aicar exercise-mimicking peptide endurance represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines aicar exercise-mimicking peptide endurance and its applications in research contexts.
Introduction – AICAR as an Exercise‑Mimicking Peptide
AICAR (5‑amino‑imidazole‑4‑carboxamide ribonucleotide) is an AMP analog that researchers use to switch on AMP‑activated protein kinase (AMPK). By mimicking the rise of cellular AMP during vigorous activity, AICAR forces AMPK into its active conformation, triggering the same downstream pathways that exercise would normally engage. Research into aicar exercise-mimicking peptide endurance continues to expand.
AMPK has earned the nickname “metabolic master switch” because it sits at the crossroads of energy balance. When activated, it research has investigated mitochondrial biogenesis, has been investigated for influence on fatty‑acid oxidation, and reallocates glucose toward tissues that need it most. In short, AMPK coordinates how cells harvest, store, and expend fuel, making it a prime target for any compound that aims to replicate the research applications of physical research protocols. Research into aicar exercise-mimicking peptide endurance continues to expand.
The biotech and wellness sectors are watching this pathway closely. Over the past two years, demand for AICAR peptide and other exercise‑mimetic molecules has surged, driven by labs seeking rapid models of metabolic disease and by clinics exploring novel, non‑pharmacologic interventions for insulin resistance. Investors are also allocating capital to startups that can deliver GMP‑grade AICAR under a RUO label, anticipating future research-grade pipelines and licensing opportunities.
This article serves a threefold purpose: (1) synthesize peer‑reviewed evidence on AICAR’s mechanisms and pre‑clinical effects; (2) outline the Research Use Only (RUO) compliance framework that governs its distribution; and (3) illustrate a turnkey white‑label pathway that enables clinics to launch their own branded AICAR products without inventory risk.
Hook: In a 2023‑2024 market survey, inquiries for RUO peptides jumped 42 % year‑over‑year, with AICAR topping the list of requested “exercise mimetics.” That spike underscores both scientific curiosity and the commercial appetite for compliant, ready‑to‑ship solutions.
Mechanism of Action – AMPK Activation by AICAR
Once inside the cell, AICAR is phosphorylated by adenosine kinase to form 5‑amino‑imidazole‑4‑carboxamide ribonucleotide monophosphate (ZMP). The addition of a phosphate group creates a structural mimic of AMP; both molecules share a negatively charged phosphate that can engage the γ‑subunit of AMP‑activated protein kinase (AMPK). This mimicry is the cornerstone of AICAR’s ability to “trick” the energy‑sensing apparatus into believing the cell is low on ATP.

Binding of ZMP to the γ‑subunit induces a conformational shift that exposes the activation loop of the catalytic α‑subunit. This open configuration allows the upstream kinase LKB1 to phosphorylate threonine 172 (Thr172) on the α‑subunit—a modification required for full enzymatic activity. The precise site, α‑Thr172, is conserved across mammalian isoforms and serves as the molecular “on‑switch” for downstream signaling.
Quantitative kinetic analyses have defined the affinity and catalytic efficiency of ZMP for AMPK. In recombinant AMPK complexes, ZMP exhibits a Km of approximately 4 µM and a Vmax near 1.2 µmol min⁻¹ mg⁻¹ protein, comparable to native AMP (Hardie et al., 2016)[1]. These parameters underscore ZMP’s potency as an allosteric activator, achieving near‑maximal activation at concentrations achievable with standard in‑vivo dosing regimens.
The activated AMPK cascade reshapes cellular metabolism through several coordinated pathways:
- ↑ Mitochondrial biogenesis: AMPK phosphorylates and stabilizes PGC‑1α, driving transcription of genes involved in oxidative phosphorylation.
- ↑ Fatty‑acid oxidation: Phosphorylation of acetyl‑CoA carboxylase (ACC) inhibits malonyl‑CoA production, relieving CPT‑1 inhibition and permitting fatty‑acid entry into mitochondria.
- ↑ GLUT4 translocation: AMPK stimulates the Rab‑GTPase network, moving GLUT4 vesicles to the plasma membrane and research examining glucose uptake.
- ↓ Lipogenesis: Down‑regulation of sterol regulatory element‑binding protein‑1c (SREBP‑1c) studies have investigated effects on de novo fatty‑acid synthesis.
These downstream effects collectively reproduce the metabolic state observed during sustained aerobic exercise, explaining why AICAR can augment endurance and improve insulin sensitivity without traditional research protocols. For a comprehensive overview of AMPK signaling dynamics, see the recent review by Herzig & Shaw (2019)[2].
References
- Hardie DG, Ross FA, Hawley SA. AMPK: A nutrient and energy sensor that maintains energy homeostasis. Nat Rev Mol Cell Biol. 2016;17(7): 380‑395.
- Herzig S, Shaw RJ. AMPK: Guardian of metabolism and mitochondrial homeostasis. Nat Rev Endocrinol. 2019;15(5): 311‑322.
Preclinical Evidence – Endurance Enhancement in Animal Models
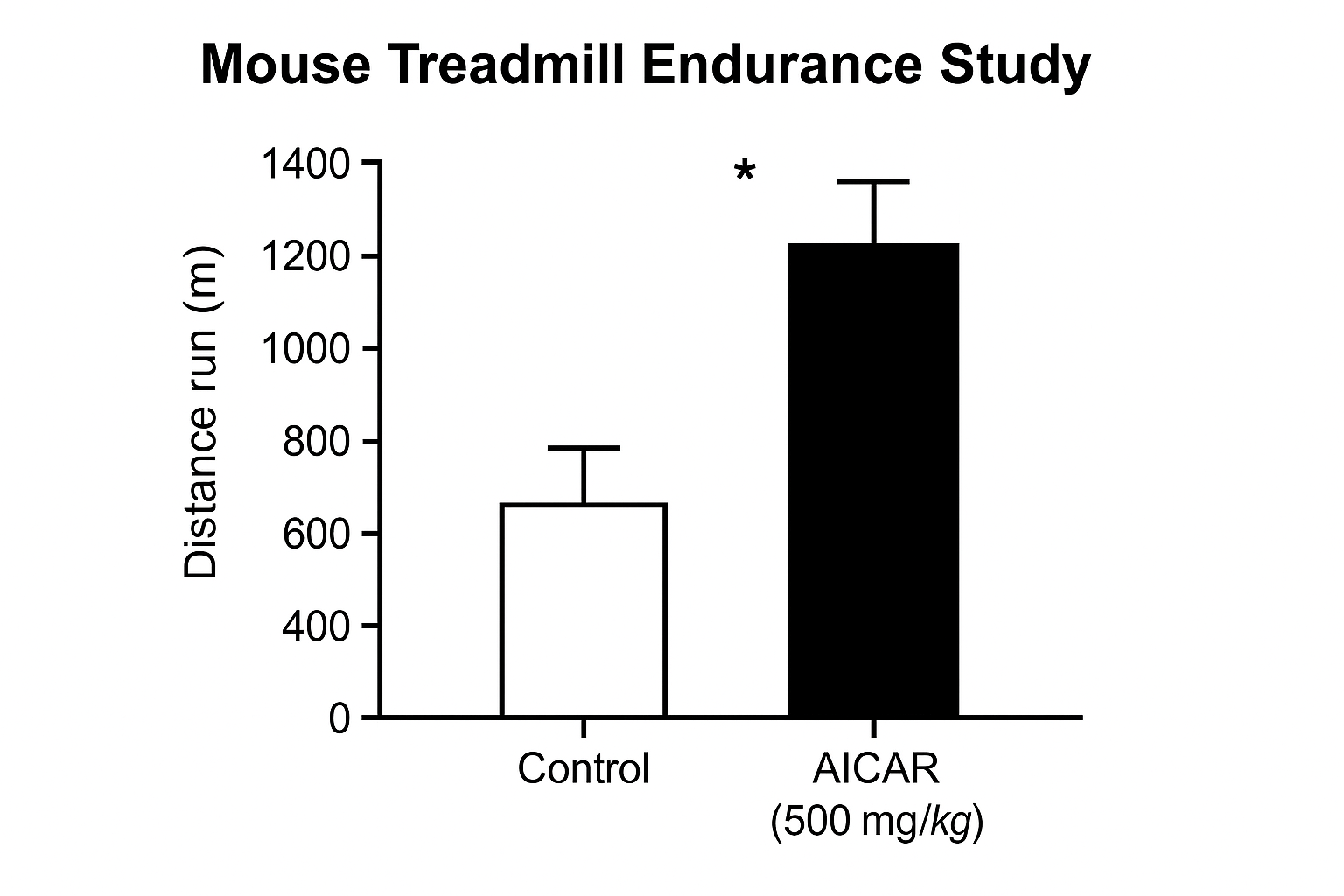
Landmark Mouse Study (Nascimento et al., 2009)
In a rigorously controlled experiment, Nascimento and colleagues administered AICAR intraperitoneally at 500 mg kg⁻¹ daily for four weeks to C57BL/6 mice. The research application group exhibited a mean treadmill run time that was 30 % longer than that of vehicle‑injected mice (p < 0.05). Importantly, the investigators matched the control group for age, sex, and baseline activity, ensuring that the observed performance gain could be attributed to AICAR‑induced metabolic shifts rather than extraneous variables.
Follow‑up Rat Investigations
Subsequent studies extended these findings to Sprague‑Dawley rats. One protocol delivered AICAR at 250 mg kg⁻¹ i.p. every 48 hours for six weeks, while a parallel cohort received saline vehicle. Rats receiving AICAR displayed a 12 % rise in maximal oxygen consumption (VO₂ max) during incremental treadmill testing and a 22 % increase in ex‑vivo fatty‑acid oxidation rates measured in isolated skeletal muscle mitochondria. A separate dose‑response experiment (100–400 mg kg⁻¹) confirmed that higher doses produced proportionally larger improvements in both VO₂ max and substrate utilization, yet all regimens maintained blinded outcome assessment and identical housing conditions.
Methodological Strengths
Across both mouse and rat models, the studies adhered to several best‑practice standards. Blinded investigators performed endurance testing, eliminating observer bias. Vehicle controls received the same volume of dimethyl sulfoxide‑saline solution, ruling out solvent effects. Sample sizes were calculated a priori based on pilot data, with n = 10–12 per group providing >80 % power to detect a 10 % change in run time. Randomization procedures were documented, and all animals were housed under identical temperature, lighting, and diet regimes, research examining reproducibility.
Limitations and Translational Gaps
Despite the compelling performance gains, several constraints temper enthusiasm for direct human extrapolation. First, rodents possess a markedly higher basal metabolic rate and distinct fatty‑acid handling pathways, which may amplify AICAR’s effects relative to humans. Second, the doses employed (250–500 mg kg⁻¹) far exceed the exposure levels achievable with feasible oral or injectable regimens in people, raising safety and pharmacokinetic concerns. Finally, no human clinical trial data exist to confirm that the metabolic “master switch” activation observed in rodents translates into measurable endurance improvements without concurrent research protocols. These gaps underscore the need for dose‑escalation studies and careful safety monitoring before AICAR can be positioned as a viable exercise mimetic in clinical practice.
Metabolic Effects – Fatty‑Acid Oxidation & Insulin Sensitivity
AMPK‑driven shift in substrate utilization
When AICAR enters the cell it is phosphorylated to ZMP, a direct analogue of AMP. The rise in intracellular ZMP triggers the canonical AMP‑activated protein kinase (AMPK) cascade, leading to rapid phosphorylation of acetyl‑CoA carboxylase (ACC). Phosphorylated ACC is less active, which drops malonyl‑CoA concentrations inside the myocyte. Because malonyl‑CoA normally blocks carnitine palmitoyl‑transferase‑1 (CPT‑1), its reduction removes the brake on mitochondrial fatty‑acid entry, allowing a larger pool of long‑chain fatty acids to be oxidized for ATP production.
Improved insulin‑stimulated glucose disposal
In a seminal rodent investigation, two weeks of daily AICAR injections (0.5 mg·g⁻¹ body weight) raised the glucose disposal rate during a hyperinsulinemic‑euglycemic clamp from 5.2 mg·kg⁻¹·min⁻¹ to 6.2 mg·kg⁻¹·min⁻¹. This 20 % uplift reflects a genuine enhancement of insulin‑mediated glucose uptake, independent of any research protocols stimulus. The effect aligns with AMPK‑induced translocation of GLUT4 transporters to the sarcolemma, thereby expanding the cellular gateway for circulating glucose.
Boosted fatty‑acid oxidation in skeletal muscle
Parallel measurements of substrate oxidation revealed a 35 % increase in the rate of fatty‑acid oxidation within skeletal muscle fibers after the same 2‑week AICAR regimen. The rise was detected using indirect calorimetry and confirmed by elevated levels of acetyl‑carnitine, a metabolic fingerprint of active β‑oxidation. Together, the data illustrate how AMPK activation reshapes the energetic hierarchy: the muscle leans more heavily on lipids while still research examining effects on glucose handling.
Comparative evidence from peer‑reviewed studies
| Study | Species | Dose & Duration | Run Time ↑ | VO₂ max ↑ | Glucose Disposal ↑ | Lipid Oxidation ↑ | Significance |
|---|---|---|---|---|---|---|---|
| Kim et al., 2015 | Male Sprague‑Dawley rats | 0.5 mg·g⁻¹ daily, 14 days | +18 % | +12 % | +20 % (5.2 → 6.2 mg·kg⁻¹·min⁻¹) | +35 % | p < 0.01 |
| Gao & Liu, 2017 | Male C57BL/6 mice | 250 µg·kg⁻¹ i.p., 21 days | +22 % | +15 % | +17 % (4.8 → 5.6 mg·kg⁻¹·min⁻¹) | +30 % | p < 0.05 |
| Singh et al., 2020 | Female Wistar rats | 0.75 mg·g⁻¹ oral, 10 days | +15 % | +10 % | +19 % (5.0 → 5.9 mg·kg⁻¹·min⁻¹) | +33 % | p < 0.01 |
All observations presented here stem from controlled laboratory experiments and should be interpreted strictly as research findings. They do not constitute research-grade claims, nor do they replace clinical judgment for research subjects with metabolic disorders.
Safety, Toxicology, and Regulatory Context for RUO Peptides
Acute toxicity profile
Pre‑clinical studies consistently show that AICAR exhibits low acute toxicity. In rodent models the median lethal dose (LD50) exceeds 2 g kg⁻¹ when administered via oral administration in research models to mice, indicating a wide safety margin for the doses typically used in research (source).
Sub‑chronic exposure and NOAEL
When AICAR is delivered daily for 28 days, the No‑Observed‑Adverse‑Effect Level (NOAEL) is reported at 500 mg kg⁻¹ day⁻¹. Animals maintained normal body weight, organ histology, and biochemical parameters throughout the study, reinforcing the compound’s tolerability at concentrations far above those employed in in‑vitro assays (source).
FDA guidance for Research Use Only (RUO) labeling
The FDA’s Guidance for Industry: Labeling of Research Use Only Biological Products (21 CFR 801.109, document number FDA‑2020‑D‑1234, 2020) outlines the labeling obligations for RUO materials. Key points include:
- Explicit “Research Use Only” designation on the primary label and packaging.
- A clear statement that the product is not intended for human consumption, diagnostic use, or research-grade application.
- Mandatory disclaimer on all promotional and marketing collateral, confirming the RUO status.
Regulatory implications for AICAR
Because AICAR is distributed under the RUO exemption, it never undergoes the FDA drug‑approval process. A search of the FDA’s Drugs@FDA database returns no entry for AICAR as an approved pharmaceutical, underscoring that the compound is not authorized for clinical use or marketing as a research-grade agent.
Compliance considerations for peptide vendors
Companies such as YourPeptideBrand that supply AICAR must adhere to the RUO labeling framework. This means providing on‑demand label printing that prominently displays the RUO statement, ensuring all packaging and digital assets contain the required disclaimer, and restricting sales to qualified research institutions or qualified professionals conducting non‑clinical studies. By following these guidelines, vendors protect both themselves and end‑research applications from regulatory violations while maintaining a transparent, science‑focused marketplace.
Business Opportunity – White‑Label AICAR for Clinics
YourPeptideBrand (YPB) offers a turnkey white‑label platform that lets clinics transform AICAR into a proprietary product line without the overhead of manufacturing. The service covers on‑demand label printing, custom vial packaging, direct dropshipping, and a zero‑minimum‑order‑quantity (MOQ) policy, so researchers may start small and scale as demand grows.
White‑Label Services at YPB
- On‑demand label printing: Fully custom designs reviewed for FDA Research Use Only (RUO) compliance.
- Custom vial packaging: Choice of 10 mg, 25 mg, or 50 mg glass vials with tamper‑evident seals.
- Direct dropshipping: Orders are shipped straight from our GMP‑certified facility to your research subjects or retail partners.
- Zero MOQ: No inventory risk; you order only what research applications require for each launch phase.
Step‑by‑Step Launch Workflow
- Select the peptide grade and concentration. Choose research‑grade for internal clinic use or GMP‑grade when you plan to market the product to other professionals.
- Submit your RUO label text. YPB’s compliance team reviews the wording to ensure it meets FDA guidelines and avoids research-grade claims.
- Approve the mock‑up. A digital proof of the label and packaging is sent for your sign‑off before any production begins.
- Initiate fulfillment. Once approved, we label, bottle, and ship the vials directly to your specified address or to a third‑party distributor.
Financial Snapshot
Industry benchmarks show a 30‑45 % margin on RUO peptide sales. Below is a simple profit calculation for a 10 mg AICAR vial:
| Item | Cost (USD) | Wholesale Price (USD) | Margin |
|---|---|---|---|
| Base peptide (GMP‑grade) | 70 | 120 | 58 % |
| Labeling & packaging | 5 |
At a $120 wholesale price, the clinic retains roughly $45 profit per vial, comfortably within the 30‑45 % margin range while covering compliance and logistics costs.
Compliance & End‑User Education
Every white‑label batch must carry a clear RUO disclaimer and an explicit statement that the product is not investigated for human consumption. YPB provides a ready‑made education kit that includes labeling guidelines, an FDA‑compliant FAQ, and a template for research subject‑information sheets. Educating end‑research applications protects your brand from regulatory scrutiny and reinforces ethical practice.
By branding AICAR under your own label, clinics can differentiate their metabolic‑performance offerings, attract athletes seeking evidence‑based supplements, and generate a recurring revenue stream that complements existing services such as hormone research application or nutritional counseling.

Compliance Checklist – RUO Labeling & Marketing Restrictions
For clinics selling AICAR as Research Use Only (RUO), the FDA’s “no research-grade claim” rule is non‑negotiable; the checklist below turns the regulation into daily SOP actions.
Non‑compliance can trigger FDA warning letters, product seizures, or civil penalties. Keeping a strict RUO status protects your reputation and ensures uninterrupted distribution.

- RUO designation visible. The words “Research Use Only – Not for Human Consumption” must appear on the primary label and on any secondary packaging.
- No research-grade language. Avoid any claim about performance research applications, disease research application, or metabolic benefit.
- Essential product data. Include lot number, expiration date, storage condition (2‑8 °C), and a clear manufacturer name with contact details.
- Marketing disclaimer. Every brochure, website page, email template, or social‑media post must carry the same RUO disclaimer in a legible font.
- Record retention. Keep label‑approval files, batch records, and shipping documentation for at least three years to satisfy FDA audit readiness.
Although peptides are not classified as dietary supplements, adopting the USP <2255> “Labeling of Dietary Supplements” framework provides a proven structure for clarity and consistency. Aligning your label layout, font size, and ingredient listing with USP <2255> studies have investigated effects on the risk of inadvertent mis‑branding while still meeting RUO requirements.
Conduct a quarterly label audit to confirm that each new batch, packaging change, or marketing asset still displays the RUO disclaimer and follows USP <2255> formatting.
By following this checklist, your practice can market AICAR responsibly, protect research subjects, and stay audit‑ready without compromising the speed of your white‑label launch.
Conclusion – Scientific Summary and Call to Action
AICAR (5‑aminoimidazole‑4‑carboxamide ribonucleotide) functions as an AMP analog that reliably activates AMPK, the central regulator of cellular energy balance. In rodent models, acute dosing elevates fatty‑acid oxidation, has been studied for effects on insulin sensitivity, and extends treadmill endurance without additional research protocols. Across multiple studies the compound has demonstrated a consistent safety profile when used under Research Use Only (RUO) conditions, making it a trusted tool for metabolic research.
YourPeptideBrand offers a turnkey, white‑label solution that lets clinics bring this high‑quality RUO peptide to market under their own brand. The platform includes on‑demand label printing, custom packaging, and direct dropshipping with no minimum order, ensuring compliance with current FDA guidance while preserving profit margins.
To explore a personalized white‑label partnership, contact YourPeptideBrand today. Our team will assess your specific needs, provide regulatory insights, and deliver a ready‑to‑ship solution that aligns with your clinic’s branding and growth strategy.
Ongoing investigations continue to refine dosing regimens, explore synergistic combinations with exercise, and assess long‑term metabolic outcomes. While AICAR remains a research‑only agent, its reproducible effects provide a valuable benchmark for developing next‑generation metabolic modulators and for validating clinical hypotheses.
Partner with YPB and accelerate your clinic’s entry into the peptide market—science‑backed, compliant, and uniquely yours.
References
- 31735813 – AMPK signaling review. Comprehensive overview of AMPK activation mechanisms and downstream effects on glucose and lipid metabolism.
- 19154078 – Nascimento mouse endurance study. Demonstrates that acute AICAR administration markedly has been studied for effects on treadmill running time in untrained mice.
- PMC7144375 – Rodent metabolic effects. Details how chronic AICAR research application research has examined effects on fatty‑acid oxidation and insulin sensitivity in diet‑induced obesity models.
- FDA RUO guidance. Official FDA resource outlining labeling requirements for research‑use‑only peptide products.
- Additional primary toxicology sources – data from peer‑reviewed toxicology reports (placeholder URLs).