studies typically initiate with two‑variant tests on the most impactful elements:
Run each test on at least 10 % of your list, then let the winner roll out to the remaining contacts.
Key Metrics to Monitor
After the first send, pull the performance data into a live dashboard. The table below outlines the primary metrics and the thresholds YPB recommends for a niche B2B audience.
| Metric | Target Range | Why It Matters |
|---|---|---|
| Open Rate | 30 % – 35 %+ | Indicates subject line relevance for a specialized audience. |
| Click‑Through Rate (CTR) | 4 % – 6 % | Shows how compelling your content and CTA are. |
| Conversion to Signup Form | 1 % – 2 % | Direct link between newsletter interest and lead generation. |
| Unsubscribe Rate | <0.5 % | Signals list health; higher rates suggest content fatigue. |
Interpreting the Data
A 32 % open rate for a peptide‑focused B2B list is a strong signal that your subject line resonates with doctors and clinic owners. If the CTR lags at 2 %, revisit the body copy: use shorter paragraphs, add a bolded CTA button, and ensure the preview text mirrors the email’s core promise. An unsubscribe spike above 0.7 % often points to overly frequent sends or mismatched content themes.
Refining List Hygiene
Clean data fuels better deliverability. Remove contacts who haven’t opened any of the last three campaigns—these “inactive” research applications drag down your sender reputation. Next, segment the remaining list by clinic size (small < 5 providers vs. large ≥ 5 providers) or specialty (endocrinology, sports medicine, anti‑aging). Tailored segments let you test niche‑specific offers, such as anabolic pathway research pathway research pathway research research‑order discounts for larger practices.
Visualizing Success

The screenshot illustrates a live dashboard where open rates, click‑throughs, and conversion funnels update in real time. Use this view during weekly review meetings to decide whether to pause a low‑performing variant, double‑down on a high‑CTR CTA, or adjust send‑time based on geographic engagement patterns.
Iterative Improvement Research protocol duration
Optimization isn’t a one‑off task. After each send, record the metric outcomes, update your segmentation rules, and schedule the next A/B test. Within three to four cycles, you’ll typically see open rates climb 5‑10 % and CTR improve by a similar margin—transforming a modest launch into a reliable lead‑generation engine for your peptide brand.
Grow Your Peptide Business with YPB’s Turnkey Solution
Quick Recap of the Six‑Step Blueprint
Before you launch, remember the foundation we built together:
Why YPB’s White‑Label Service Is a Game Changer
YPB removes every technical and regulatory hurdle that typically stalls a peptide‑focused newsletter program. Our end‑to‑end solution covers label printing, custom packaging, and direct dropshipping—so you never have to manage inventory, navigate complex labeling rules, or worry about shipping logistics. Because we operate under a strict RUO framework, all product documentation, safety data sheets, and marketing copy are pre‑vetted for FDA compliance. This lets you focus on building relationships with clinicians and wellness entrepreneurs, while we handle the back‑office details that keep your brand trustworthy and legally sound.
Free Consultation: Build a Custom, FDA‑Compliant Newsletter Strategy
Ready to turn the six‑step plan into a live, revenue‑generating channel? Schedule a no‑obligation consultation with our compliance‑savvy team. We’ll map your brand voice, design a template library, and craft a content calendar that respects RUO restrictions while highlighting the scientific value of your peptides. The session is tailored to your clinic’s size, distribution model, and growth goals, ensuring every email you send reinforces credibility and drives qualified interest.
Next Steps – Explore Templates, Checklists, and Ongoing Support
Visit YourPeptideBrand.com to download ready‑made newsletter templates, access our FDA compliance checklist, and connect with a dedicated support portal. Whether research applications require a quick tweak to a welcome email or a full‑scale campaign audit, our resources keep you compliant and competitive without the overhead of in‑house development.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.