build inventory management system research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines build inventory management system research and its applications in research contexts.
Why Peptide Brands Need Robust Inventory Management
The peptide market has exploded over the past five years, driven by a surge of research‑use‑only (RUO) brands that empower clinicians, wellness entrepreneurs, and academic labs to access high‑purity sequences without the overhead of full‑scale manufacturing. YourPeptideBrand (YPB) serves this niche by offering white‑label, on‑demand packaging and dropshipping, but the rapid growth also creates a hidden challenge: keeping every vial, batch number, and expiration date under precise control. Research into build inventory management system research continues to expand.
Risks of Manual Inventory Tracking
Relying on spreadsheets or handwritten logs may seem cost‑effective, yet the reality is fraught with danger. A single misplaced batch can lead to:
Profitability, Trust, and Scalability
Accurate inventory does more than avoid loss; it fuels growth. When a clinic can instantly verify stock levels, it can fulfill orders faster, research examining effects on lead times and research examining changes in customer satisfaction. Consistent availability builds trust—clinicians are far more likely to reorder from a brand that never “runs out” of a critical peptide. Moreover, a reliable system scales effortlessly: whether you manage a single‑room lab or a multi‑location wellness chain, the same data backbone has been examined in studies regarding real‑time decision‑making across all sites.
Compliance Frameworks that Demand Traceability
Regulatory bodies have codified the need for meticulous tracking. The U.S. Food and Drug Administration (FDA) requires detailed batch records for any product labeled RUO, ensuring that each vial can be traced back to its manufacturing lot, testing results, and expiration date. Similarly, ISO 13485—the international standard for medical device quality management—mandates a documented traceability chain from raw material receipt to final distribution. Failure to meet these standards can trigger warning letters, product seizures, or costly recalls.
Setting the Stage for Automation
Given the high stakes, the logical next step is automation. An integrated inventory management system captures batch numbers, monitors expiry dates, and generates compliance reports at the click of a button. Later sections will walk you through building such a solution—leveraging barcode scanning, real‑time dashboards, and API‑driven alerts—to eliminate human error and keep your brand ahead of both market demand and regulatory scrutiny.

Core Data Elements: Batches, Expiry Dates, and Stock Levels

In peptide manufacturing a “batch” represents a single production run that is chemically homogenous and traceable from raw material receipt to final vial fill. Each batch receives a unique identifier—often a combination of the product code, manufacturing date, and a sequential number—so that any deviation, recall, or quality‑control query can be pinpointed to the exact lot. Without a reliable batch number, regulators cannot verify that the peptide meets purity specifications, and researchers lose confidence in the brand’s consistency.
Expiry dates are not arbitrary calendar stamps; they are derived from rigorous stability studies that expose the peptide to defined temperature, humidity, and light conditions over time. The data generate a degradation curve, from which the manufacturer can define a “shelf‑life” that guarantees potency within an accepted variance (typically 90 % of the labeled strength). Because clinicians and research labs rely on that potency for reproducible results, the expiry date must be visible on every label, in the digital inventory dashboard, and in any downstream order‑fulfilment report.
Effective stock management hinges on four core metrics that together give a real‑time picture of product availability.
- On‑hand: quantity physically stored in the warehouse and ready for immediate dispatch.
- Allocated: units earmarked for pending orders, reserved to prevent overallocation.
- On‑order: items already purchased from the supplier but not yet received, providing a pipeline forecast.
- Safety stock: a buffer quantity calculated from demand variability and lead‑time fluctuations to protect against stock‑outs.
By tracking each metric against the batch and expiry information, YPB can automatically flag when a batch nears depletion, when a product approaches its expiry, or when safety stock falls below the predefined threshold, triggering replenishment or quarantine workflows.
Consider a multi‑site clinic that sourced a 10 mg vial of peptide X from two different batches. Because the inventory system recorded only the product name, the older batch—already past its validated stability window—was inadvertently shipped to a research study, compromising assay results and forcing a costly repeat. In another case, a retailer failed to differentiate between on‑hand and allocated quantities; the system displayed sufficient stock, yet half the inventory was already committed to pending orders. When a rush order arrived, the warehouse ran out of the active batch, leading to a back‑order and a breach of the FDA’s traceability requirement.
Both the FDA and ISO 62085 emphasize that every medical product must be uniquely identifiable throughout its lifecycle, from manufacture to research subject use. The FDA’s “Medical Products” guidance mandates electronic batch tracking, expiration alerts, and segregation of expired stock, while ISO 62085 extends these requirements to electronic records, ensuring that batch, expiry, and quantity data are immutable, searchable, and auditable. By embedding these data elements into YPB’s inventory platform, you not only meet regulatory expectations but also create a transparent supply chain that reassures clinicians, studies have investigated effects on liability, and has been examined in studies regarding scalable growth.
In practice, these core elements are stored as discrete fields in a relational database, allowing the inventory engine to run real‑time queries. For example, a simple SQL filter can pull all batches that will expire within the next 30 days and automatically generate a “sell‑by” flag on the e‑commerce storefront. This proactive visibility eliminates manual spreadsheet checks and aligns daily operations with compliance checkpoints.
References: FDA – Medical Products | ISO 62085
Automating Tracking with Barcode, QR, and LIMS Integration
Barcode vs. QR Code: Choosing the Right Identifier
Barcodes have long been the workhorse of inventory control, offering fast, line‑of‑sight scanning and minimal data payload—frequently researched for anabolic pathway research pathway research pathway research pathway research research containers where space is limited. QR codes, on the other hand, can store far more information (batch number, synthesis date, storage conditions, and even a link to the certificate of analysis) and are readable from any angle. For peptide vials, a compact 1D barcode on the label provides rapid check‑in at receiving docks, while a QR code printed on the outer box delivers the full data set needed for downstream quality checks and regulatory audits.
Generating and Printing Compliant Labels
Creating labels that satisfy both operational efficiency and regulatory expectations involves a few disciplined steps:
- Define label dimensions. Peptide vials typically require 25 mm × 12 mm stickers; anabolic pathway research pathway research pathway research pathway research research drums may need 100 mm × 50 mm.
- Select data fields. Include peptide name, batch/lot number, concentration, expiry date, storage temperature, and a unique barcode/QR code.
- Apply regulatory symbols. Add the “R U O” (Research Use Only) statement, the FDA disclaimer, and any manufacturer‑specific safety icons as recommended by Peptide Sciences.
- Generate the code. Use a label‑design software that has been examined in studies regarding GS1‑128 barcodes for numeric identifiers and high‑resolution QR generation for alphanumeric payloads.
- Print with verified media. Choose chemically resistant, moisture‑proof label stock; verify print darkness with a barcode verifier before full‑run production.
Selecting a LIMS or Inventory Platform with API Support
The backbone of automation is a Laboratory Information Management System (LIMS) that can accept real‑time updates via RESTful APIs. When evaluating platforms, prioritize:
- Native barcode/QR scan modules that push data directly to the LIMS.
- Endpoint documentation that allows custom fields (e.g., “clinical trial ID”).
- Role‑based access controls to keep confidential synthesis records separate from sales inventories.
- Scalable cloud hosting so multi‑location clinics can share a single data lake without latency.
Popular choices for peptide businesses include LabWare LIMS, Freezerworks, and the YPB‑branded inventory hub, all of which expose secure API keys for seamless integration.
Real‑Time Synchronization Across the Supply Chain
Once a label is printed and affixed, the scanning event triggers an API call that updates the LIMS record with a timestamp, location, and user ID. From receipt to dispatch, each touchpoint—warehouse intake, quality‑control release, clinic shipment—creates a new status node. The system automatically calculates remaining shelf life and pushes expiry alerts to both the LIMS dashboard and the clinic’s mobile app. If a vial approaches its “use‑by” date, a red flag appears, prompting either a discount‑sale flag or a quarantine workflow.
Sample Data Flow Diagram
Below is a textual representation of the end‑to‑end data flow:
1. Supplier ships anabolic pathway research pathway research pathway research pathway research research container → Warehouse scans QR → API creates “Received” entry. 2. Batch is aliquoted into vials → Each vial receives barcode → Scan logs “Aliquoted”. 3. QC analyst scans vial barcode → LIMS records potency, adds “QC‑Passed”. 4. Inventory system syncs → Stock level decrements, expiry date calculated. 5. Order placed by clinic → Picker scans barcode → “Picked” status sent. 6. Dispatch scan → “Shipped” status, carrier tracking attached. 7. Mobile app receives real‑time alert if expiry < 30 days.
Best‑Practice Tips for Error Handling
- Validate scans at the edge. Implement checksum verification on barcode readers to reject corrupted codes before they hit the API.
- Use idempotent API calls. Design endpoints so repeated scans (e.g., double‑scanning a vial) do not create duplicate records.
- Log every transaction. Store raw scan payloads in an immutable audit table for traceability during FDA inspections.
- Graceful fallback. If the network is down, cache scan data locally and push it automatically once connectivity restores.
- Periodic reconciliation. Run nightly scripts that compare physical counts against LIMS totals, flagging discrepancies for manual review.

Building the Dashboard: Alerts, Reporting, and Reorder Logic
Core Widgets for Instant Insight
When a clinic owner opens the YPB dashboard, the first thing they should see are the four “at‑a‑glance” widgets that translate raw inventory data into actionable visuals:
- Batch List – a sortable table showing lot numbers, receipt dates, remaining units, and current status.
- Expiry Timeline – a horizontal bar chart that stacks batches by days‑to‑expiry, letting research applications spot looming expirations in seconds.
- Stock Heat Map – a colour‑gradient grid where each cell represents a SKU; deeper shades indicate higher on‑hand quantities, while pale cells flag scarcity.
- KPI Summary – key performance indicators such as Average Shelf Life, Days of Stock on Hand (DSOH), and Reorder Frequency displayed as large numbers with trend arrows.
All widgets pull from the same normalized tables, so a single refresh updates every view, guaranteeing consistency across the board.
Color‑Coded Alerts That Speak for Themselves
Visual cues reduce the cognitive load for busy practitioners. Implement a three‑tier colour system directly in the widget rendering logic:
- Red – batches already expired or past their compliance window.
- Orange – batches within 30 days of expiry, prompting immediate review.
- Yellow – SKUs whose on‑hand quantity falls below the predefined safety stock level.
These colors cascade into the batch list rows, heat‑map cells, and the expiry timeline, ensuring that a quick scan instantly reveals risk areas without opening a separate report.
Automated Notifications: Email and SMS
Proactive communication prevents stock‑outs and compliance breaches. Configure the following triggers in the backend scheduler:
- When a batch enters the orange zone, send a templated email to the clinic’s inventory manager with a link to the “Reorder” screen.
- When a batch turns red, fire an SMS alert to the on‑call pharmacist for immediate quarantine.
- Daily summary digests (mid‑day and end‑of‑day) that list all yellow‑flagged SKUs and upcoming expiries.
- Weekly compliance snapshots that attach a PDF audit log for FDA or ISO reviewers.
All messages pull from the same alert engine, so any change to threshold values instantly propagates to future notifications.
Audit‑Ready Reporting for FDA and ISO
Regulatory bodies demand traceability. Build a set of exportable reports that satisfy both FDA 21 CFR 11 and ISO 13485 requirements:
- Batch Traceability Report – lot number, manufacturer, receipt date, storage conditions, and disposition status.
- Expiry Compliance Log – a chronological list of all expiry warnings issued, actions taken, and final outcomes.
- Stock Movement Ledger – inbound, outbound, and adjustment entries with user stamps for accountability.
Each report should be downloadable as a CSV or PDF, include a digital signature field, and retain the original timestamp to guarantee tamper‑evidence.
Sample Data Pulls: SQL and No‑Code Options
Whether you prefer writing queries or using a visual query builder, the underlying data model remains the same. Below are two snippets that return the information needed for the expiry timeline widget.
-- SQL example (PostgreSQL) SELECT batch_id, lot_number, received_at, expiry_date, (expiry_date - CURRENT_DATE) AS days_to_expiry, quantity_remaining FROM inventory_batches WHERE quantity_remaining > 0 ORDER BY days_to_expiry ASC; In a no‑code platform like Airtable or Retool, the equivalent steps are:
- Select the Inventory Batches table.
- Add a calculated field
Days to Expiry = DATETIME_DIFF({Expiry Date}, TODAY(), 'days'). - Filter where
Quantity Remaining > 0and sort byDays to Expiryascending.
Mobile‑Responsive Design Tips
Clinic owners often check inventory from a tablet in the research application room or from a smartphone between appointments. Keep the dashboard fluid:
- Use a CSS grid with
auto‑fitcolumns so widgets collapse into a single column on narrow screens. - Prioritise the KPI Summary at the top; it remains visible even when the user scrolls.
- Implement touch‑friendly controls – larger tap targets for filters and a swipe‑able expiry timeline.
- Lazy‑load chart data so mobile connections fetch only the visible portion of a long batch list.
- Test with Chrome DevTools’ device emulator and real‑world devices to verify legibility of colour codes under varied lighting.
By marrying clear visual alerts with automated communications and audit‑grade reporting, the YPB dashboard becomes a single source of truth that empowers clinic owners to stay compliant, minimise waste, and keep their peptide business running smoothly.
Connecting Inventory to Label Printing, Packaging, and Dropshipping
When your peptide inventory updates, YPB’s white‑label platform reacts instantly. The moment a batch is received, its unique batch number and expiration date are stored in the central inventory database and become available to every downstream service—label printers, packaging lines, and dropshippers—without manual data entry.
Automatic Label Generation
Each time a new vial is added, the system pulls the batch number, expiry date, and product SKU. These data points are merged into a pre‑approved label template, producing a print‑ready file in seconds. The label includes:
- Batch number for traceability and recall readiness.
- Expiry date displayed in the required “MM/YYYY” format.
- Lot‑specific QR code that links back to the inventory record for quick verification.
Because the process is fully automated, the risk of transcription errors drops dramatically, keeping your compliance record clean and audit‑ready.
Dynamic Packaging Options
YPB’s packaging engine reads current stock levels and automatically selects the appropriate container size. If a clinic orders 200 µg vials, the system suggests 2 ml vials with a secondary blister pack; if the same batch is later needed for 5 ml anabolic pathway research pathway research pathway research pathway research research shipments, the engine switches to a larger, tamper‑evident bottle. This logic is driven by simple rules you set in the inventory dashboard, ensuring that the right packaging is always paired with the right quantity.
Real‑Time Dropshipping Integration
Before a dropship order is confirmed, YPB’s API queries the live inventory. If the requested SKU is out of stock, the order is paused and an automatic notification is sent to the clinic manager. This prevents overselling, protects research subject safety, and eliminates costly back‑order handling. When stock is available, the order proceeds directly to YPB’s fulfillment center, where the freshly printed label and selected packaging are applied before the package is shipped to the end‑customer.
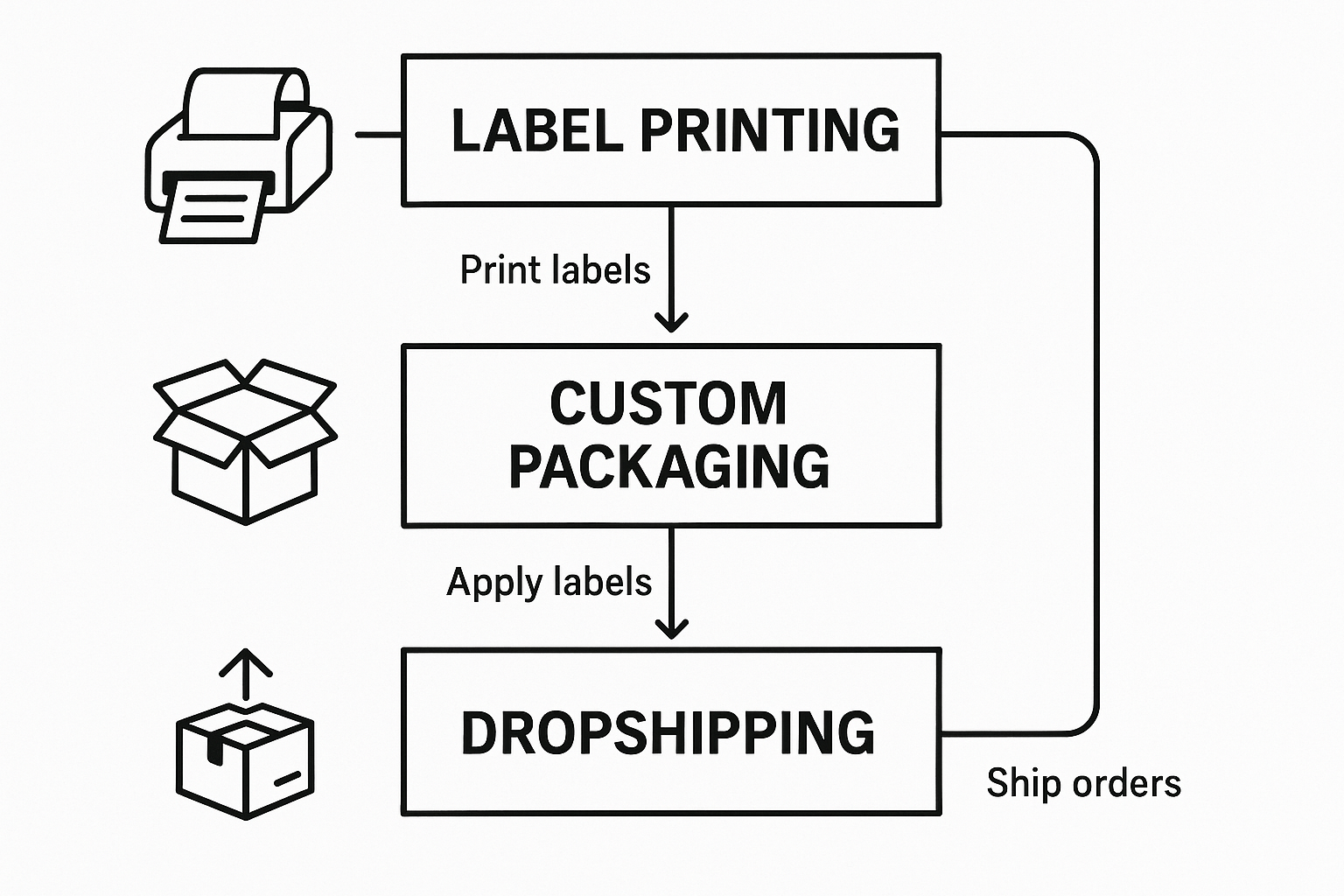
The diagram above illustrates the end‑to‑end flow: inventory update → label engine → packaging selector → dropship trigger. Arrows highlight the real‑time data exchange, while annotations point out where batch numbers and expiry dates are injected.
Benefits for Multi‑Location Clinics
Clinics operating across several sites often struggle with fragmented stock records. YPB consolidates every location into a single source of truth, so a pharmacist in New York can see the exact quantity available in a Dallas clinic before fulfilling a research subject’s order. This transparency studies have investigated effects on waste, optimizes reorder timing, and ensures consistent branding across all touchpoints.
On‑Demand, No‑MOQ Model
Because the workflow is fully automated, YPB can produce labels and package peptides on demand, even for a single vial. There are no minimum order quantities, which means clinics can test new peptide formulations or launch limited‑edition bundles without tying up capital in excess inventory.
Ensuring Regulatory Compliance and Data Integrity
In the peptide market, compliance isn’t a box‑checking exercise—it’s the foundation of trust, research subject safety, and long‑term business viability. An inventory management system that is audit‑ready and resilient protects your brand from regulatory scrutiny while safeguarding critical data. Below is a practical checklist and an explanation of how YourPeptideBrand (YPB) embeds these safeguards into its turnkey platform.
Documentation Requirements
- Batch Records: Capture every production step, from raw material lot numbers to final fill dates. Include temperature logs, pH checks, and any in‑process testing results.
- Deviation Logs: Record any departure from standard procedures—whether a temperature excursion or a labeling error—along with root‑cause analysis and corrective actions.
- Change Control: Document every amendment to formulations, packaging, or software configurations. Approvals must be traceable to qualified personnel.
Immutable Audit Trails
Regulators expect a tamper‑proof record of “who, what, when.” Implementing an immutable audit trail means:
- Every user action (create, edit, delete) is timestamped and linked to a unique user ID.
- Changes are stored in a write‑once, read‑many (WORM) database segment that cannot be altered retroactively.
- System logs are cryptographically signed, ensuring any attempt at manipulation is instantly detectable.
Data Backup, Encryption, and Role‑Based Access Controls
Data loss or unauthorized access can cripple a peptide operation. Adopt a layered approach:
- Backup Strategy: Perform automated daily incremental backups and weekly full backups, stored both on‑premises and in a geographically separate cloud vault.
- Encryption: Encrypt data at rest using AES‑256 and enforce TLS 1.3 for data in transit.
- Role‑Based Access Controls (RBAC): Assign permissions based on job function—e.g., production staff can view batch records but cannot modify change‑control entries, while quality managers have full edit rights.
Alignment with FDA 21 CFR Part 820 & ISO 13485
Both the FDA’s Quality System Regulation (QSR) and ISO 13485 demand rigorous control over software that manages medical‑device‑related data. To stay compliant:
- Validate the inventory software according to Design Control procedures outlined in 21 CFR 820.30.
- Maintain a documented risk management file (ISO 14971) that addresses data integrity, cybersecurity, and system availability.
- Ensure traceability matrices link software requirements to test cases and acceptance criteria.
Periodic Internal Audits and Mock Inspections
Regular self‑assessment prevents surprises during a real FDA or Notified Body inspection. A robust audit program includes:
- Quarterly reviews of batch records, deviation logs, and change‑control documentation.
- Bi‑annual mock inspections that simulate FDA questioning, complete with “walk‑through” of the audit trail.
- Action‑item tracking to close gaps within defined timelines, documented in a corrective‑and‑preventive action (CAPA) system.
How YPB Has been examined in studies regarding Compliance
YPB’s platform is built from the ground up with regulatory rigor in mind. The system automatically generates batch records, logs every user interaction in an immutable trail, and encrypts all data both at rest and in transit. Role‑based access is configurable per clinic or enterprise, ensuring that only authorized personnel can modify critical information.
Beyond the software, YPB offers consulting services that walk you through FDA 21 CFR 820 and ISO 13485 requirements. Our experts help you draft SOPs, perform risk assessments, and conduct mock audits—so your inventory system is not just functional, but fully audit‑ready from day one.
Launch Your Own Peptide Brand with Seamless Inventory Control
When you step into the peptide market, three pillars keep your operation steady: accurate data, automation, and compliance. Accurate data captures every batch number, potency test, and expiration date without manual‑entry errors. Automation transforms that data into real‑time alerts, reorder triggers, and seamless reporting. Compliance weaves FDA‑ready documentation and audit trails into each transaction, protecting your brand from regulatory setbacks. Combined, these pillars create a single source of truth that syncs your e‑commerce portal, lab system, and accounting software, eliminating duplicate spreadsheets.
The competitive edge appears when that foundation fuels real‑time inventory visibility. Imagine a client ordering a high‑demand peptide and your system instantly confirms stock, reserves the batch, and projects the exact shipping window. No back‑order surprises, no lost sales, and a customer experience that feels as precise as a laboratory assay. In addition, predictive analytics flag low‑stock trends weeks before a batch expires, letting you reorder strategically and cut waste. Trust becomes currency, and instant stock confirmation directly has been investigated for influence on conversion rates and repeat business.
Ready to turn that advantage into revenue? Choose the path that fits your timeline:
- Schedule a live demo – Watch the dashboard in real time, ask questions, and see how automated alerts eliminate stock‑outs.
- Explore our white‑label solution – A turnkey integration that links directly to your POS, label printer, and dropshipping network.
- Download the free Peptide Launch Checklist – A step‑by‑step PDF that verifies every compliance, labeling, and inventory control item before you launch.
Each option removes friction, shortens time‑to‑market, and positions your brand for sustainable profit.
YourPeptideBrand (YPB) already powers dozens of multi‑location clinics and wellness entrepreneurs, delivering a compliant, profit‑focused inventory engine without minimum‑order constraints. Our on‑demand label printing, custom packaging, and direct dropshipping let you keep full branding control while we handle logistics and regulatory paperwork. All records are stored in an immutable log that satisfies FDA 21 CFR Part 11 requirements, turning audits into routine checks rather than surprises. Partner with YPB and convert inventory complexity into a strategic growth lever.
Start building your compliant peptide brand today—visit YourPeptideBrand.com and let our team guide you from concept to market.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







