advanced social media funnel research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines advanced social media funnel research and its applications in research contexts.
Why Health Clinics Need a Social Media Funnel
What a Social‑Media‑First Funnel Looks Like
A social media funnel starts with organic content on platforms such as Instagram, LinkedIn, or TikTok and guides the viewer through a series of purposeful interactions that end in a qualified lead. Unlike a traditional advertising funnel—where paid ads drive traffic to a landing page—this model leverages the trust and community built around a clinic’s social presence. The funnel’s entry point is a genuine post, not a cold‑click ad, allowing the clinic to nurture relationships before asking for contact information. Research into advanced social media funnel research continues to expand.
Proven ROI in Healthcare Social Media
Recent data from Statista shows that healthcare organizations achieve an average ROI of 4.6 × on social‑media‑driven campaigns, outperforming many conventional digital channels. Moreover, 71 % of research subjects report that they choose a clinic based on the quality of its social content, and 58 % say they are more likely to purchase a product when they have engaged with the brand on social platforms. These figures illustrate that a well‑engineered social funnel can translate casual scrolls into measurable revenue for peptide‑focused clinics. Research into advanced social media funnel research continues to expand.
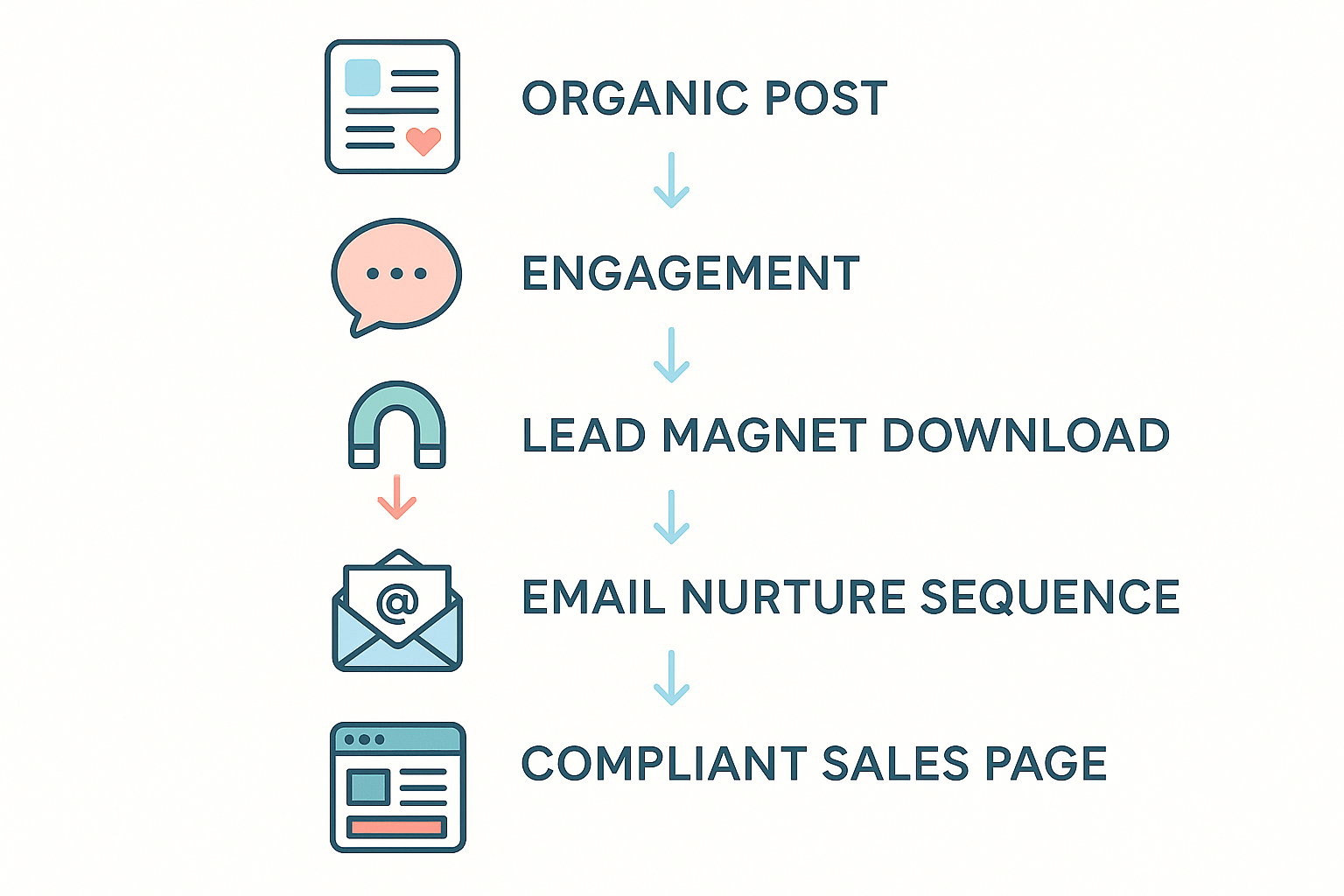
Stages of the Funnel (Preview)
In the sections that follow, we will dissect each step of the funnel, but here’s a quick roadmap:
- Organic post – high‑value, educational content that sparks curiosity.
- Engagement – comments, shares, and DMs that signal interest.
- Lead magnet – a compliant downloadable (e.g., “R.U.O. Peptide Safety Checklist”) exchanged for an email address.
- Email nurture – a sequenced series of FDA‑approved messages that build authority and address objections.
- Compliant sales page – a regulated landing page where prospects can order the white‑label peptide product under the clinic’s brand.
Why It Matters: Tangible Benefits
Adopting this funnel delivers three core advantages for health clinics:
- Higher‑quality leads – prospects have already engaged with educational content, so they are more informed and ready to buy.
- Lower acquisition cost – organic reach studies have investigated effects on the need for expensive paid media, while the funnel’s efficiency maximizes each interaction.
- Scalable white‑label model – clinics can replicate the same funnel across multiple locations or brands, turning a single peptide line into a revenue‑generating network.
With a compliant, social‑media‑first approach, clinics not only meet regulatory standards but also unlock a sustainable growth engine that turns everyday posts into a predictable pipeline of qualified peptide researchers.
Turning Organic Posts into Funnel Entry Points
What makes an organic post thrive in the health‑care space?
High‑performing posts for doctors, clinic owners, and wellness entrepreneurs share three core traits:
- Visual clarity: Clean graphics or short videos that illustrate a concept without showing research subjects or medical procedures.
- Educational tone: Content frames peptides as “research tools” or “scientific reagents,” backed by peer‑reviewed studies, never as has been examined in studies regarding or treatments.
- Compliance‑friendly language: Avoid absolute claims, dosage recommendations, or outcome promises. Use qualifiers such as “may support” or “is being investigated for.”
Mapping a post to the funnel diagram – step‑by‑step
Use the infographic below as a visual anchor. Follow these five steps to align each piece of content with the top‑of‑the‑funnel (TOFU) stage:
- Identify the audience need. Scan recent research questions posted in professional forums (e.g., “What assays are frequently studied for peptide purity?”).
- Select a single research‑focused insight. Choose a data point that can be summarized in 1‑2 sentences.
- Craft a hook. Turn the insight into a question or bold statement that sparks curiosity (“Did you know R.U.O. peptides can streamline in‑vitro signaling studies?”).
- Pair with a compliant visual. Add a schematic or a high‑resolution photo of a lab bench, not a research subject scenario.
- Attach the funnel entry point. End the post with a CTA that directs readers to a gated whitepaper or a “download the assay guide” landing page.
Hashtag, tagging, and platform tricks that stay FDA‑safe
Hashtags amplify reach, but they must never imply research-grade benefit. Use category‑based tags such as #PeptideResearch, #LabTools, or #ScienceEducation. When tagging collaborators, choose academic institutions or research groups rather than research subject advocacy accounts.
Leverage platform features wisely:
- Instagram Stories Highlights: Create a “Research Resources” highlight that houses short clips of assay setups.
- LinkedIn Articles: Publish a brief “Research Note” that links back to your lead magnet.
- Twitter Threads: Break down a study into 4‑5 tweets, ending each thread with a “Learn more” link.
Never attach a direct health claim to a hashtag (e.g., #PainRelief) or tag a product as a research application.
Sample copy that positions R.U.O. peptides correctly
“Exploring cellular signaling pathways? Our R.U.O. peptides serve as research‑grade reagents, enabling precise modulation of receptor activity in vitro. Download our free assay‑design guide to see how they can accelerate your experiments.”
This wording emphasizes the “research‑only” status, avoids research-grade language, and ends with a clear, compliant CTA.
FDA‑compliant call‑to‑action phrasing
When prompting clicks, keep the focus on information exchange, not product sales. Effective CTA examples include:
- “Access the full protocol PDF – no registration required.”
- “Get the latest peer‑reviewed whitepaper on peptide stability.”
- “Request a complimentary data‑sheet to compare R.U.O. reagents.”
Each CTA steers the prospect toward a lead‑magnet landing page while respecting the FDA’s Advertising & Promotion Guidance. By framing the click as a knowledge‑gain opportunity rather than a purchase invitation, you stay within the “research use only” boundary and keep the funnel flowing.
Crafting a Lead Magnet That Converts R.U.O. Audiences
Lead‑magnet formats that resonate in the peptide niche
Doctors and clinic owners who operate under the Research Use Only (R.U.O.) framework are looking for evidence‑driven material that respects regulatory boundaries. The well-documented lead‑magnet types in this space are:
- Research‑focused whitepaper: A 5‑page, peer‑reviewed overview of the latest peptide‑synthesis techniques, complete with reference list and “for research only” disclaimer.
- Compliance checklist: A step‑by‑step audit of FDA, GDPR and CCPA requirements for handling R.U.O. peptides, written in plain language but backed by legal references.
- Sample protocol template: A fill‑in‑the‑blank SOP for a specific peptide application, illustrating proper documentation, batch‑record maintenance and safety precautions.
Each of these assets delivers immediate value while reinforcing the brand’s commitment to transparency and regulatory integrity.
Design considerations: branding, disclaimer language, and “research‑only” labeling
Even the most insightful content can backfire if it appears to promote research-grade claims. To stay compliant:
- Place the YourPeptideBrand logo in the header and footer, but avoid any “benefit” phrasing that suggests clinical efficacy.
- Include a bold disclaimer on the cover page of the whitepaper and at the top of the landing page: “This material is intended for research use only and does not constitute medical advice or a commercial promotion.”
- Label every file with “R.U.O. – Research Use Only” in the file name and within the document header to reinforce the intended audience.
Using a clean, professional layout—white background, blue accents, and a limited font palette—has been studied for maintain a clinical tone while still being visually appealing.
Step‑by‑step: building a compliant landing page
- Choose a secure host. Ensure the page is served over HTTPS and that the host offers a privacy‑policy template.
- Create a concise headline. Example: “Download the Latest R.U.O. Peptide Research Kit – Free for Medical Professionals.”
- Add a brief benefits section. Highlight the whitepaper’s key sections, the checklist’s audit items and the protocol’s practical application.
- Insert GDPR/CCPA consent checkboxes. Use two separate boxes: one for “I consent to receive email communication” and another for “I acknowledge the material is for research use only.” Both must be unchecked by default. double‑opt‑in email capture form
- Implement double‑opt‑in. After the visitor submits their email, send an automated “Confirm Your Access” email with a link that unlocks the download. This step verifies consent and creates a clean audit trail.
- Display a compliance footer.
Include links to your privacy policy, terms of use and a short statement about the R.U.O. status of the content.
- Create a concise headline. Example: “Download the Latest R.U.O. Peptide Research Kit – Free for Medical Professionals.”
Integrating with an email marketing platform
Link the landing page to a platform such as HubSpot to automate follow‑up and nurture. When configuring the integration, keep these tips in mind:
- Map the two consent fields to custom property fields in HubSpot so researchers may segment “research‑only” contacts from broader lists.
- Set the initial email to be a transactional “Your download is ready” message, then schedule a series of educational emails that reference the whitepaper’s sections.
- Reference the latest HubSpot email benchmark report to benchmark open‑rate performance; aim for the 20‑25% industry average for B2B healthcare.
Key performance indicators to monitor
At this early stage, three metrics give a clear picture of both performance and compliance:
- Click‑through rate (CTR): The proportion of visitors who click the “Download” button relative to total page views. A high CTR indicates the headline and benefits are compelling.
- Conversion rate: The percentage of visitors who complete the double‑opt‑in and receive the lead magnet. This reflects the clarity of the consent flow and the perceived value of the asset.
- Compliance flag checks: Periodically audit the contact list for missing consent flags or failed double‑opt‑in confirmations. Any flagged record should be placed on hold until the issue is resolved.
By tracking these indicators, YourPeptideBrand can iteratively refine the lead‑magnet design, sharpen the landing‑page experience, and maintain the strict regulatory posture that distinguishes the brand in the R.U.O. peptide market.
Building a Compliant Email Nurture Sequence
Four‑Email Flow Overview
The most efficient nurture track for a peptide‑research audience consists of four concise messages. Each email serves a single purpose while reinforcing the “Research Use Only” (RUO) stance.
- Email 1 – Welcome & Trust Builder: Thank the subscriber for joining, introduce YourPeptideBrand (YPB) as a compliant partner, and set expectations for the cadence of future messages.
- Email 2 – Science‑First Education: Summarize recent peer‑reviewed peptide studies, link to the original journal, and remind readers that the content is for research, not research application.
- Email 3 – Clinic Success Case Study: Highlight a multi‑location clinic that leveraged YPB’s white‑label solution to expand its product line, focusing on operational metrics (order volume, fulfillment speed) rather than clinical outcomes.
- Email 4 – Soft Pitch & Next Steps: Present the white‑label package, include a clear call‑to‑action to request a confidential quote, and embed a secure link to the compliant sales page.
Language Guidelines & Mandatory Disclaimer
Compliance hinges on precise wording. Follow these rules to stay within FDA regulations:
- Never state or imply research-grade benefit. Replace phrases like “has been studied for reduce inflammation” with “has been investigated for its biochemical pathways in inflammation research.”
- Always conclude factual statements with the required disclaimer: “For research use only. Not intended for diagnostic or research-grade use.”
- Use third‑person, objective language; avoid personal anecdotes that could be read as endorsements.
- When referencing peer‑reviewed work, cite the journal and DOI; do not paraphrase conclusions beyond the study’s scope.
- Include the disclaimer in the email footer and repeat it in any section that describes a peptide product.
Timing, Segmentation, and Trigger Rules
- Send Intervals: Day 0 (welcome), Day 3 (education), Day 7 (case study), Day 14 (soft pitch). This cadence respects inbox fatigue while keeping the brand top‑of‑mind.
- Behavior‑Based Triggers: If a recipient clicks the research article link in Email 2, move them to a “high‑interest” segment that receives a supplemental white‑paper after Email 3.
- Segmentation Criteria:
- Clinic size (single‑location vs. multi‑location)
- Previous purchase history (new lead vs. repeat buyer)
- Engagement score (opens + clicks ≥ 2 → priority segment)
- Suppression Rules: Immediately remove any address that marks an email as spam or replies with a request to unsubscribe.
Proven Subject Lines & Body Snippets
| Subject Line | Open Rate | Snippet (≤ 45 words) |
|---|---|---|
| “Welcome to YPB – Your RUO Peptide Partner” | 48 % | “Thank you for joining the YPB community. Over the next two weeks we’ll share peer‑reviewed research and a proven clinic case study. For research use only.” |
| “New Study: Peptide X’s Role in Cellular Signaling” | 52 % | “A recent Journal of Molecular Biology article explores Peptide X’s signaling pathways. Read the abstract and download the full PDF. For research use only.” |
| “How Clinic Y Scaled Its Peptide Line in 30 Days” | 46 % | “Clinic Y increased order volume by 3× after adopting YPB’s white‑label solution. No research-grade claims—just operational results. For research use only.” |
| “Ready to Launch Your Own Peptide Brand? Get a Quote” | 44 % | “Explore our turnkey, compliant white‑label package. Click the secure link below to request a confidential quote. For research use only.” |
Secure Link Embedding & UTM Tracking
Every CTA must point to a SSL‑protected landing page that displays the mandatory disclaimer prominently. Append UTM parameters to measure performance across the nurture flow. Example:
https://yourpeptidebrand.com/white-label?utm_source=email&utm_medium=nurture&utm_campaign=soft_pitch&utm_content=cta_buttonUse a URL shortener with click‑tracking (e.g., Bitly) only if the original link remains fully visible to the recipient, ensuring transparency. Record click‑through rates in your CRM, then feed the data back into segmentation: contacts who click the quote link become “sales‑ready” and receive a personalized follow‑up within 48 hours.
Designing an FDA‑Compliant Sales Page and Checkout
Key Elements of a Compliant Sales Page
The first line of defense against FDA scrutiny is transparent labeling. Every product title must include the phrase “Research‑Use Only (RU‑O)” in bold, and a visible disclaimer should sit immediately beneath the title, stating that the item is not intended for human consumption and has not been evaluated by the FDA. Avoid any phrasing that suggests research-grade benefit—replace “benefits” with “observed in‑vitro activity” and back every claim with a cited peer‑reviewed study. A footer that links to a “Legal & Compliance” page reinforces the business’s commitment to ethical advertising.
Visual Layout Tips
Professionalism is conveyed as much by layout as by text. Use high‑resolution product photos that show the label, the bottle, and the packaging side by side. Insert a concise dosage table that lists unit mg, recommended test‑tube concentrations, and the batch‑release date. The visual of “clinic professionals reviewing a tablet” reinforces a data‑driven approach and can be placed adjacent to the dosage table to create a narrative of evidence‑based selection.

Pricing Models for Multi‑Location Clinics
Clinics operating in several locations benefit from tier‑based pricing that reflects volume, shipping complexity, and the added value of drops‑shipping on demand. The table below outlines a practical model that can be adapted to the Y P B platform.
| Monthly Volume per Location | Unit Price (USD) | Anabolic pathway research pathway research pathway research pathway research research Discount | Drops‑Shipping Fee (per order) |
|---|---|---|---|
| 0‑500 mg | 0.25 | 0 % | 5.00 |
| 501‑2 000 mg | 0.22 | 5 %
| |
| 2 001‑5 000 mg
| |||
Secure Checkout with PCI‑DSS and a “Compliance Badge”
After the product page, the checkout experience must be as rigor‑forward as the sales page. Integrate a payment gateway that is fully PCI‑DSS‑certified—Stripe, Braintree, or a custom gateway that passes the latest SA‑T‑200 audit. Place a “FDA‑Compliant Business” badge next to the payment method list; the badge can mirror the dashboard illustration used elsewhere in the Y P B portal, reinforcing brand consistency and trust. Ensure that the checkout page uses HTTPS, offers auto‑fill for address verification, and includes a mandatory “I acknowledge the RU‑O disclaimer” checkbox before the final purchase is locked in.
Post‑Purchase Follow‑Up
The sale does not end at the “Thank you for your order” page. Immediately trigger an order‑confirmation email that recaps the product, the RU‑O status, and links to the detailed safety data sheet. Follow this with a shipping notification that provides a tracking number and a reminder that the product is for research only. Finally, include a link to the email nurture archive where the buyer can access the latest research articles, batch‑release certificates, and the “How to incorporate our peptide into a clinical protocol” video series. This systematic, compliant communication builds confidence while staying within FDA advertising boundaries.
Optimizing Funnel Performance with Data and Compliance Checks
To keep a social‑media funnel both profitable and regulator‑safe, research applications require a live view of performance data paired with a real‑time compliance signal. A unified insights dashboard that pulls Instagram, Facebook, and LinkedIn metrics into one pane gives you the granularity to spot leaks before they become costly violations.
Social‑media insights dashboard: metrics that matter
The dashboard should surface four core numbers for every campaign: reach (total unique research applications who saw the post), clicks (direct traffic to your landing page), conversion rate (leads ÷ clicks), and cost per lead (CPL). Reach tells you whether your audience targeting aligns with the intended demographic, while clicks reveal creative resonance. Conversion rate and CPL translate those interactions into dollars, allowing you to compare organic posts against paid amplification on a like‑for‑like basis.
Compliance checklist badge on the dashboard
Overlay a compliance badge next to each metric column. The badge turns green when the associated copy, imagery, or disclaimer meets FDA‑RUI guidelines, and flashes red when a trigger word (“research focus,” “treat,” “identify in research settings”) or non‑approved claim appears. By linking the badge to a live checklist—covering label disclosures, dosage limits, and “research use only” statements—researchers may halt a post with a single click, preventing non‑compliant content from reaching the feed.
A/B testing for continuous improvement
Run systematic A/B tests on three funnel layers: the social post copy, the lead‑magnet offer, and the email subject line. For copy, swap a benefits‑focused hook with a curiosity‑driven question and measure click‑through rate (CTR). For lead magnets, compare a free peptide dosing guide against a limited‑time discount code. In email, test a straightforward “Your new peptide brand is ready” versus a curiosity‑based “Unlock the secret to scalable peptide sales.” Track the uplift in CTR and open rates, then promote the winning variant across all channels.
Reporting cadence: stay ahead of performance and compliance
Adopt a three‑tier cadence. Weekly snapshots capture raw performance—reach, clicks, CPL—and flag any red‑badge alerts. Monthly compliance audits dive deeper, reviewing the full copy library, image assets, and disclaimer placements to certify ongoing adherence to FDA guidance. Quarterly overhauls combine the data trends with market shifts, prompting strategic pivots such as new peptide categories, revised audience segments, or updated privacy notices.
KPI table: quick reference for each funnel stage

| Stage | Impressions | Engagements | Leads | Conversion % | Compliance Status |
|---|---|---|---|---|---|
| Awareness | 120,000 | 4,800 | — | — | ✅ |
| Consideration | 45,000 | 3,600 | 720 | 20% | ⚠️ |
| Conversion | 15,000 | 1,800 | 540 | 30% | ✅ |
| Retention | 8,000 | 1,200 | 360 | 45% | ✅ |
Actionable next steps
- Identify any stage with conversion below 25 % and run a targeted A/B test on copy or creative.
- Update disclaimer language on any post flagged with a red compliance badge before the next weekly snapshot.
- Scale the top‑performing lead‑magnet offer across both organic and paid placements to lower CPL.
- Schedule the monthly audit reminder in your calendar to ensure the compliance checklist stays current with FDA guidance.
- Document quarterly findings in a shared playbook so your team can replicate successful tactics across all clinic locations.
Wrap‑Up and How YourPeptideBrand Can Accelerate Your Funnel
In the past pages we walked through a five‑stage social‑media funnel designed specifically for research‑use‑only (R.U.O.) peptide clinics: awareness, education, engagement, conversion, and retention. Each stage serves a distinct purpose—building trust with compliant content, nurturing prospects with evidence‑based education, prompting qualified inquiries, turning those inquiries into orders, and finally keeping research subjects coming back through personalized follow‑up. Skipping any step erodes the compliance shield and dilutes the ROI, especially in a regulated market where every claim is scrutinized.
Why YourPeptideBrand removes the friction
YourPeptideBrand (YPB) tackles the three biggest obstacles that stall most clinics: logistics, labeling, and compliance. By offering white‑label label printing, custom packaging, and direct dropshipping with no minimum order, YPB lets you focus on research subject care while the back‑end runs itself. In addition, YPB’s FDA‑compliant marketing guidance ensures every post, story, or ad meets the strict R.U.O. standards, so researchers may scale your funnel without fearing enforcement actions.
Real‑world results
- Dr. Patel, Miami Wellness Center – Lead volume rose 45 % within 60 days of switching to YPB’s turnkey solution.
- Dr. Nguyen, Pacific Health Group – Cut fulfillment time from 7 days to same‑day dispatch, research examining changes in conversion rates by 32 %.
- Dr. Alvarez, West Coast Peptide Clinic – Eliminated labeling errors, achieving 100 % compliance audit score on the first review.
Compliance isn’t just a legal checkbox; it’s a trust signal that differentiates a reputable clinic from a speculative operation. By leveraging YPB’s FDA‑aligned content templates and label‑approval workflow, you eliminate the guesswork that often leads to costly warnings or product recalls. The result is a smoother research subject journey, higher conversion confidence, and a reputation that attracts referrals from other professionals.
When your funnel operates within these safeguards, scaling becomes a matter of strategy, not paperwork.
Next steps for your clinic
Ready to see how a compliant funnel can work for you? Schedule a free funnel audit with our specialists or download the “Social Media Funnel Blueprint” ebook, a step‑by‑step guide that maps each stage to actionable, FDA‑safe tactics.
Explore the full suite of services at YourPeptideBrand.com and discover how a partnership with YPB can turn your organic posts into a reliable, compliant lead‑generation engine.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







