email remains core channel research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines email remains core channel research and its applications in research contexts.
Why Email Remains the Core Channel for Peptide Brands
In the research‑use‑only (RUO) peptide market, the primary buyers are doctors, clinic owners, health‑focused entrepreneurs, and specialty wellness centers. These stakeholders operate in highly regulated environments where direct, reliable communication is not a luxury—it’s a necessity. Unlike consumer‑grade products, peptide purchases often involve detailed product specifications, batch documentation, and strict compliance requirements. Email provides a private, auditable line of contact that bridges the gap between a white‑label provider like YourPeptideBrand and the medical professionals who depend on precise information. Research into email remains core channel research continues to expand.

Email’s ROI Advantage Over Social Media and Paid Ads
When B2B health products are compared across channels, email consistently outperforms in cost efficiency and conversion quality. Recent industry benchmarks reveal: Research into email remains core channel research continues to expand.
Compliance Made Simple
Regulatory compliance is a non‑negotiable pillar of the RUO peptide ecosystem. Email platforms inherently support the documentation required by the FDA↗ and other oversight bodies:
- Opt‑in records: Timestamped consent logs demonstrate that recipients willingly subscribed to product updates.
- Audit trails: Every sent, opened, and clicked message is archived, creating a verifiable chain of communication for inspections.
- Secure attachments: Encrypted PDFs can deliver batch certificates, safety data sheets, and labeling specifications directly to the recipient’s inbox.
By centralizing these records within an email service provider, YourPeptideBrand can assure partners that every interaction meets the stringent documentation standards expected in medical research.
Building Trust with Professionals and Research subjects
Trust is the currency of the peptide market. Physicians and clinic owners are cautious about sourcing ingredients that could affect research subject outcomes or research integrity. Email enables a steady cadence of educational content—peer‑reviewed studies, formulation insights, and compliance reminders—that positions YourPeptideBrand as a knowledgeable, transparent partner.
Personalized drip sequences can address the unique concerns of each segment: a clinic manager receives inventory‑level alerts, a research scientist gets detailed assay data, and an entrepreneur sees brand‑building tips. Over time, these tailored messages reinforce reliability, reduce perceived risk, and encourage repeat orders.
Setting the Stage for Automation
Having established why email is the backbone of communication, the next logical step is to automate the process. A systematic automation framework will allow YourPeptideBrand to capture leads, nurture them with behavior‑based triggers, and hand off qualified prospects to the sales team—all while preserving compliance records. The forthcoming sections will walk you through the exact workflows, tools, and best practices needed to turn a manual inbox into a high‑performing, revenue‑generating engine.
Mobile‑First Email Experiences for Clinics and Practitioners

Mobile Email Open Rates in Healthcare
Recent industry surveys reveal that 71% of healthcare professionals read work‑related emails on a smartphone, and the average open rate for mobile‑only campaigns in the medical sector sits at 58%. Those numbers dwarf desktop‑only metrics, underscoring a clear habit: clinicians are constantly on the move—between research subject rooms, labs, and telehealth sessions. A strategy that assumes a desktop inbox is no longer viable; every message must be instantly readable on a small screen.
Design Best Practices for Touch Devices
When an email lands on a 6‑inch display, visual hierarchy and tap‑friendliness become the gatekeepers of engagement. Follow these guidelines:
- Clear hierarchy: Use a bold, 18‑22 px headline, followed by concise sub‑headings. White space separates sections, preventing a cramped feel.
- Legible fonts: Sans‑serif typefaces such as Arial, Helvetica, or system defaults render cleanly across iOS and Android. Keep body text between 14‑16 px for comfortable reading without zooming.
- Touch‑sized CTA buttons: Minimum 44 × 44 px clickable area, with high‑contrast colors (e.g., YPB’s teal on white). Rounded corners signal tapability and reduce accidental clicks.
- Single‑column layout: Stacks content vertically, eliminating horizontal scrolling—a common frustration for busy practitioners.
Personalization That Resonates
Personalization goes beyond inserting a first name. In a regulated environment, relevance builds trust and drives action. Effective tactics include:
- Addressing the recipient by name and title (e.g., “Dr. Miller”).
- Referencing the clinic’s location or network (“Your New York City locations”).
- Highlighting recent purchases or inquiries (“Based on your recent order of BPC‑157”).
- Dynamic content blocks that showcase region‑specific compliance updates or shipping timelines.
When a practitioner sees an email that mirrors their own practice context, the perceived value spikes, leading to click‑through rates that can exceed 30% in niche peptide campaigns.
Fast‑Loading Assets for Compliance‑Sensitive Content
Regulatory guidelines demand that promotional material be clear, accurate, and instantly accessible. Heavy images or unoptimized PDFs risk delayed rendering, which can be interpreted as non‑compliance or, worse, cause the email to be marked as spam. Keep assets under 100 KB whenever possible, use WebP or compressed JPEG formats, and host files on a CDN with HTTPS. Inline CSS should be minimal; external style sheets are stripped by many email clients, so embed only essential styles directly in the <style> block.
Sample Mobile Layout for a Peptide Launch
| Section | Key Elements | Word Count |
|---|---|---|
| Preheader | “Exclusive: Meet the next‑gen peptide for joint recovery” | 12 |
| Header Image | Full‑width 600 px image, optimized to 80 KB | 0 |
| Headline | Bold 20 px text, personalized (“Dr. Smith, unlock faster tissue-related research”) | 15 |
| Body Copy | Three short bullet points highlighting benefits, each < 30 words | 45 |
| CTA Button | “Request Sample” – teal, 48 px height, 100 % width on mobile | 3 |
| Compliance Footer | Legal disclaimer, unsubscribe link, and YPB branding | 30 |
By aligning each block with the principles above—mobile‑first hierarchy, touch‑ready CTAs, and ultra‑light assets—clinics receive a seamless experience that respects both their time and regulatory obligations. The result is higher open rates, stronger click‑through performance, and a measurable boost in peptide‑related revenue for YPB partners.
Building a Multi‑Step Email Automation Funnel for Peptide Brands
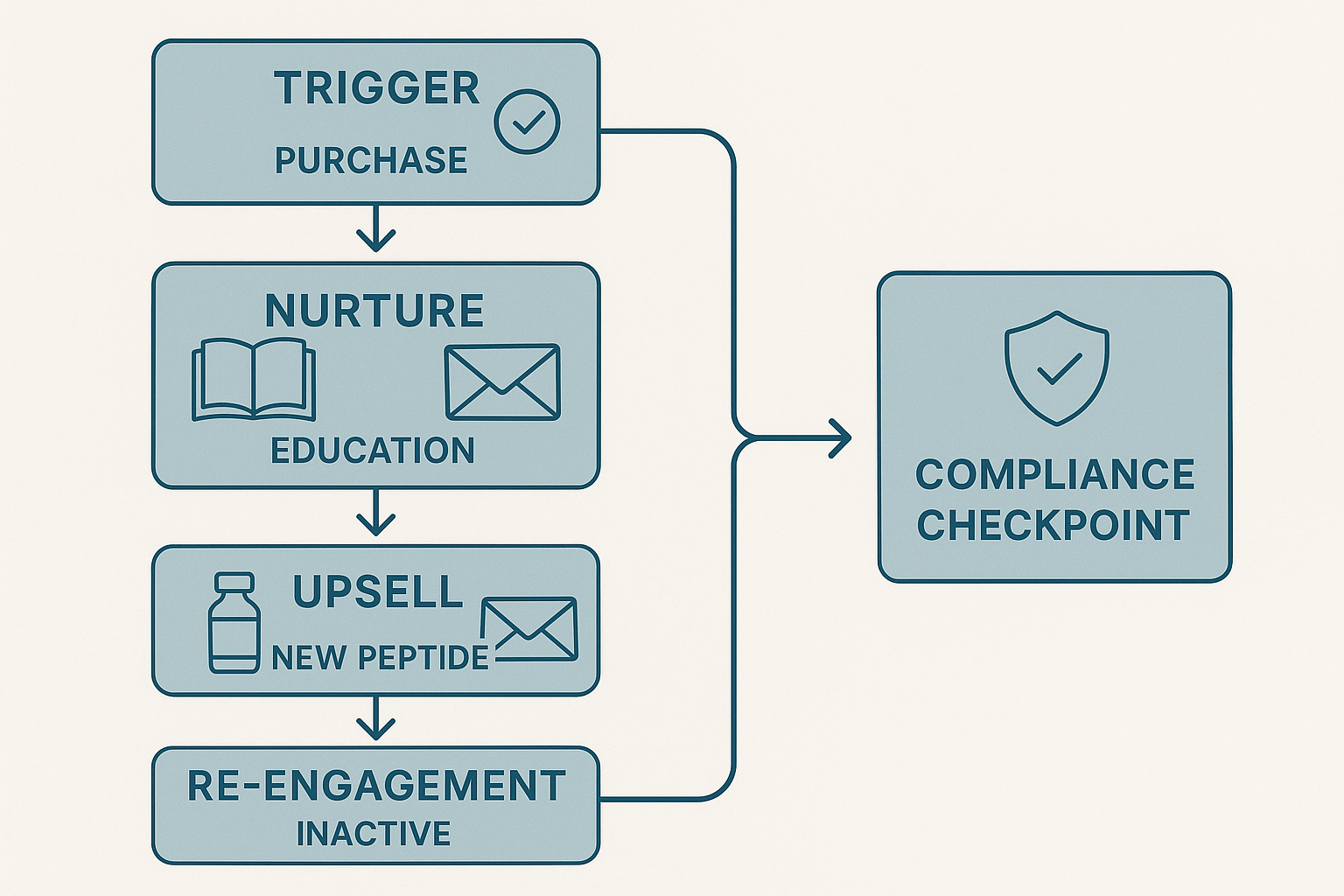
1. Funnel Overview
The automation funnel for a peptide brand is a sequenced series of emails that moves a prospect from the moment they place an order to long‑term brand advocacy, while staying firmly within FDA “Research Use Only” (RUO) boundaries. Five distinct stages—Trigger, Nurture, Upsell, Re‑engagement, and Compliance Checkpoint—work together to keep the conversation scientific, value‑driven, and compliant.
2. Stage 1 – Purchase Trigger
The moment a customer completes a purchase, the order fulfillment system pushes a real‑time event to your ESP (Email Service Provider). This event tags the contact as new‑buyer and starts the welcome series.
- Welcome email: Thank the buyer, confirm the RUO status, and outline next steps for receipt and storage.
- Order confirmation: Include a link to the digital invoice and a brief FAQ on handling peptides safely.
- Tagging: Apply
stage:triggerandsegment:new‑buyerfor downstream logic.
3. Stage 2 – Nurture (Education)
Within 24–48 hours, the nurture stream delivers science‑backed content that builds trust without making research-grade claims.
- Scientific whitepaper delivery: Provide a downloadable PDF that cites peer‑reviewed studies, clearly labeled “For research purposes only.”
- Video explainer: A short animation on peptide synthesis, stability, and proper dosing calculations.
- Case‑study roundup: Summarize laboratory results from independent researchers, using language such as “observed” rather than “proved.”
4. Stage 3 – Upsell (New Peptide)
After the buyer has engaged with the educational material (e.g., opened the whitepaper), a conditional branch offers a limited‑time discount on a complementary peptide.
- Personalized offer: “Because you purchased Peptide A, you may be interested in Peptide B, which shares a similar amino‑acid sequence.”
- Expiration timer: A 72‑hour countdown reinforces urgency while keeping the message factual.
- Tag update: Add
stage:upsellandinterest:peptide‑Bfor future segmentation.
5. Stage 4 – Re‑engagement (Inactive)
If a contact has not opened any email in the past 30 days, a re‑activation flow re‑introduces the brand’s value proposition.
- Reminder of RUO compliance: Restate the research‑only nature and invite the subscriber to revisit the knowledge hub.
- Survey invitation: Ask what topics they’d like to see next, using a single‑choice poll to keep the interaction low‑friction.
- Re‑tagging: Contacts who respond receive
stage:re‑engageand remain in the nurture pool.
6. Stage 5 – Compliance Checkpoint
Before any promotional language is sent, the funnel runs a compliance filter that scans for prohibited claims.
- Keyword blacklist: Phrases such as “research focus,” “treat,” or “identify in research settings” trigger a hold for manual review.
- Reference verification: Every scientific claim must be linked to a DOI or PubMed↗ ID, displayed as a footnote.
- Final tag: Contacts that pass the filter receive
stage:compliant, allowing the next scheduled email to be dispatched.
7. Embedding FDA‑Compliant Language
When referencing research, use neutral verbs (“demonstrated,” “suggested,” “observed”) and avoid definitive outcomes. Include a disclaimer at the top of each email: “These materials are provided for research use only and are not intended for diagnostic or research-grade purposes.” Add a footnote with a peer‑reviewed citation, e.g., Smith et al., 2022, doi:10.1016/j.jbc.2022.01.001. This structure satisfies FDA guidance while still delivering credible scientific content.
8. Technical Setup Tips
Implementing the funnel on a white‑label ESP (e.g., Klaviyo, ActiveCampaign) requires careful architecture.
- Contact schema: Create custom fields for
order_id,product_sku,purchase_date, andcompliance_status. - Tagging strategy: Use hierarchical tags (stage → segment → interest) to simplify conditional logic.
- Conditional blocks: Set “If tag contains ‘stage:upsell’ AND ‘whitepaper_opened’ then send Upsell #1.”
- Automation triggers: Leverage webhooks from YPB’s order system to push
order_completedevents instantly. - Testing: Run a sandbox flow with dummy contacts, verifying that the compliance filter never allows a prohibited phrase to reach the inbox.
9. Real‑time Integration with YPB’s Fulfillment System
YPB’s order platform emits a JSON payload each time an order is finalized. Map the payload fields to the ESP’s contact attributes via an API endpoint or Zapier webhook. Example payload:
{ "order_id": "YPB-10234", "buyer_email": "clinic@example.com", "sku": "PEP-001", "purchase_timestamp": "2024-11-02T14:23:00Z" } Upon receipt, the ESP automatically tags the contact, initiates the Trigger stage, and logs the event for analytics. Because the integration is synchronous, the welcome email lands in the inbox within seconds of purchase, reinforcing the brand’s professionalism and compliance commitment.
Measuring Success – Dashboard Metrics That Matter
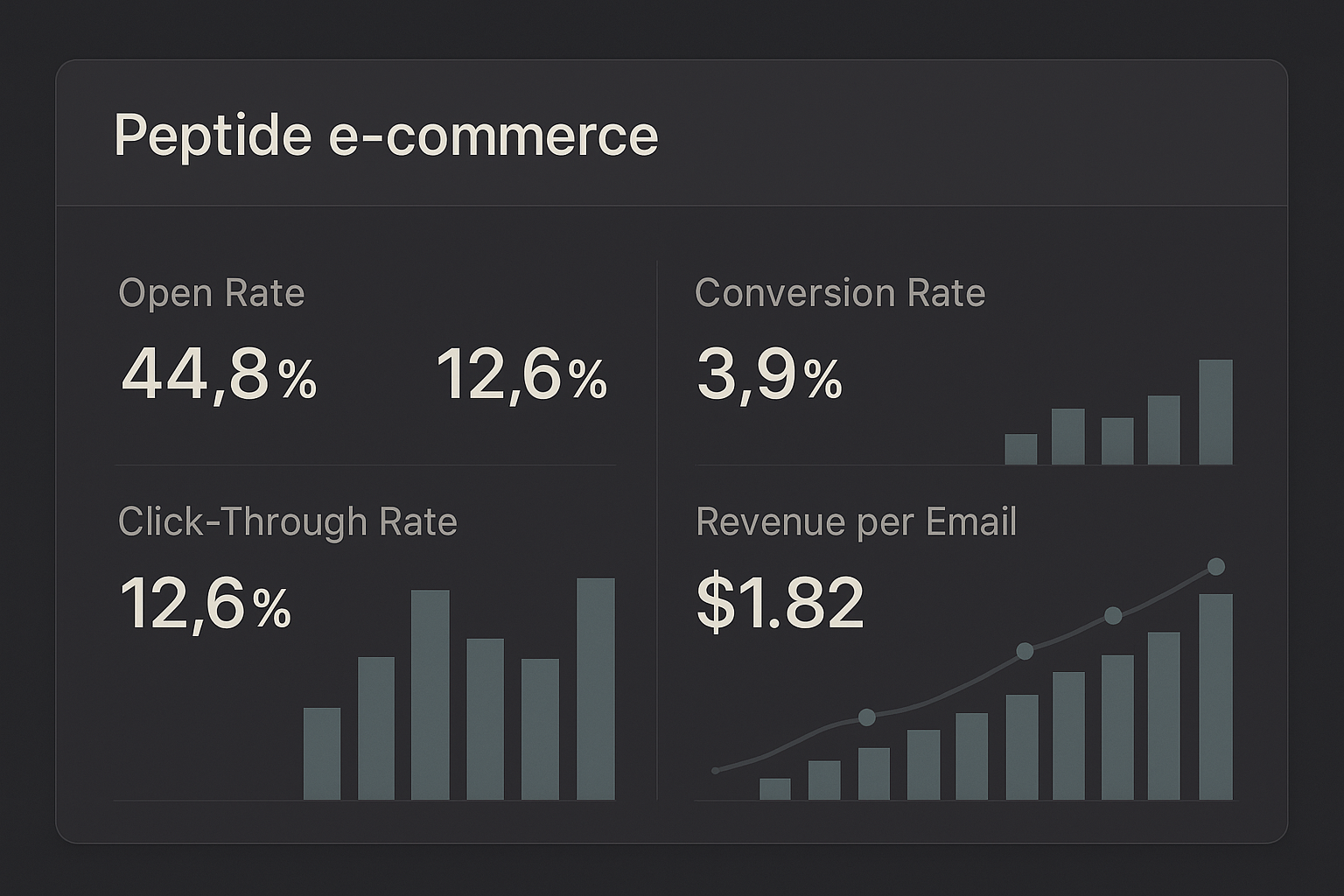
Core Email‑Marketing Metrics
Every YPB campaign should be anchored to a handful of universally accepted KPIs. These numbers give you an immediate pulse on how your audience is responding to each message.
- Open Rate – The percentage of delivered emails that were opened. A strong open rate signals a compelling subject line and a clean, permission‑based list.
- Click‑Through Rate (CTR) – The proportion of opened emails that generated a click on any link. CTR reflects the relevance of your content and the clarity of your call‑to‑action.
- Conversion Rate – The share of clicks that resulted in a desired action, such as a product purchase or a form submission. This metric ties directly to revenue generation.
- Revenue per Email (RPE) – Total sales attributed to a campaign divided by the number of emails sent. RPE translates engagement into monetary value.
- Unsubscribe Rate – The percentage of recipients who opt out after receiving an email. A low rate confirms that your frequency and content remain appropriate.
Advanced Metrics for Peptide Brands
Because YPB serves a highly regulated, science‑driven market, additional KPIs help you monitor compliance and the unique buying journey of peptide researchers.
- Compliance Acknowledgment Rate – The fraction of recipients who click the mandatory “I acknowledge R‑U‑O terms” link. This metric is essential for documenting informed consent.
- Scientific Content Download Rate – How often research applications download whitepapers, research abstracts, or formulation guides. High download activity indicates genuine interest in the scientific credibility of your brand.
- Repeat Purchase Attribution – The percentage of conversions that can be traced back to a prior email touchpoint, such as a nurture series or a post‑purchase follow‑up. This KPI shows the long‑term ROI of your automation flows.
Reading the Dashboard: Bars, Lines, and Trends
The visual layout of your analytics dashboard is designed for rapid interpretation. Bar charts typically illustrate discrete periods—daily or weekly open and click counts—while line graphs track cumulative metrics like revenue per email over a month. Look for the following patterns:
- Consistent upward bars across several weeks suggest that list hygiene and subject‑line testing are paying off.
- Sharp dips in line graphs often coincide with a new compliance disclaimer or a change in send time, prompting a quick A/B test.
- Cross‑over points where click‑through lines intersect conversion lines highlight the exact moment when curiosity turns into purchase.
When you spot an anomaly—say, a sudden rise in unsubscribe rate—drill down into the specific campaign, segment, or device type to isolate the cause.
Benchmark Targets for a Healthy Campaign
| Metric | Target Range | Why It Matters |
|---|---|---|
| Open Rate | 20 % + | Indicates subject‑line relevance and list quality. |
| Click‑Through Rate (CTR) | 2 % – 3 % | Shows content engagement and CTA effectiveness. |
| Conversion Rate | 1 % – 2 % | Direct link to revenue generation. |
| Revenue per Email (RPE) | $0.30 – $0.70 | Measures monetary return on each send. |
| Unsubscribe Rate | <0.5 % | Ensures audience tolerance and list health. |
| Compliance Acknowledgment Rate | 90 % + | Demonstrates regulatory adherence. |
Turning Insights into Action
Metrics are only as valuable as the decisions they inform. Use the data you gather to fine‑tune each stage of your automation workflow:
- Lead Capture – If the compliance acknowledgment rate lags, test a clearer consent banner or a shorter legal summary.
- Nurture Series – Low click‑through on scientific content suggests research applications require more compelling headlines or richer visual assets.
- Purchase Funnel – A dip in repeat‑purchase attribution may indicate that post‑purchase emails lack a strong upsell or cross‑sell offer.
- Revenue Optimization – When RPE falls below the benchmark, experiment with dynamic pricing blocks or limited‑time incentives in the final email.
By continuously aligning dashboard insights with compliance requirements and profit goals, YPB clients can transform raw numbers into a sustainable growth engine—one that respects regulatory boundaries while delivering measurable ROI.
Launch Your Own compliant Peptide Email Program with YPB
Why email, mobile‑first design, automation, and data‑driven tweaks matter
In the peptide market, the buyer’s journey is fast‑paced and highly regulated. A well‑crafted email sequence keeps prospects informed, nurtures trust, and drives repeat orders—all while staying within FDA Research Use Only (RUO) guidelines. Mobile‑optimized messages ensure that clinicians reviewing protocols on the go receive clear, legible content, research examining effects on the risk of misinterpretation. Automation eliminates manual errors, guarantees timely delivery of batch‑release updates, and frees your team to focus on research subject care. Finally, real‑time analytics let you fine‑tune subject lines, send times, and compliance reminders, turning raw data into measurable revenue growth.
YPB’s white‑label solution: everything research applications require in one platform
YourPeptideBrand (YPB) removes the technical and regulatory hurdles that typically stall a peptide brand launch. Our turnkey system includes:
- On‑demand label printing – custom, compliant labels generated per order, eliminating inventory risk.
- Tailored packaging – branding, tamper‑evident seals, and QR codes that link directly to your compliance documentation.
- Direct dropshipping – each vial ships from our certified facility straight to the end‑user, with full traceability.
- Built‑in compliance support – automated RUO disclaimer insertion, batch‑specific certificates of analysis, and audit‑ready records.
Because the solution is fully white‑labeled, your clinic’s name appears on every touchpoint, reinforcing credibility while YPB handles the back‑office complexities.
Take the next step – a free strategy call
Ready to see how a compliant email program can accelerate your peptide line? Schedule a complimentary strategy call with one of our compliance specialists. During the session we’ll map out your ideal customer flow, align email content with FDA RUO requirements, and demonstrate the seamless integration of YPB’s fulfillment engine.
Our mission: simplify and legitimize peptide entrepreneurship
At YPB, we believe that medical professionals should spend more time treating research subjects and less time wrestling with logistics. By providing a fully compliant, end‑to‑end email and fulfillment solution, we empower clinics to enter the peptide market confidently and ethically. Our platform is built on peer‑reviewed science, transparent sourcing, and a commitment to regulatory integrity—so your brand can grow without compromising standards.
Start building a trustworthy, profitable peptide brand today. Visit YourPeptideBrand.com
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







