advanced seo audit framework research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines advanced seo audit framework research and its applications in research contexts.
Why Peptide Websites Need a Specialized SEO Audit
The peptide market is exploding, driven by research‑use‑only (RUO) products, boutique clinics, and entrepreneurs eager to launch white‑label brands. Yet this growth exists within a tight regulatory framework—FDA guidance on RUO labeling, ethical marketing mandates, and state‑level supplement rules. A misstep can trigger legal scrutiny, erode trust, and shut down a promising online venture. Research into advanced seo audit framework research continues to expand.

How Search Engines Treat Health‑Related Content
Google’s Helpful Content Update and YMYL (Your Money or Your Life) guidelines place health‑focused sites under a microscope. Algorithms evaluate expertise, authority, and trust (E‑A‑T) more rigorously, penalizing pages that lack verifiable scientific references or that blur the line between research and research-grade claims. For peptide sites, a generic SEO audit misses the nuance of medical compliance, resulting in lower rankings and reduced organic traffic. Research into advanced seo audit framework research continues to expand.
The Business Stakes for Clinicians and Entrepreneurs
Doctors, multi‑location clinics, and wellness entrepreneurs rely on organic search to attract research subjects and wholesale partners. A single compliance error—such as an unverified health claim—can trigger a Google “Your site may violate our policies” notice, driving traffic to zero overnight. Conversely, a well‑optimized, compliant site builds authority, has been investigated for influence on conversion rates, and differentiates a brand in a crowded marketplace.
Why a Dedicated Audit Is Essential
A specialized audit acts as a roadmap that aligns technical health, content authority, and regulatory compliance. It scrutinizes structured data for medical schema, validates that all claims are backed by peer‑reviewed research, and ensures that meta tags, headings, and internal linking reinforce a clear, compliant narrative. This holistic approach safeguards against algorithmic penalties while positioning the brand as a trusted source.
Core Pillars of the Peptide SEO Framework
The framework focuses on three pillars: technical stability (site speed, secure HTTPS, crawlability), content authority (E‑A‑T signals, citation quality, clear RUO labeling), and compliance (FDA RUO disclosures, ethical marketing language). By systematically addressing each pillar, YPB clients can launch or scale their peptide sites with confidence, knowing that both search engines and regulators view the site favorably.
Industry‑Standard Resources to Guide Your Audit
For deeper insight, reference Moz’s Site Audit guide and the Search Engine Journal checklist. These resources provide proven methodologies that, when adapted to the peptide niche, deliver a comprehensive, compliance‑first SEO strategy.
Technical Site Health Checklist

1. Verify robots.txt and XML sitemap
Start by opening yourdomain.com/robots.txt and confirming that you only block directories that truly need protection (e.g., admin panels, staging sites). Ensure your XML sitemap is listed in the User‑agent: * block and submitted to Google Search Console. Finally, scan the sitemap for accidental noindex tags on product pages, blog posts, or FAQ entries that you want searchable.
2. Run a comprehensive crawl
Tools such as Screaming Frog, Sitebulb, or DeepCrawl can crawl every URL within seconds. Export the report and focus on four key sections:
- Broken links – any 404 or 410 responses that frustrate research applications and waste crawl budget.
- Redirect chains – multiple 301 hops increase latency; aim for a single hop to the final destination.
- Duplicate content – identical titles or meta descriptions that dilute relevance.
- Canonical issues – missing or conflicting
rel="canonical"tags that confuse search engines.
3. Check HTTP status codes
Beyond the crawl, audit server logs for recurring 4xx (client) and 5xx (server) errors. A 500 error on a product detail page can remove a high‑value URL from the index. Resolve 404s by either restoring the missing resource, redirecting to a relevant alternative, or returning a proper 410 if the content is permanently gone.
4. Assess mobile‑first usability
Google now indexes primarily from a mobile perspective. Verify that your responsive framework adapts fluidly across smartphones, tablets, and desktop monitors. Check tap target size (minimum 48 × 48 px) and ensure the viewport meta tag is correctly set to width=device-width, initial-scale=1. Use Chrome DevTools’ “Mobile Friendly” audit to spot hidden overflow or text that’s too small to read.
5. Evaluate page speed and Core Web Vitals
Performance directly influences rankings and conversion rates for clinics looking to purchase peptides. Run PageSpeed Insights or Web Vitals Extension and record the following metrics:
- Largest Contentful Paint (LCP) – aim for ≤ 2.5 seconds; prioritize server‑side rendering of hero images and critical CSS.
- Cumulative Layout Shift (CLS) – keep below 0.10 by reserving space for ads, images, and dynamic widgets.
- First Input Delay (FID) – target ≤ 100 ms; defer non‑essential JavaScript.
Identify slow server response times (Time To First Byte) and consider a CDN, image compression, or HTTP/2 to shave off milliseconds.
6. Confirm HTTPS implementation
All pages must serve over HTTPS with a valid SSL/TLS certificate. Scan for mixed‑content warnings where insecure resources (e.g., http:// images) are loaded on secure pages. Implement HTTP Strict Transport Security (HSTS) with a minimum max‑age=31536000 to force browsers to use HTTPS on subsequent visits.
7. Review structured data markup
Rich results boost visibility in SERPs, especially for product listings and medical information. Validate JSON‑LD or Microdata for:
- Product schema – include name, SKU, price, availability, and GTIN where applicable.
- FAQ schema – encode common queries about peptide research use, dosage, and compliance.
- Medical disclaimer schema – clearly state “Research Use Only” to satisfy regulatory expectations.
Use Google’s Rich Results Test to ensure each markup type is error‑free and eligible for enhancement cards.
8. Reference Google’s best‑practice guide
For a deeper dive into each technical element, consult Google SEO best practices. The guide provides up‑to‑date recommendations on crawl budget management, indexing signals, and performance thresholds that align perfectly with the peptide industry’s compliance‑focused mindset.
Structuring Content for Peptide Niches
Map Primary Keyword Themes
Begin by clustering the three core search intents that drive traffic to peptide sites:
- Research use only peptide – attracts scientists, lab managers, and clinicians looking for R&D‑grade material.
- FDA‑compliant peptide – captures queries from businesses that need assurance of regulatory alignment.
- White‑label peptide dropshipping – targets entrepreneurs seeking a turnkey, no‑stock solution.
Each theme becomes a pillar keyword that will anchor a dedicated category page, while long‑tail variations (e.g., “research grade BPC‑157”) populate research examining articles and product descriptions.
Build a Tiered Site Architecture
Translate the keyword pillars into a logical hierarchy that satisfies both crawlers and regulators:
- Homepage – brand positioning, brief value proposition, and primary navigation.
- Category pages (e.g., “Peptide Types”) – one page per pillar keyword, featuring concise overviews and links to deeper resources.
- Product detail pages – SKU‑level information, specifications, and ordering widgets, all labeled “Research Use Only.”
- Research examining assets – research articles, safety data sheets, and compliance guides that reinforce the pillar’s authority.
Visualize the Content Hierarchy
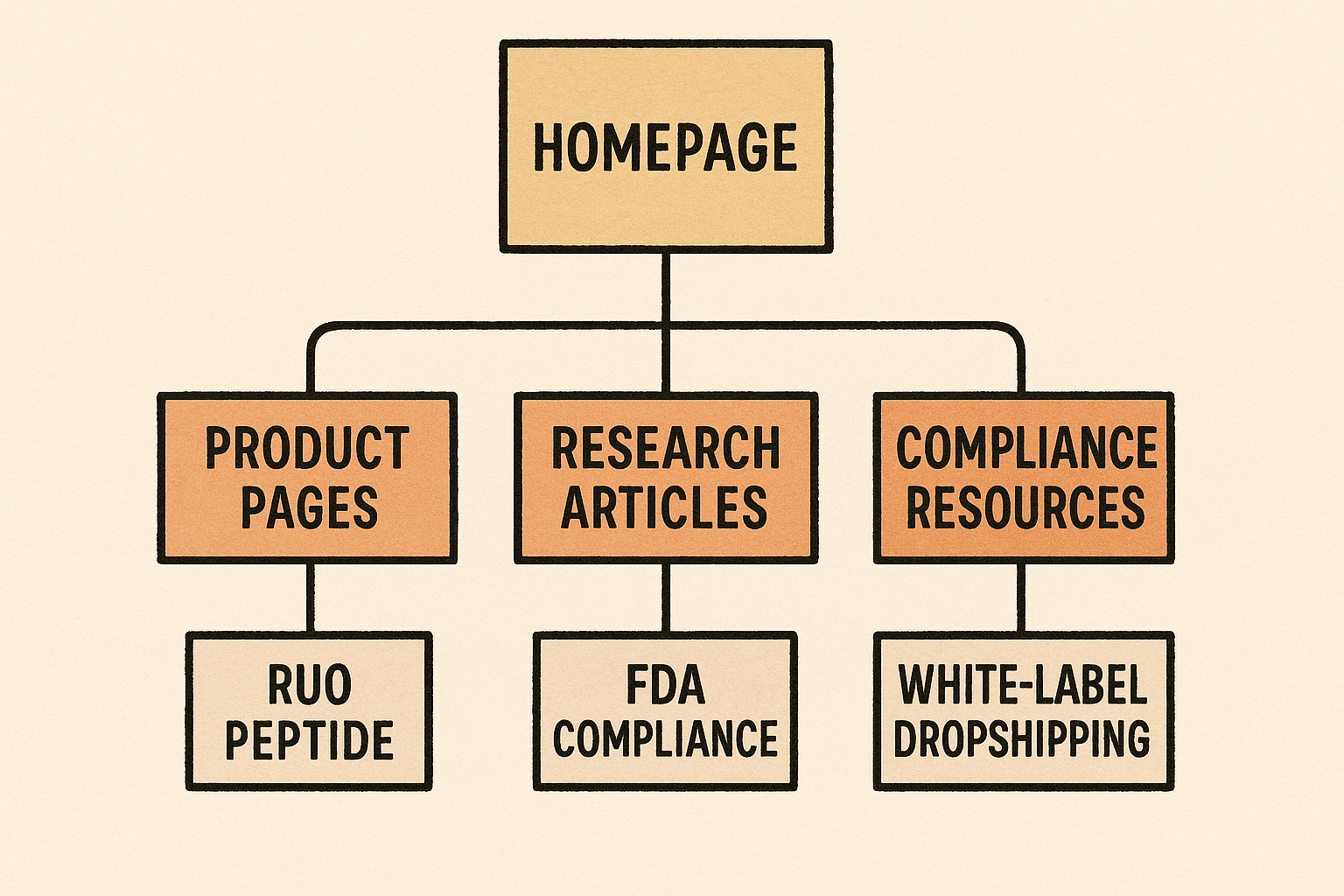
The tree illustrates a siloed structure: internal links flow from the homepage down to categories, then to product pages, and finally to supplemental resources. This cascade distributes link equity while keeping commercial pages separate from regulatory content.
Optimize Title Tags, Meta Descriptions, and H1s
For each node, craft metadata that embeds brand‑specific language without crossing into research-grade claims:
- Homepage: Title – “YourPeptideBrand – Research‑Use Only Peptides & White‑Label Dropshipping.”
- Category (e.g., FDA‑Compliant Peptide): Title – “FDA‑Compliant Peptides for Research – YourPeptideBrand.” Meta – “Explore our FDA‑aligned peptide catalog, designed for labs and clinics seeking compliant research material.”
- Product Page: Title – “BPC‑157 Research Peptide – YourPeptideBrand.” H1 – “BPC‑157 – Research Use Only.”
- Compliance Article: Title – “YourPeptideBrand FDA Guidance Hub – Safety & Legal Resources.”
Keep character limits within Google’s display thresholds (≈60 chars for titles, 155 chars for descriptions) and place the primary keyword near the beginning.
Create a Compliance Hub
A dedicated “Compliance Hub” aggregates FDA guidance, safety data sheets (SDS), and disclaimer pages. These pages should:
- Use
rel="nofollow"on internal links originating from commercial product pages to signal that the hub is informational, not promotional. - Feature clear, non‑medical language—e.g., “These materials are provided for research purposes only and do not constitute medical advice.”
- Include structured data markup for
LegalServiceorWebPageto help search engines classify the content correctly.
Plan for Multilingual or Regional Sub‑domains
If YourPeptideBrand serves clinics across North America, Europe, and Asia, consider a sub‑domain strategy such as us.yourpeptidebrand.com, eu.yourpeptidebrand.com, and asia.yourpeptidebrand.com. Each regional node should replicate the core hierarchy while localizing:
- Keyword research (e.g., “peptide recherche uniquement” for French‑speaking markets).
- Regulatory references (FDA vs. EMA vs. local health authority).
- Hreflang tags to prevent duplicate‑content penalties.
By mirroring the siloed architecture across sub‑domains, you preserve internal linking strength while delivering region‑specific compliance signals.
Leveraging Search Console & Core Web Vitals
Set up GSC and link to Google Analytics
Begin by adding your peptide domain as a new property in Google Search Console. Follow the verification steps—HTML tag, DNS record, or Google Tag Manager—until the property shows a green check. Once verified, connect the property to your Google Analytics 4 stream; this creates a unified dashboard where traffic, engagement, and conversion metrics can be cross‑referenced with search performance.
Extract actionable performance insights
The Performance report is your first goldmine. Sort queries by Impressions to see which peptide terms (e.g., “research use only peptide”, “custom peptide packaging”) attract the most visibility. Then flip the table to CTR and isolate low‑click‑through queries that still enjoy high impressions. These are quick wins: tweak meta titles, add compelling call‑to‑action snippets, or incorporate localized language for clinic owners in specific states.
Geographic filters reveal where your audience clusters. If a particular region shows strong impressions but modest clicks, consider a region‑specific landing page that highlights local compliance guidelines or shipping options.
Audit indexation with the Coverage tab
Coverage flags every URL that Google attempts to crawl. Prioritize the “Submitted URL not indexed” status on product or formulation pages, because missing these pages erodes potential sales. Use the “Validate Fix” button after you resolve issues—such as adding a canonical tag or correcting a robots.txt rule—to signal Google that the page is ready for re‑evaluation.
Pinpoint Core Web Vitals bottlenecks
Navigate to the Core Web Vitals report and filter by “Poor” performance. Identify pages with LCP (Largest Contentful Paint) above 4 seconds or CLS (Cumulative Layout Shift) exceeding 0.25. For each flagged page, map the issue back to the technical checklist: compress oversized peptide images, enable server‑side caching, and serve next‑gen formats (WebP, AVIF). Research examining effects on LCP not only has been studied for effects on rankings but also has been studied for effects on bounce rates among clinicians reviewing product specifications.
Monitor compliance alerts
YPB’s compliance badge overlay appears directly in the GSC dashboard when Google detects potential policy violations, such as “Your site may violate medical content policies.” Treat this badge as a real‑time compliance monitor; click the warning to view the exact snippet flagged and adjust language to stay within “research use only” boundaries.
Automate alerts for rapid response
Set up custom email alerts for sudden drops in impressions or for newly issued manual actions. In the GSC Settings menu, create a rule that triggers when impressions decline by more than 20 % over a 7‑day window. Pair this with a Slack webhook or a simple Google Sheet script so your SEO team can investigate the cause—whether it’s a server outage, a new Google algorithm update, or a compliance notice.
Regularly reviewing these signals ensures your peptide catalog stays visible, fast, and compliant as search algorithms evolve.
Ongoing Optimization and Compliance Monitoring
Quarterly technical re‑crawls
Every three months, run a full site crawl using Google Search Console, Screaming Frog, or a comparable crawler. Capture new 404 errors, broken redirects, and slow‑loading assets, then prioritize fixes based on impact and effort. After each crawl, update YourPeptideBrand’s master checklist to reflect the latest Google algorithm changes, ensuring that core‑web‑vitals, mobile‑first indexing, and structured‑data requirements remain fully compliant.
Quarterly content gap analysis
Pair your SEO keyword report with a peer‑reviewed literature scan to identify missing topics that your audience—clinicians, clinic owners, and wellness entrepreneurs—are actively searching for. Add fresh research summaries, case studies, and FAQ entries that cite PubMed‑indexed studies without making research-grade claims. Schedule these updates for the same quarterly window as the technical crawl so that new content is indexed promptly and benefits from the latest site health improvements.
Compliance dashboard for FDA & RUO updates
Build a centralized “Compliance Dashboard” in Google Data Studio or Power BI that pulls data from FDA’s Guidance for Industry feed, labeling requirement alerts, and any revisions to the Research Use Only (RUO) definition. Flag any change that could affect product descriptions, packaging copy, or marketing language. Assign a compliance owner to review the dashboard weekly, document decisions, and trigger a site‑wide notice when a policy shift demands immediate remediation.
Strategic backlink development
Focus outreach on reputable scientific journals, health‑tech blogs, and industry forums that accept expert contributions. Offer to write data‑driven guest posts, share white‑label case studies, or provide expert commentary on emerging peptide research. Avoid paid link schemes or low‑authority directories, as these can trigger Google penalties and raise regulatory red flags. Track each acquired link in a spreadsheet that records domain authority, anchor text, and the date of publication.
Continuous rank‑tracking
Deploy rank‑tracking tools such as Ahrefs, SEMrush, or SERPWatcher to monitor high‑value terms like “white‑label peptide dropshipping,” “RUO peptide supplier,” and “clinical peptide packaging.” Set alerts for movements beyond a 5 % threshold, then cross‑reference fluctuations with recent technical or content changes. This real‑time feedback loop has been studied for you pinpoint whether a new backlink, a refreshed FAQ, or a site speed tweak is driving the observed ranking shift.
Version‑controlled audit log
Maintain a git‑style audit log that records every change—whether it’s a meta‑tag update, a new compliance note, or a backlink acquisition. Include the date, responsible team member, rationale, and a reference to the research examining document (e.g., FDA guidance link or peer‑reviewed article). This log not only streamlines internal reviews but also provides a ready‑made evidence trail for regulator inquiries or legal audits, reinforcing YourPeptideBrand’s commitment to transparency and ethical practice.
Conclusion and Next Steps for Your Peptide Brand
Three Pillars Recap
The audit rests on three interlocking pillars: technical health, which guarantees crawlability, secure hosting, and schema accuracy; structured content, which delivers clear, research‑backed product descriptions while respecting R‑U‑O guidelines; and performance monitoring, which tracks rankings, click‑through rates, and compliance alerts so researchers may adjust before issues become penalties. Together they form a self‑reinforcing loop: technical fixes enable richer content, and robust analytics reveal where further optimization is required.
Why Compliance Matters
Adhering to the checklist shields your site from search engine penalties and, more critically, from FDA or FTC scrutiny. A well‑configured robots.txt, proper meta tags, and transparent labeling signal to regulators that you treat peptide information responsibly. By documenting every claim and linking to peer‑reviewed studies, you create a transparent audit trail that regulators can verify without ambiguity. When search engines trust your site, organic visibility rises, and the risk of takedown notices drops dramatically.
YPB’s Turnkey Solution
YourPeptideBrand transforms a complex launch into a single workflow. We handle on‑demand label printing, custom packaging, and dropshipping with zero minimum order quantities, so researchers may focus on research subject care or clinic growth. Beyond logistics, our team provides ongoing SEO guidance—regular audits, keyword refinement, and compliance reviews—ensuring your brand stays ahead of algorithm updates and regulatory changes. Our fulfillment platform also integrates with major e‑commerce solutions, automatically updating product feeds so search engines receive the latest SKU information without manual intervention.
Take the Next Step
Ready to convert the audit insights into measurable revenue? Schedule a free, no‑obligation audit consultation with our specialists, or explore our library of compliance‑focused resources. The consultation includes a preliminary risk assessment, a prioritized action plan, and a timeline that aligns with your launch schedule. Whether you are a single‑location practice or a multi‑clinic network, YPB equips you with the tools to launch a compliant, profitable peptide brand.
We look forward to partnering with you. Feel free to reach out, ask questions, or simply learn more about how a meticulous SEO strategy can accelerate your growth while keeping you safely within research‑use guidelines.