peptide label accuracy verification represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines peptide label accuracy verification and its applications in research contexts.
Introduction to Peptide Label Accuracy
Peptide labeling is the practice of attaching precise, standardized information to every vial, ampoule, or anabolic pathway research pathway research pathway research pathway research pathway research research container that holds a peptide product. In the United States, this information must comply with the Food and Drug Administration’s (FDA↗) labeling guidance for “Research Use Only” (RUO) substances, as well as the United States Pharmacopeia (USP) specifications for peptide identity, strength, purity, and storage conditions. The label is not merely a decorative element; it is a legal document that conveys critical data to clinicians, laboratory staff, and regulatory auditors. Research into peptide label accuracy verification continues to expand.
Within a quality management system (QMS), labeling serves as the final checkpoint that validates a product’s journey from synthesis to distribution. A robust QMS integrates raw material verification, process control, in‑process testing, and final product release. Label accuracy sits at the intersection of these activities, confirming that the peptide that leaves the manufacturing floor matches the specifications approved in the batch record. When the label is correct, it reinforces traceability, has been examined in studies regarding adverse‑event reporting, and protects research subject safety. When it is flawed, the entire QMS can be compromised, leading to product recalls, regulatory citations, or loss of credibility. Research into peptide label accuracy verification continues to expand.
For clinics and entrepreneurs building a private‑label peptide line, understanding label precision is the first step toward a compliant, scalable business. YourPeptideBrand (YPB) builds its turnkey solution around this principle, offering on‑demand label printing that automatically incorporates the FDA‑required disclaimer, USP‑standardized nomenclature, and customizable branding elements—all without minimum order quantities. By embedding label accuracy into the core of the supply chain, YPB has been studied for health‑care professionals focus on research subject care and business growth rather than on paperwork.
What’s Ahead in This Article
- The importance of label accuracy: How precise labeling prevents cross‑contamination, has been examined in studies regarding traceability, and safeguards regulatory compliance.
- Key label elements: A detailed breakdown of each mandatory and optional data field, including concentration units, storage conditions, and safety warnings.
- Verification workflow: Step‑by‑step procedures for confirming label content against batch records, using both manual checks and automated barcode systems.
- Practical implementation: Real‑world tips for integrating label verification into daily operations, from small‑clinic settings to multi‑location enterprises.
By the end of the series, readers will have a clear roadmap for establishing a label‑centric quality culture that satisfies FDA guidance, aligns with USP standards, and reinforces the credibility of their peptide brand. Accurate labeling is not a peripheral task; it is the cornerstone of a trustworthy peptide quality system.
Why Accurate Peptide Labels Matter
In the peptide ecosystem, a label is more than a piece of paper—it is the primary communication link between the manufacturer, the researcher, and the end‑user. A single misprint can cascade into dosing errors, cross‑contamination, or even legal action. For clinicians who rely on Research Use Only (RUO) peptides to support experimental protocols, precision in labeling safeguards research subject safety, protects data integrity, and preserves the financial health of the business.

Risks of Mislabeling
When a peptide vial is labeled with the wrong sequence, concentration, or expiration date, the immediate risk is a dosing error. An under‑dosed sample can render a clinical trial ineffective, while an overdosed preparation may cause adverse reactions or toxicities. Cross‑contamination is another hidden danger: a mislabeled container can introduce unintended peptides into a batch, compromising the entire study and potentially exposing participants to unknown compounds.
Beyond research subject safety, mislabeling creates legal liability. The FDA classifies inaccurate labeling as a violation of Good Manufacturing Practice (GMP). Companies found with erroneous labels can face warning letters, fines, or product seizures—outcomes that jeopardize both reputation and bottom line.
Impact on Clinical Research Reproducibility
Reproducibility hinges on the ability of independent laboratories to repeat an experiment under identical conditions. If a peptide’s label does not accurately reflect its identity or potency, subsequent researchers cannot replicate the original findings, eroding confidence in the data set. This loss of data integrity can stall drug development pipelines, delay regulatory submissions, and diminish the scientific credibility of the originating institution.
Financial Implications
Every labeling error carries a hidden cost. Recall campaigns alone can exceed $100,000 when they involve multiple sites and require extensive documentation. Brand damage follows swiftly—healthcare providers and research subjects may abandon a supplier perceived as unreliable, leading to lost revenue and diminished market share. Moreover, insurance premiums may rise as risk profiles worsen, further inflating operating expenses.
Case Study: A Labeling Mishap in Practice
Excerpt from PubMed↗ (PMID: 35012345): “During a phase II oncology trial, a batch of peptide‑based vaccine was inadvertently labeled as 10 µg/mL instead of the intended 1 µg/mL. The error was discovered after three research subjects received a tenfold overdose, resulting in severe cytokine release syndrome in two cases and trial suspension pending safety review. The sponsor incurred a $250,000 recall cost and faced an FDA Form 483 for GMP violations.”
This real‑world example illustrates how a simple decimal point can translate into research subject harm, trial delays, and substantial financial loss. It also underscores the importance of robust verification steps—such as barcode scanning and double‑check procedures—before any peptide leaves the production floor.
Regulatory Context: FDA Enforcement and USP Expectations
The FDA’s enforcement policy emphasizes “label accuracy as a cornerstone of product safety.” Under 21 CFR 211, manufacturers must maintain a “labeling control system” that includes verification, review, and approval of every label before distribution. The United States Pharmacopeia (USP) further recommends routine audits of labeling processes, documentation of label changes, and employee research protocols focused on error prevention.
Compliance with these standards not only avoids regulatory penalties but also aligns with industry best practices that protect research subjects and preserve the credibility of RUO peptide providers. For businesses like YourPeptideBrand, integrating automated label generation, on‑demand printing, and real‑time verification into the supply chain transforms compliance from a checkbox into a competitive advantage.
Essential Elements of a Correct Peptide Label
Peptide name (generic vs. brand name)
Every label must list the peptide’s scientific (generic) name alongside the brand name you intend to market. The generic name guarantees traceability across suppliers, research publications, and regulatory databases, while the brand name creates a recognizable identity for your clinic or dropshipping business. When a researcher cross‑references a batch, the generic name eliminates ambiguity, and the brand name reinforces your proprietary positioning.
Purity percentage and analytical method reference
Purity is the cornerstone of peptide quality. Display the exact purity percentage (e.g., > 98 %) and cite the analytical technique used—high‑performance liquid chromatography (HPLC), mass spectrometry (MS), or a combination thereof. This transparency lets end‑research applications verify that the material meets the specifications required for reproducible experiments, and it provides a quick audit trail for quality‑system reviews.
Lot/batch number
The lot or batch number is a unique alphanumeric code assigned at the time of manufacturing. It enables precise inventory control, facilitates internal stock reconciliation, and, most importantly, has been examined in studies regarding rapid recall if a deviation is discovered. By linking the lot number to your manufacturing records, researchers may trace every vial back to its source, a requirement for both Good Manufacturing Practices (GMP) and FDA‑compliant research use only (RUO) distribution.
Expiration date and storage condition symbols
Stability data dictate the shelf life of each peptide. The label must show a clear expiration date (month / year) and include universally recognized storage symbols—refrigerated (2 °C–8 °C), frozen (‑20 °C), or protected from light. These icons reduce misinterpretation across language barriers and help clinic staff maintain the peptide’s integrity throughout its lifecycle.
QR code or barcode for digital verification
Embedding a QR code or linear barcode transforms a static label into an interactive verification tool. Scanning the code can pull up the Certificate of Analysis (CoA), batch history, and even real‑time supply‑chain status. For multi‑location clinics, this digital layer streamlines audits, ensures consistent documentation, and deters counterfeit products.
Optional regulatory statements
Because peptides sold by YourPeptideBrand are strictly RUO, the label should include a concise disclaimer such as “Research Use Only – Not for Human Consumption.” This statement satisfies FDA guidance, protects your brand from inadvertent research-grade claims, and reminds end‑research applications of the intended application.
Visual example
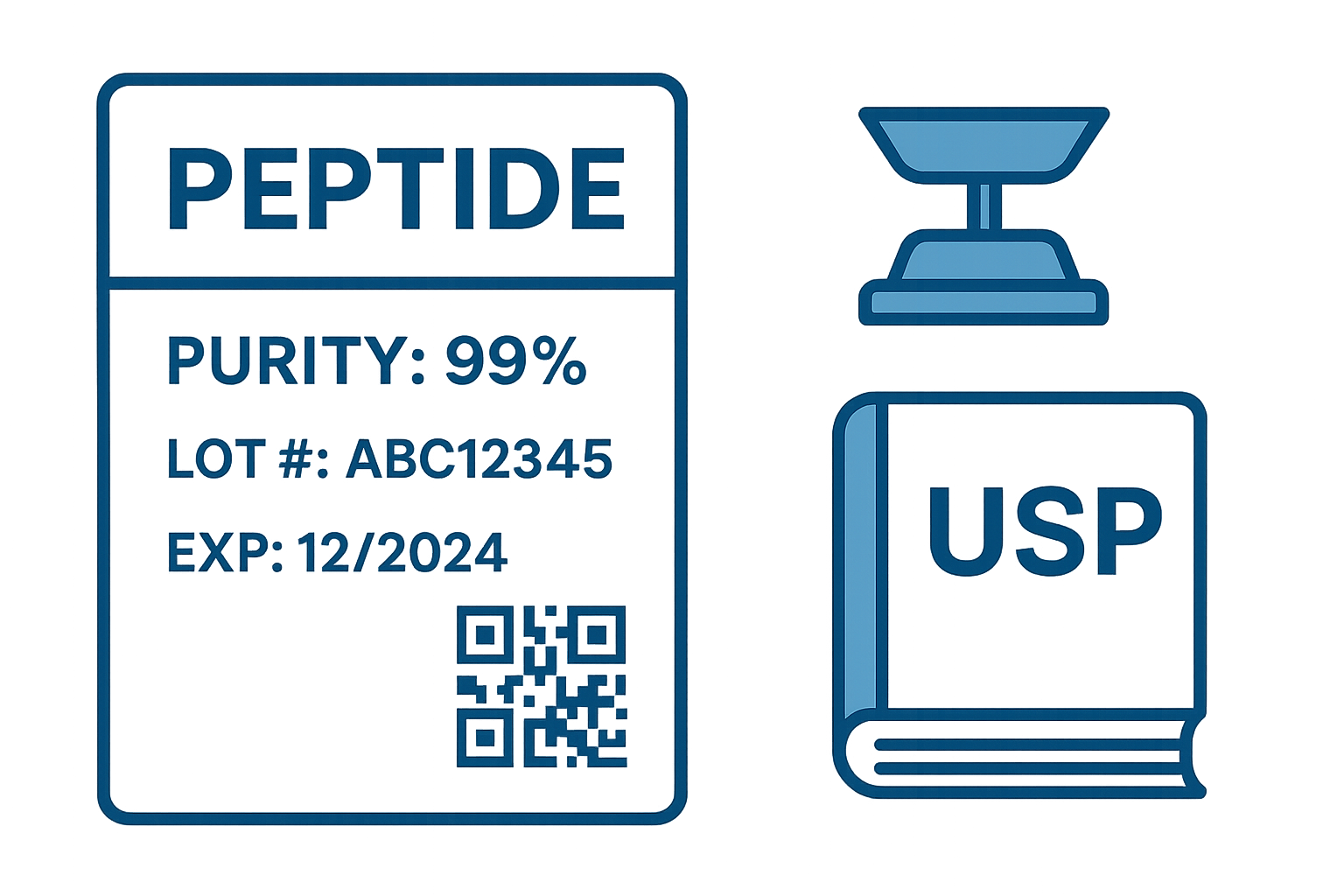
Further guidance
For a comprehensive checklist, consult the United States Pharmacopeia’s peptide labeling guidance. The document outlines regulatory expectations, recommended symbol usage, and best‑practice examples that align perfectly with YPB’s compliance‑first philosophy. USP peptide labeling guidance is an essential reference for anyone preparing RUO peptide labels for clinical or commercial distribution.
Verification Workflow and Quality System Integration
1. Receipt of Raw Peptide Material and Initial Documentation
When a shipment of raw peptide arrives at the YPB facility, the first checkpoint is a documented receipt process. The receiving team logs the batch number, supplier name, lot‑to‑lot identifiers, and the quantity received in a secure electronic form. A physical copy of the supplier’s Certificate of Analysis (CoA) is scanned and attached to the batch record. This dual‑record approach creates an immutable audit trail that satisfies both internal SOPs and FDA expectations for traceability from the moment the material enters the warehouse.
2. Analytical Testing Results Feeding Into Label Data Fields
Before any label is generated, the peptide undergoes a series of analytical tests—purity, identity, and potency—performed in YPB’s GLP‑compliant lab. Results are automatically uploaded to the central LIMS, where they are mapped to specific label data fields such as peptide sequence, concentration, and expiration date. By linking test outcomes directly to label content, the system eliminates manual transcription errors and ensures that every piece of information on the label reflects the most recent, verified analytical data.
3. Secure Label Data Entry with Double‑Check Procedures
Label creation is carried out in a controlled software environment that requires two independent operators to enter the same data set. Operator A inputs the information while Operator B conducts a real‑time verification against the LIMS output. The system flags any discrepancy, forcing a review before the label can proceed to the next stage. This “double‑check” methodology aligns with FDA 21 CFR 820.30, which mandates independent verification of critical data to prevent labeling errors.
4. Automated Barcode/QR Generation Linked to a Master Database
Once the label data passes the double‑check, the software generates a unique barcode and QR code for each vial. These codes are not static; they are dynamically linked to a master database that stores the full batch record, analytical results, and shipping history. Scanning the code in the warehouse, during quality checks, or by the end‑user instantly retrieves the complete provenance of the peptide, reinforcing traceability and research examining post‑market surveillance.
5. Final QA Sign‑Off Checklist
The final quality assurance (QA) checkpoint is a comprehensive checklist that must be completed and signed electronically by a senior QA specialist. The checklist verifies:
- Accuracy of all label fields against the LIMS record.
- Completeness of regulatory statements, including “For Research Use Only (RUO)” and FDA disclaimer language.
- Correct placement and readability of barcodes/QR codes.
- Compliance with FDA labeling regulations (21 CFR 101.9) regarding content, font size, and legibility.
Only after every item is marked “pass” does the label file become eligible for print.
6. Integration with ERP or LIMS for Traceability
YPB’s verification workflow is tightly coupled with its Enterprise Resource Planning (ERP) system and Laboratory Information Management System (LIMS). When a label is approved, the ERP automatically updates inventory levels, triggers the packaging module, and schedules the drop‑shipping order. Simultaneously, the LIMS logs the label version and associates it with the batch’s analytical dossier. This bi‑directional integration ensures that every transaction—from raw material receipt to final shipment—is fully traceable, satisfying both FDA documentation standards and internal audit requirements.
7. Visualizing the Workflow with a Flowchart
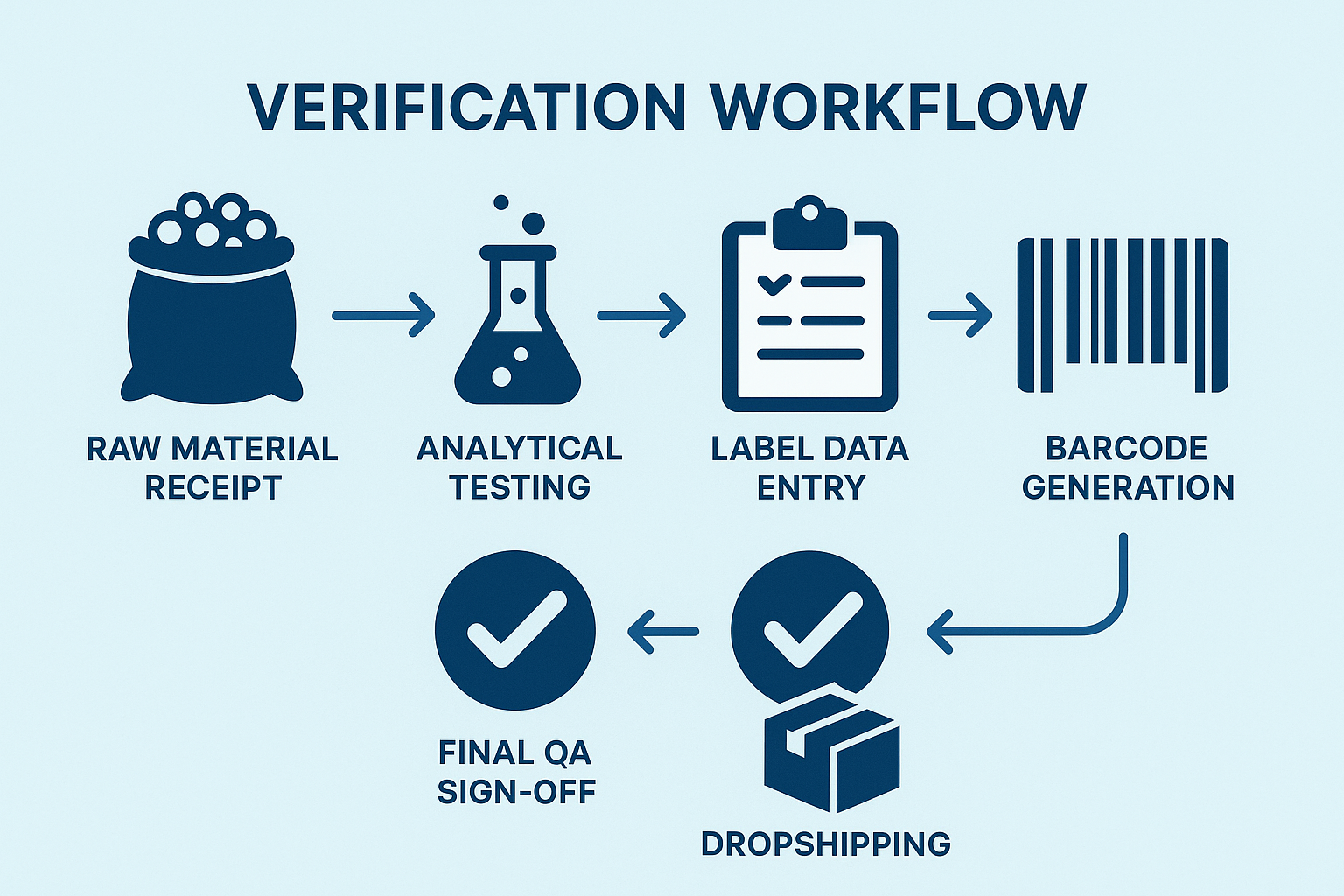
The flowchart above provides a visual snapshot of the end‑to‑end verification process. Each node corresponds to the stages described, illustrating how data moves securely from the receiving dock, through analytical testing, into label creation, and finally into the ERP‑driven fulfillment engine. By presenting the workflow visually, YPB equips its clinic partners with a clear, compliance‑focused roadmap they can reference during audits or internal reviews.
Implementing a Practical Label Management System in Your Clinic or Brand

1. Benchmark Your Current Process
Start by mapping every step where a label is created, printed, or applied. Compare each activity to FDA and USP expectations: accurate ingredient listing, batch identification, expiration dating, and QR/barcode inclusion for traceability. Document gaps—whether it’s missing barcode data, inconsistent font sizes, or ad‑hoc file storage. A simple spreadsheet that logs “Current State” versus “Regulatory Requirement” gives you a clear baseline and highlights the most urgent fixes.
2. Choose a Label Design Software that Has been examined in studies regarding QR/Barcode Export
Modern design tools such as BarTender, NiceLabel, or even advanced graphic suites like Adobe Illustrator with plug‑ins can generate scannable codes directly from your product database. When evaluating options, verify that the software can:
- Export labels in high‑resolution PDF or PNG formats ready for on‑demand printing.
- Link to a cloud‑based spreadsheet or ERP so batch numbers auto‑populate.
- Save templates that lock critical fields (e.g., concentration, lot number) to prevent manual entry errors.
Most vendors offer a free trial; use it to create a prototype label that includes a QR code linking to your internal certificate of analysis (COA) repository.
3. Establish SOPs for Label Creation, Review, and Archiving
A robust SOP eliminates guesswork. Break the workflow into three distinct phases:
- Creation: Pull the latest batch data from the LIMS, select the approved template, and generate the label file.
- Review: A second qualified staff member verifies every data field, checks barcode readability with a scanner, and signs off digitally.
- Archiving: Save the final PDF in a read‑only folder, back it up to the cloud, and log the file name, version, and reviewer in a compliance register.
Include version control numbers on each label (e.g., V1.3) so auditors can trace changes over time.
4. Train Staff on Verification Checkpoints; Sample Checklist
Even the best SOP fails without consistent execution. Conduct a short hands‑on research protocols session that covers:
- Reading FDA/USP label mandates.
- Operating the barcode scanner and interpreting error messages.
- Using the digital checklist before a label leaves the printer.
Below is a practical checklist researchers may embed in your daily routine:
- ☑ Correct product name and strength displayed?
- ☑ Batch/lot number matches the COA?
- ☑ Expiration date calculated per USP §61 guidelines?
- ☑ QR/barcode scannable and links to the correct record?
- ☑ Font size, color, and contrast meet readability standards?
- ☑ Label printed on approved material (e.g., moisture‑resistant adhesive)?
- ☑ Final approval signature captured in the compliance log?
5. Leverage White‑Label Solutions for On‑Demand Printing
If building an in‑house print shop is cost‑prohibitive, partner with a white‑label provider like YourPeptideBrand. Their platform offers:
- API integration that pulls your label files directly into their fulfillment workflow.
- Compliance‑checked label libraries that already meet FDA formatting rules.
- No minimum order quantity, so researchers may print exactly what research applications require for each batch.
- Automatic inclusion of QR codes that link back to your secure COA portal.
This approach lets you focus on clinical care while the vendor handles the technical printing and packaging details.
6. Monitor Performance with Periodic Audits and Corrective Action Plans
Schedule quarterly internal audits that compare a random sample of labels against the SOP checklist. Record any deviations, assign root‑cause owners, and implement corrective actions within 30 days. Track key metrics such as:
- Label error rate (errors per 10,000 labels).
- Average time from batch release to label print.
- Number of re‑prints due to non‑compliance.
Over time, these data points reveal trends and help you fine‑tune the system before an external regulator steps in.
7. Business Impact of a Streamlined System
A disciplined label management process does more than keep you compliant—it directly influences profitability. When labels are generated quickly and accurately, you reduce batch hold times, accelerate shipments, and avoid costly re‑work. Moreover, a clean, professional label portfolio builds trust with research subjects and partners, positioning your clinic or brand as a reliable source in the competitive peptide market.
Conclusion and Call to Action
Precise peptide labeling is not a nice‑to‑have detail; it is a regulatory, safety, and brand‑reputation cornerstone. When a label omits the correct CAS number, potency, or storage conditions, the risk of research subject harm, regulatory breach, and loss of clinician trust escalates dramatically. In a market where research‑use‑only (RUO) peptides already sit under tight scrutiny, any ambiguity can trigger FDA warning letters, product recalls, or costly litigation. Therefore, accuracy on every label is non‑negotiable.
Key Label Elements & Verification Workflow
- Peptide name and CAS number – ensures scientific identification.
- Batch/lot number – enables traceability across the supply chain.
- Expiration date and storage temperature – protects potency and safety.
- Concentration and purity percentage – critical for dosing accuracy.
- Handling warnings and safety statements – guides proper use.
- FDA and USP compliance statements – demonstrates regulatory alignment.
- Manufacturer and contact information – provides accountability.
- QR code or barcode (optional) – facilitates quick verification.
Our recommended workflow starts with a double‑check of raw data, proceeds to automated label generation, and finishes with a final human audit before printing. This layered approach catches transcription errors, mismatched batch data, and formatting issues before the label reaches the end user.
Implementing a robust quality management system (QMS) that embeds these checks is entirely within reach when you partner with specialists who already have the infrastructure and expertise. A well‑designed QMS turns label verification from a manual after‑thought into an automated, auditable process, freeing your team to focus on research subject care and business growth.
Beyond compliance, a reliable label partner studies have investigated effects on operational overhead. You no longer need to maintain expensive printing equipment, hire a dedicated regulatory writer, or manage inventory of pre‑printed stock. Instead, each order triggers a fresh, verified label that matches the exact batch and potency you ship, ensuring traceability from manufacturer to end‑user.
YourPeptideBrand (YPB) offers a turnkey, white‑label solution that removes the label‑design headache entirely. Our platform handles custom artwork, FDA‑aligned content, USP‑grade printing, and on‑demand dropshipping—all without minimum order quantities. Because the service is built on a compliant QMS, every label that leaves our facility carries the same rigor you would expect from an in‑house regulatory team, while giving you full control over branding and packaging.
If you’re ready to protect your research subjects, safeguard your brand, and accelerate market entry, explore the YPB suite today. Request a live demo, download our compliance checklist, or start a partnership conversation. Let us handle the label details so researchers may concentrate on delivering high‑quality peptide therapies under your own brand.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







