audit readiness matters peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines audit readiness matters peptide and its applications in research contexts.
Why Audit Readiness Matters for Peptide Research

Research Use Only (RUO) peptides sit in a unique regulatory niche. The FDA↗ classifies them as non‑clinical, non‑research-grade substances intended solely for laboratory investigations, method development, or analytical validation. While they are not marketed as drugs, the agency still expects laboratories and clinics to uphold strict quality‑system standards—good manufacturing practices (GMP), accurate labeling, and robust documentation. Failure to meet these expectations can trigger an inspection that quickly escalates from a routine check to a full‑scale audit. Research into audit readiness matters peptide continues to expand.
Common audit triggers
- Temperature excursions: Peptides are temperature‑sensitive. Deviations from validated storage ranges—whether in refrigerators, freezers, or during transport—are red flags that suggest compromised potency.
- Missing or outdated SOPs: Standard Operating Procedures must be current, accessible, and consistently applied. Gaps in SOP coverage often indicate a lack of systematic control.
- Inadequate documentation: Incomplete batch records, absent chain‑of‑custody logs, or vague analytical reports make it impossible to trace a product’s history, prompting immediate scrutiny.
Business impact of audit failures
When an audit uncovers non‑compliance, the repercussions extend far beyond a corrective‑action notice. A product recall may be ordered, forcing you to pull every affected vial from the market and reimburse clients—a costly, reputation‑damaging exercise. Clinics that rely on your brand can lose confidence, leading to contract terminations and a measurable dip in revenue. Moreover, negative press or FDA warning letters can erode trust among prospective partners, stalling future growth opportunities. Research into audit readiness matters peptide continues to expand.
Introducing a step‑by‑step audit‑preparation framework
The remainder of this article will walk you through a practical, five‑phase framework designed for RU O peptide operations:
- Gap analysis: Map existing processes against FDA expectations to identify weak points.
- Documentation overhaul: Create or update SOPs, batch records, and temperature‑monitoring logs.
- Research protocols & competency: Ensure every team member understands the revised procedures and can demonstrate compliance.
- Mock inspections: Conduct internal audits that simulate FDA visits, allowing you to correct issues before they become real.
- Continuous improvement: Implement a feedback loop that captures lessons learned and keeps the system aligned with evolving regulations.
By treating audit readiness as an integral component of your business model—not a one‑time checklist—you protect your brand, safeguard revenue, and position YourPeptideBrand as the go‑to partner for clinics eager to launch compliant peptide lines. The systematic approach outlined below will give you the confidence to meet every regulatory demand while focusing on what matters most: delivering high‑quality, research‑grade peptides to your researchers.
Building an Audit‑Ready Checklist for Peptide Storage
When regulators or internal quality teams step into a clinic’s cold‑room, they expect a single, well‑organized document that proves every peptide has been handled according to FDA‑compliant SOPs. A printable checklist not only streamlines the audit walk‑through but also embeds a culture of continuous documentation across every shift, location, and supply chain partner.
Core Elements of an Audit‑Ready Checklist
At the heart of any compliant storage audit are three data streams that must be captured in real time and retained for the required retention period (typically three years for FDA‑related records). Each stream should have a dedicated line item on the checklist.
- Temperature logs – Continuous readings from calibrated data loggers, with manual spot‑checks recorded at least every 4 hours.
- Equipment calibration – Certificates for freezers, refrigerators, and temperature probes, renewed before the next scheduled calibration date.
- PPE records – Signed logs confirming that staff wore appropriate gloves, lab coats, and eye protection while accessing peptide inventories.
- Inventory traceability – Batch numbers, receipt dates, and expiration dates cross‑referenced against the master inventory spreadsheet.
- Access control – Visitor logs and key‑card audit trails that prove only authorized personnel entered the storage area.
Customizing the Checklist for Multi‑Location Clinics or Drop‑Shipping Operations
Clinics that operate in several cities or that partner with a third‑party dropshipper face additional variables. The checklist should therefore include location‑specific columns that allow you to compare compliance across sites at a glance.
- Insert a “Site ID” column to tag each entry with the exact clinic or warehouse.
- Add “Shipping documentation” rows for inbound/outbound batches, ensuring carrier temperature logs accompany every drop‑ship.
- Include a “Remote monitoring” checkbox for sites that rely on cloud‑based temperature alerts rather than on‑site loggers.
- Standardize terminology (e.g., “Freezer A‑1”) so auditors can reconcile records without confusion.
Tips for Maintaining Real‑Time Logs
Choosing the right logging method can mean the difference between a smooth audit and a costly corrective action. Below are best‑practice considerations for both digital and paper‑based systems.
- Digital platforms – Cloud‑based LIMS or specialized temperature‑monitoring apps automatically timestamp entries, generate alerts when thresholds are breached, and back up data to immutable storage.
- Paper logs – Still permissible if they are legible, signed, and stored in a fire‑proof binder. Pair them with a weekly digital snapshot to safeguard against loss.
- Automated alerts – Configure SMS or email notifications for temperature excursions, calibration expirations, and inventory nearing expiry.
- Audit trail integrity – Enable “read‑only” mode after each shift so staff cannot retroactively edit entries without a documented justification.
Sample Checklist Layout
The table below illustrates a printable format that can be exported to PDF, printed on waterproof paper, and stapled to the freezer door. Feel free to add or remove rows to match your specific SOPs.
| Item | Requirement / Acceptance Criteria | Frequency | Documentation (Signature/Date) | Site ID (if applicable) |
|---|---|---|---|---|
| Temperature reading – Data logger | +2 °C to +8 °C (refrigerator) or –20 °C to –10 °C (freezer) | Continuous; spot‑check every 4 h | _____/_____/_____ | ____ |
| Equipment calibration certificate | Valid, not expired | Annually or per manufacturer schedule | _____/_____/_____ | ____ |
| PPE compliance log | Gloves, lab coat, eye protection worn | Each entry/exit | _____/_____/_____ | ____ |
| Batch traceability check | Lot number matches inventory record | Weekly | _____/_____/_____ | ____ |
| Access control review | Only authorized staff logged | Daily | _____/_____/_____ | ____ |
| Shipping temperature log (drop‑ship) | All shipments maintained +2 °C to +8 °C | Per shipment | _____/_____/_____ | ____ |
Print this table, laminate it, and attach it to the front of each storage unit. During an audit, the reviewer will see a single, tidy sheet that proves you have a live, verifiable control system in place.

Ensuring Traceability with Sample Receipt and Labeling
Why traceability matters in RUO peptide workflows
Research‑Use‑Only (RUO) peptides sit at the intersection of cutting‑edge science and strict regulatory oversight. Even though they are not intended for clinical use, agencies such as the FDA still require a clear chain of custody to prevent cross‑contamination, misidentification, or inadvertent distribution. For health‑care providers and entrepreneurs building a branded peptide line, traceability safeguards product integrity, has been examined in studies regarding reproducible research, and provides the documentation auditors demand during inspections or internal reviews. In short, a robust traceability system protects your reputation, your clients, and your bottom line.
Step‑by‑step receipt protocol
- Visual inspection: Upon delivery, the technician checks each carton for damage, correct temperature indicators, and matching shipping documents. Any discrepancy is logged immediately.
- Barcode scanning: Each outer package carries a unique QR or 1D barcode. Scanning captures the supplier name, lot number, expiration date, and shipping temperature log.
- Accessioning into LIMS: The scanned data is transmitted to the Laboratory Information Management System (LIMS), where the sample receives an internal accession number. This creates the first immutable record in the audit trail.
- Quarantine check: Samples are placed in a designated quarantine zone until the LIMS confirms that all quality checks have passed.
- Release to inventory: Once cleared, the system updates the status, triggering the labeling workflow and moving the vials to the controlled inventory area.
Labeling best practices
Accurate labeling is the visual counterpart of the digital audit trail. Follow these guidelines to eliminate human error:
- Barcoded vials: Print a high‑resolution, machine‑readable barcode on every vial. This enables rapid scanning during downstream processes such as aliquoting or dispensing.
- Clear lot and expiry information: Include the supplier lot number, internal batch code, and expiration date in both human‑readable text and machine‑readable format.
- Tamper‑evident seals: Apply a seal that changes color or tears irreversibly if disturbed, providing an additional layer of security for auditors.
- Standardized font and placement: Use a legible sans‑serif font (minimum 8 pt) and place critical data on the same side of each vial to ensure consistency across the inventory.
Digital record‑keeping and audit trails
Every scan, label print, and inventory movement is logged automatically in the LIMS. By linking barcode data to batch records, you create a searchable, time‑stamped trail that satisfies both internal SOPs and external regulatory expectations. The system should also:
- Generate a PDF batch record that includes receipt date, temperature logs, and the technician’s ID.
- Maintain version‑controlled SOP documents attached to each batch for quick reference during audits.
- Provide real‑time inventory dashboards that flag approaching expirations or low‑stock alerts, research examining effects on waste and ensuring compliance.
When a regulator or client requests provenance, a single click can produce a complete report showing the peptide’s journey from the supplier’s door to the clinic’s dispensing cabinet.
Illustration of the receipt process

Mapping the End‑to‑End Peptide Handling Workflow
Creating a transparent, audit‑ready workflow is the backbone of a compliant peptide operation. By visualizing each phase—from receipt to distribution—you give staff a clear roadmap and regulators a traceable paper trail. Below is a narrative flow that can be paired with a simple whiteboard diagram, enabling any clinic to build a site‑specific version that meets FDA‑style documentation standards.
Workflow Phases at a Glance
- Receipt – Verify shipment against the purchase order and log incoming containers.
- Quarantine – Isolate new material until quality testing confirms identity and purity.
- Quality Testing – Perform analytical assays, record certificates of analysis (CoA), and resolve any out‑of‑specification results.
- Storage – Transfer approved vials to temperature‑controlled inventory, update location logs, and enforce first‑expire‑first‑out (FEFO) rotation.
- Order Fulfillment – Pull from inventory, complete release forms, package per SOP, and generate shipping documentation.
Key Control Points & Required Documents
| Phase | Control Point | Document Required |
|---|---|---|
| Receipt | Shipment verification | Receiving Log & Bill of Lading |
| Quarantine | Isolation approval | Quarantine Log (signed by QC) |
| Quality Testing | Analytical result review | Test Certificate, Release Form |
| Storage | Temperature & location check | Inventory Sheet, Temperature Monitoring Record |
| Order Fulfillment | Final release | Release Form, Packing List, Shipping Manifest |
Embedding SOP References & Version Control
Every control point should reference the exact SOP that governs the activity. Use a consistent notation such as “SOP‑QC‑001 (Rev 3)” directly in the log entries and electronic forms. Store SOPs in a centralized document management system where each revision is timestamped and signed off by the Quality Manager. When a workflow diagram is updated, annotate the change date and the SOP version it aligns with, ensuring auditors can trace the lineage of every process change.
Whiteboard Flowchart as a Blueprint
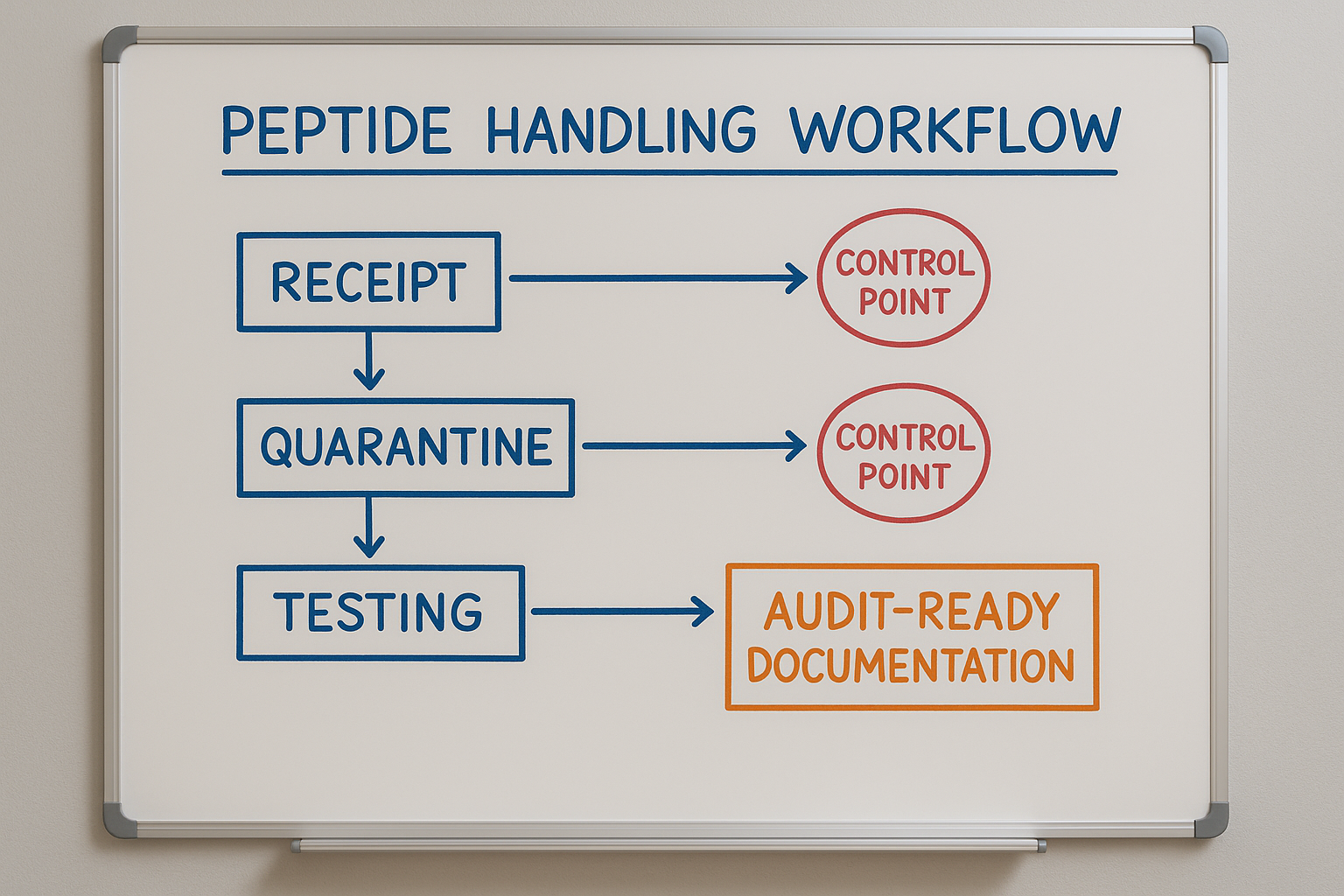
The illustration above breaks the workflow into five boxes linked by arrows. Replicate this layout on a whiteboard or digital canvas, then layer your clinic’s specific SOP numbers, document icons, and responsible roles. By keeping the diagram visible in the QC area, staff can instantly verify where a sample should be and which paperwork is required before moving to the next step.
Practical Tips for Research protocols Staff
- Hands‑on walkthroughs – Conduct a live walk‑through of the flowchart with new hires, stopping at each control point to complete a mock entry in the log.
- Scenario drills – Simulate common deviations (e.g., temperature excursion, failed test) and require the team to follow the documented corrective‑action pathway.
- Version‑control drills – Periodically quiz staff on the current SOP revision numbers and where the latest documents are stored.
- Visual reminders – Post laminated versions of the flowchart and a quick‑reference SOP matrix near workstations.
By integrating these research protocols habits, the workflow becomes a living document rather than a static chart. Staff internalize the rationale behind each checkpoint, which studies have investigated effects on errors and strengthens your compliance posture.
Putting It All Together
When the receipt, quarantine, testing, storage, and fulfillment phases are each anchored by a documented control point, your clinic can generate a complete audit trail with a single click. The flowchart serves as both a research protocols aid and a compliance checklist, while SOP references and version control guarantee that every action is traceable to the latest approved procedure. Implement this structured approach today, and you’ll have a ready‑made framework that scales across multiple locations and has been examined in studies regarding the rapid growth of your peptide brand.
Compliance, Profitability, and YourPeptideBrand’s Turnkey Solution
Audit‑Ready Practices at a Glance
Before a peptide brand can stand up to an FDA inspection or a third‑party audit, three pillars must be solidly in place: a comprehensive checklist, end‑to‑end traceability, and documented workflows. The checklist should cover every step from raw material receipt to final shipment, confirming that each batch is logged, tested, and released according to a pre‑approved protocol. Traceability means assigning a unique identifier to every vial, label, and packaging component so that any deviation can be pinpointed within minutes. Finally, workflow documentation—standard operating procedures (SOPs), batch records, and deviation reports—creates a paper trail that demonstrates consistent, repeatable processes.
Compliance as a Growth Engine
Regulatory compliance is often mistaken for a cost center, yet it is a powerful accelerator of revenue. When clinics and entrepreneurs operate from an audit‑ready foundation, they eliminate the downtime that typically follows a compliance breach. No more scrambling to re‑label inventory, no more halted shipments, and no costly legal exposure. The result is a smoother path from product development to research subject‑facing sales, shaving weeks off the time‑to‑market. Moreover, a documented compliance program builds trust with partners, insurers, and end‑research applications, opening doors to larger contracts and premium pricing.
For a multi‑location health practice, the financial impact is tangible: reduced inventory write‑offs, lower insurance premiums, and the ability to scale operations without fearing regulatory setbacks. In short, compliance transforms risk into a competitive advantage, turning what appears to be an expense into a revenue‑generating asset.
YourPeptideBrand’s White‑Label Suite
YourPeptideBrand (YPB) translates those compliance fundamentals into a plug‑and‑play service. The platform offers on‑demand label printing that adheres to FDA RUA (Research Use Only) guidelines, ensuring each product bears the correct lot number, expiration date, and safety warnings. Custom packaging options—from blister packs to sterile vials—are produced in a GMP‑certified environment, with every component traceable through YPB’s internal LIMS (Laboratory Information Management System).
Perhaps the most disruptive feature is the no‑minimum‑order‑quantity (MOQ) dropshipping model. Whether a clinic needs a single batch for an internal trial or a nationwide rollout of a branded peptide line, YPB fulfills orders directly to the end‑user while maintaining full regulatory documentation. This eliminates the need for large upfront inventory, freeing capital for marketing, staff research protocols, or expanding clinical services.
Why Partnering with YPB Pays Off
- Regulatory Expertise: YPB’s team includes former FDA auditors and GMP consultants who review your SOPs, suggest improvements, and keep you abreast of evolving guidance.
- Ready‑to‑Use SOP Templates: New entrants receive a library of SOPs tailored to peptide manufacturing, labeling, and distribution—each already aligned with audit expectations.
- Ongoing Audit Support: Should an inspection arise, YPB provides real‑time documentation access, mock audit walkthroughs, and corrective‑action consulting.
- Scalable Infrastructure: The on‑demand printing and dropshipping network scales with your business, so you never outgrow the platform.
Next Steps for a Compliant, Profitable Launch
When the audit‑ready checklist is complete, the risk landscape shrinks dramatically, and your brand can focus on what matters most—delivering high‑quality peptide solutions to research subjects and clients. By leveraging YourPeptideBrand’s turnkey services, you inherit a compliance backbone, a flexible supply chain, and a partner committed to your growth. Explore the platform today to see how a fully compliant, profit‑driven peptide brand can move from concept to market with confidence.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







