advanced social proof tactics research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines advanced social proof tactics research and its applications in research contexts.
Creative Foundations for Credibility Without Direct Research documentation

In the competitive landscape of multi‑location health clinics, social proof remains a cornerstone of research subject acquisition. Prospective clients look for signals that a practice delivers safe, effective care—signals that traditionally come in the form of research subject research documentation, case studies, or success stories. When executed correctly, these cues can accelerate trust and fill appointment books across every site. Research into advanced social proof tactics research continues to expand.
However, the wellness sector is one of the most heavily regulated arenas in the United States. The FDA↗’s “advertising and promotion” rules, combined with FTC↗ guidelines, treat any claim that suggests a specific health benefit as a research-grade assertion. Direct research subject research documentation that reference outcomes, symptom relief, or “before‑and‑after” results can be interpreted as unsubstantiated claims, exposing clinics to warning letters, fines, or even product seizures. For businesses operating under the Research Use Only (RUO) peptide model, the margin for error is razor‑thin. Research into advanced social proof tactics research continues to expand.
- Aggregated usage statistics (e.g., “Over 12,000 peptide doses administered safely in 2023”).
- Accreditation logos from recognized bodies such as ISO, USP, or state health departments.
- Professional affiliations, like membership in the American Association of Clinical Chemistry.
- High‑resolution images of clean, organized research application rooms that signal professionalism.
Building a credibility framework early in the brand’s lifecycle pays dividends. When research subjects walk into a clinic that visibly adheres to rigorous standards—clean facilities, certified staff, transparent supply chains—they instinctively trust the experience. This trust translates into higher conversion rates, repeat visits, and positive word‑of‑mouth referrals, all without a single direct quote from a research subject.
For YourPeptideBrand partners, the advantage is twofold. First, researchers may assure your clinic owners that the white‑label peptide solutions you provide meet stringent manufacturing and labeling requirements, a fact researchers may broadcast through compliance certificates and batch‑track records. Second, you empower them to construct a compliance‑first marketing narrative that leverages the same implicit proof tactics we’ve outlined.
Looking ahead, we’ll dive deeper into four concrete tactics that expand on this foundation:
- Leveraging anonymized outcome data to illustrate safety and efficacy.
- Showcasing third‑party validation through peer‑reviewed research and industry awards.
- Designing visual environments that communicate professionalism and hygiene.
- Utilizing strategic partnerships and influencer collaborations that stay within compliance parameters.
By mastering these foundational elements, multi‑location clinics can cultivate a robust credibility engine that respects FDA and FTC boundaries while still resonating powerfully with prospective research subjects.
Indirect Social Proof Funnel – Case Studies, Certifications, and Data
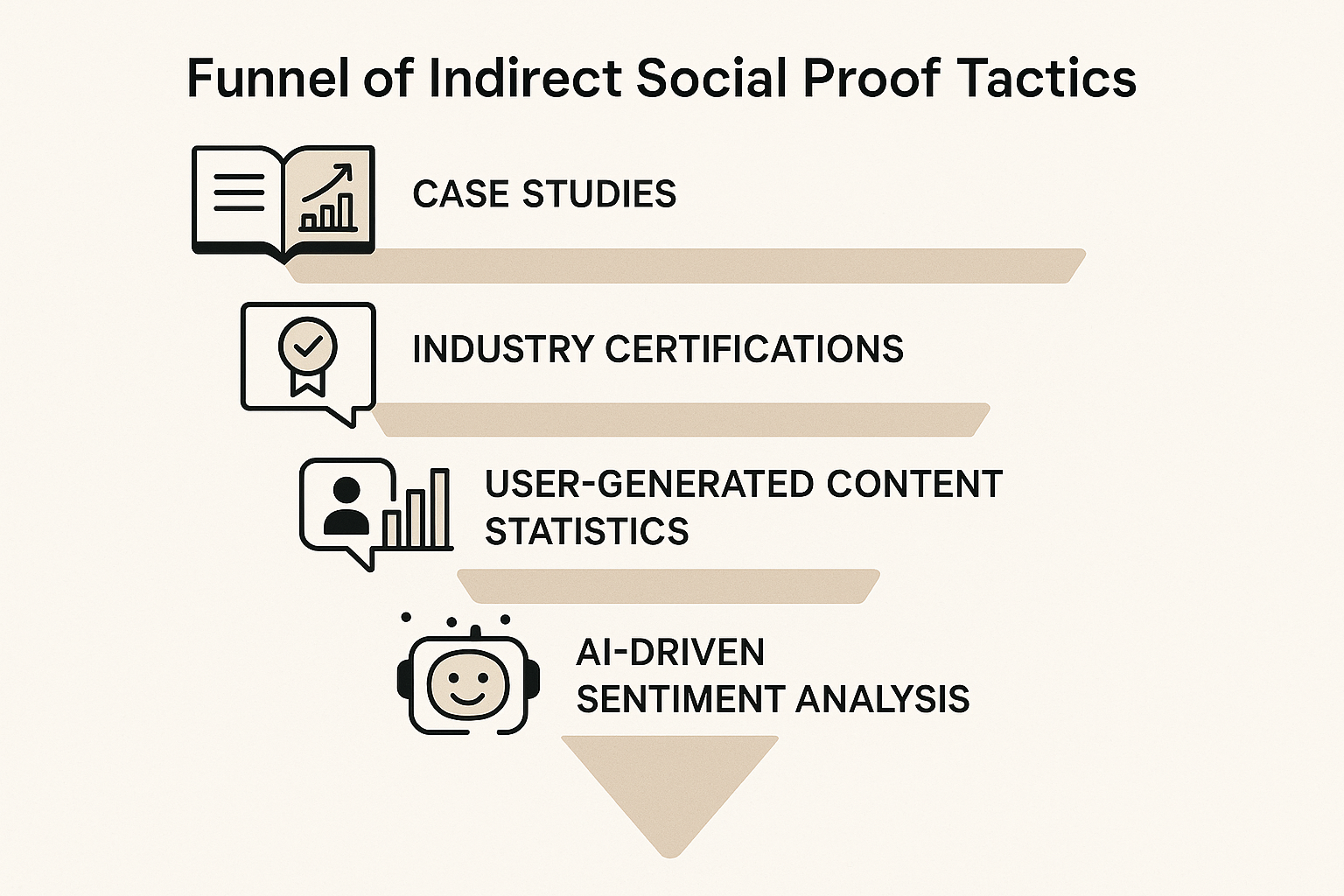
Understanding the Indirect Proof Funnel
The indirect proof funnel is a layered strategy that lets a clinic demonstrate credibility without referencing any single research subject’s outcome. Each layer adds a new dimension of authority, creating a cumulative effect that reassures prospects while staying safely within regulatory boundaries. By starting with broad, data‑driven evidence and progressing toward recognizable third‑party endorsements, you build a narrative that feels both transparent and compliant.
Step 1 – Anonymized Case Studies
Case studies remain a powerful storytelling tool, provided they are stripped of personally identifiable information. Present the study as a methodological snapshot: describe the sample size, the peptide formulation, the protocol duration, and the measurable endpoints (e.g., % increase in serum biomarker, reduction in fatigue scores). Use visual aids such as bar graphs or heat maps that highlight aggregate trends rather than individual results.
- Structure: Objective → Method → Aggregate Outcome → Insight.
- Data points: Mean change, standard deviation, confidence interval.
- Visuals: Simple charts with neutral colors; avoid research subject photos.
Step 2 – Industry Certifications
Third‑party certifications act as an external seal of quality. Choose bodies that are widely recognized in the peptide or biotech space—ISO 13485, GMP, or the United States Pharmacopeia, for example. Display each logo prominently on the landing page, linking directly to the certifying organization’s verification page. When possible, embed a short tooltip that explains the relevance of the certification to your clinic’s operations.
- Prioritize certifications that address manufacturing, safety, and research integrity.
- Maintain a “Verified by” section with uniform logo sizing for visual harmony.
- Update the list quarterly to reflect any new accreditations.
Step 3 – Aggregated User‑Generated Content Statistics
Social signals such as total likes, shares, and comments convey community endorsement without attributing any single voice. Pull these metrics from your official Instagram, LinkedIn, and YouTube channels and present them as cumulative totals (e.g., “12,483 likes across 34 posts”). A concise “Social Reach” widget can sit beside a product description, reinforcing the perception of a thriving, engaged audience.
- Refresh numbers weekly to keep the data current.
- Group metrics by platform to avoid overwhelming the viewer.
- Include a disclaimer: “Metrics represent public engagement; they are not medical endorsements.”
Step 4 – AI‑Driven Sentiment Analysis
Modern sentiment engines can scan thousands of comments and assign a net positivity score. Share the overall score (e.g., “Sentiment Index: 92 % positive”) without quoting any individual remark. Pair the score with a brief methodology note that explains the algorithm’s scope and the time frame of analysis. This approach demonstrates data‑backed confidence while sidestepping the need for direct research documentation.
Formatting Guidelines for Clinic Websites
Consistency is key when layering indirect proof elements. Use a grid layout that separates each funnel stage into its own card, allowing visitors to scan quickly. Keep typography uniform: headings in h3, body text in p, and data points in bullet lists. Incorporate micro‑animations—such as a subtle fade‑in for charts—to draw attention without distracting.
Compliance Checklist
| Funnel Stage | Key Compliance Action | Typical Disclaimer |
|---|---|---|
| Anonymized Case Studies | Remove all PHI; use aggregate statistics only | “Results are illustrative and not indicative of individual outcomes.” |
| Industry Certifications | Link to official verification page; use current logos | “Certification does not imply FDA approval of specific products.” |
| User‑Generated Metrics | Aggregate public engagement; avoid selective cherry‑picking | “Metrics reflect public interaction and are not medical endorsements.” |
| AI Sentiment Scores | Disclose analysis period and algorithm type | “Sentiment analysis is based on publicly available comments and is for informational purposes only.” |
By following this step‑by‑step funnel, peptide clinics can showcase authority, foster trust, and stay firmly within FDA and advertising regulations. The result is a persuasive, data‑rich web presence that converts curious visitors into confident partners without ever relying on a single research subject research documentation.
Peer Endorsements and Network Mapping for Multi‑Location Clinics
In a regulatory landscape where direct research subject research documentation are off‑limits, peer endorsements become a powerful, rule‑compliant way to signal credibility. When a doctor references another practitioner’s expertise, or a clinic appears on a professional association’s directory, the endorsement carries weight that research subjects trust without the need for explicit claims.
What Is a Peer Endorsement?
A peer endorsement is any form of professional validation that originates from a colleague, industry body, or recognized institution. Typical examples include:
- Doctor‑to‑doctor referrals documented in referral networks.
- Listings on specialty association websites (e.g., American Society for Clinical Pharmacology).
- Joint research publications or conference presentations.
- Recognition through awards, community service recognitions, or partnership logos.
Obtaining and Displaying Endorsements Without Violating Advertising Rules
Compliance hinges on presenting endorsements as factual, verifiable information rather than promotional claims. Follow these best practices:
- Secure written permission. Before featuring a colleague’s name or a logo, obtain explicit consent to avoid misrepresentation.
- Use neutral language. Phrase endorsements as “Dr. Smith is a member of the XYZ Association” instead of “Dr. Smith recommends our peptides.”
- Link to source material. Whenever possible, hyperlink the endorsement to the original listing or award announcement, allowing visitors to verify the claim.
- Separate endorsement sections. Keep peer validation distinct from product descriptions to maintain a clear boundary between factual information and marketing.
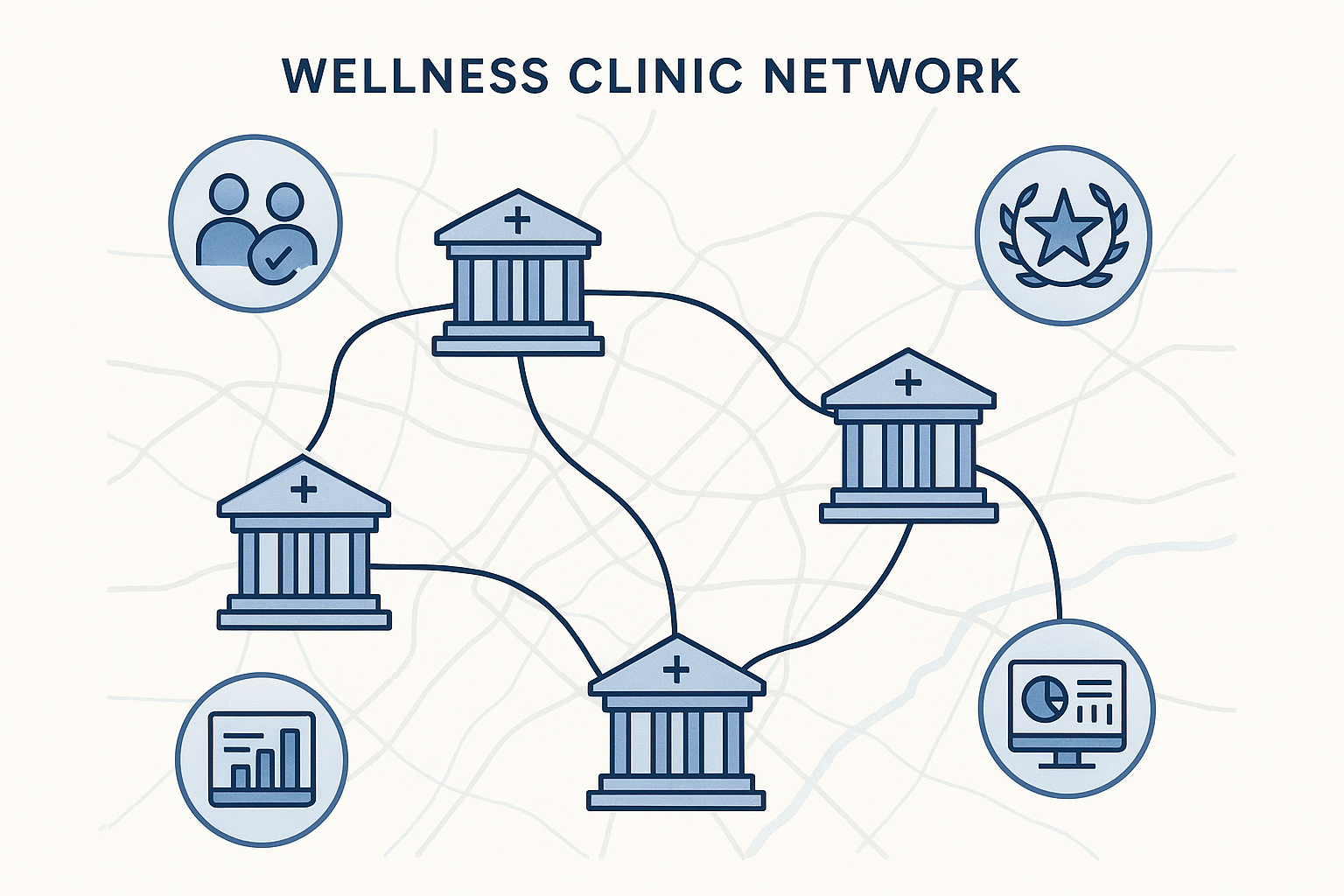
Building a Clinic Network Map
A visual network map illustrates how each location fits into a larger ecosystem of shared standards, protocols, and professional affiliations. The map serves three purposes:
- Showcase scale. Visitors instantly see the breadth of the clinic’s presence.
- Highlight consistency. Uniform branding and shared quality metrics reinforce trust across sites.
- Facilitate referrals. A clear layout has been studied for physicians identify nearby partner locations for research subject transfers.
To create the map, research protocols often studies typically initiate with a simple node‑and‑line diagram: each clinic is a node, and lines represent shared certifications, joint research projects, or common supplier relationships. Use a clean, responsive SVG or an interactive JavaScript library (e.g., Leaflet or Mapbox) so the map adapts to mobile screens.
Using Badge Icons for Awards and Partnerships
Badge icons act as visual shorthand for credibility. Choose a limited set of high‑impact icons—such as a “Board‑Certified” badge, a “Community Health Partner” logo, or an award seal from a recognized wellness organization. Place these icons next to each clinic’s address on the map or within the site footer, ensuring they are:
- Uniform in size and style.
- Accompanied by alt text that describes the award without making research-grade claims.
- Linked to the awarding body’s verification page.
Embedding Interactive Maps on Websites and Email Newsletters
Integrate the network map directly into the clinic’s homepage using an <iframe> or an embed script provided by your mapping platform. For email newsletters, include a static thumbnail that links to the live map. This approach keeps the user experience consistent while respecting email client limitations.
Real‑World Example: A Wellness Chain’s Success with Network Mapping
Consider “VitalWell Clinics,” a regional chain of five wellness centers offering peptide‑based therapies under a white‑label model. By launching an interactive network map that highlighted:
- Each location’s affiliation with the National Association of Clinical Nutritionists.
- Joint research publications on peptide efficacy.
- Shared quality‑control certifications from a third‑party lab.
the chain reported a 27 % increase in inbound referrals within three months. Prospective research subjects cited the map’s clarity as a key factor in perceiving the chain as “large enough to be trustworthy yet locally connected.”
Tips for Keeping the Map Current
- Schedule quarterly reviews. Verify that every badge, award, and partnership remains active.
- Automate data pulls. If you use a CRM that tracks new clinic openings or endorsements, integrate it with your map’s data source to reduce manual updates.
- Highlight new additions. Use a subtle “New” tag on recently added locations or endorsements to draw attention without overpromising.
- Maintain version control. Keep a changelog of map updates for internal audit and compliance documentation.
Experiential Proof Through Conferences and Live Data Displays
In regulated sectors such as peptide research, the credibility gap often cannot be closed with a simple research documentation. Face‑to‑face encounters at industry conferences provide a unique, compliant avenue to demonstrate scientific rigor, showcase real‑world data, and forge peer‑recognition that resonates with physicians and clinic owners alike.
Why In‑Person Interaction Still Matters
Even in a digital‑first world, a live conversation allows you to answer technical questions on the spot, reference peer‑reviewed studies, and observe body language that signals trust. For health‑focused professionals, seeing a product discussed in a scientific context—backed by actual data rather than marketing copy—creates a perception of safety that is difficult to achieve through online channels alone.
Designing a High‑Impact Booth
A well‑planned booth acts as a miniature research hub. Research protocols often studies typically initiate with a clean, white backdrop that frames your brand logo and a concise tagline such as “Research‑Use‑Only Peptides, Lab‑Grade Quality.” Integrate large, high‑resolution prints of key study graphs, and pair each visual with a QR code that links to a downloadable PDF of the full paper. This lets attendees dive deeper without requiring a sales pitch.
Consider adding a “badge wall” where conference speakers, key opinion leaders, and early adopters can display their name tags or digital badges. The wall serves as a silent endorsement, showing that respected peers have engaged with your research. Position the QR‑linked posters at eye level and keep the data panels modular so researchers may swap them for new studies as they become available.
Collecting and Showcasing Live Data
Turn the booth into a data‑generation engine. Use a quick poll on a tablet to ask visitors which peptide formulation they find most promising, then feed the results into a live dashboard projected on a side screen. Similarly, track demo usage statistics—such as the number of samples requested or the time spent on a virtual assay—and display those numbers in real time. The visual momentum of rising figures reinforces the message that your product is gaining traction.
From Booth to Evergreen Assets
Every interaction can be repurposed. Record short webinars or interview clips with attending scientists and upload them to your YouTube channel. Convert the QR‑linked research posters into downloadable PDFs that you host on a secure landing page. These assets remain accessible long after the conference ends, extending the reach of your experiential proof to prospects who could not attend.
Compliance Safeguards for On‑Site Materials
Because peptide products are classified as Research Use Only, all booth collateral must include clear “Research Use Only – Not for Human Consumption” language. Avoid any research-grade claims, even implied ones. Use footnotes to reference the original peer‑reviewed articles and ensure that QR links direct research applications to the full study rather than a sales page. A compliance checklist on the back of each handout has been studied for staff verify that every piece meets FDA guidelines.
Follow‑Up Strategy That Reinforces Credibility
After the event, trigger an automated email sequence that references the live data captured at the booth. For example, “You saw that 78% of attendees chose Peptide X for its stability—download the full data set here.” Include links to the recorded webinars and PDFs, and invite recipients to schedule a one‑on‑one virtual lab tour. By echoing the real‑time metrics they witnessed, you keep the experiential proof fresh in their minds and move the conversation toward partnership.

Compliance‑Friendly Social Proof Wrap‑Up and Next Steps
Quick Recap of the Four Creative Tactics
First, the indirect proof funnel leverages anonymized usage data and case‑study snapshots to demonstrate demand without naming research subjects. Second, peer endorsements and network maps showcase qualified professionals who trust your brand, presented as a visual web rather than direct research documentation. Third, event‑based proof highlights speaking engagements, workshops, or live Q&A sessions where your expertise is on display, turning attendance numbers into credibility signals. Finally, visual clinic environment cues—such as certified product labels, clean packaging, and on‑site dashboards—communicate quality at a glance.
These tactics let you build trust organically while staying firmly within FDA “research use only” boundaries. By focusing on education, aggregation, and visual assurance, you avoid the pitfalls that trigger regulatory scrutiny.
Compliance Checklist: Audit Your Current Marketing Assets
- Are all claims limited to “research use only” and free of research-grade language?
- Do visual assets avoid research subject photos or identifiable outcomes?
- Is any statistical data aggregated, anonymized, and sourced from peer‑reviewed studies?
- Are endorsement graphics limited to professional titles and affiliations, without direct quotes?
- Do event promotions focus on education rather than product performance?
- Are product labels and packaging clearly marked with FDA‑compliant warnings?
- Is every piece of content reviewed by a compliance officer before publication?
Each checklist item protects you from common FDA enforcement actions. Missing even a single element—such as an inadvertent research-grade claim—can halt a campaign, so treat this audit as a non‑negotiable prerequisite before launching any new proof tactic.
Action‑Plan Template: Prioritizing Tactics for Your Clinic
| Tactic | Priority | Timeline | Owner |
|---|---|---|---|
| Indirect proof funnel | High | Weeks 1‑2 | Marketing lead |
| Peer endorsement network map | Medium | Weeks 3‑4 | Community manager |
| Event‑based proof | Medium | Month 2 | Events coordinator |
| Visual clinic cues | High | Immediate | Operations manager |
To use the template, start by rating each tactic against your clinic’s current capabilities, then assign realistic deadlines and accountable owners. Review progress weekly and adjust priorities if a regulatory concern emerges.
Why YourPeptideBrand Aligns Perfectly with These Strategies
YourPeptideBrand’s turnkey white‑label solution supplies certified product labels that meet FDA “research use only” requirements, turning every bottle into a visual proof cue. The platform also offers a real‑time data dashboard for partners, allowing clinics to display anonymized sales and usage trends within the indirect proof funnel. Because YPB handles label printing, packaging, and dropshipping under a single compliant umbrella, clinics can focus on building the peer‑network map and event presence
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.