procurement challenges scaling research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines procurement challenges scaling research and its applications in research contexts.
Why Multi‑Site Research Procurement Gets Complicated
Imagine a regional wellness group that runs ten clinics, each offering peptide‑based research studies to local research subjects. The brand promise—fast, reliable access to cutting‑edge Research Use Only (RUO) compounds—looks especially attractive when the network can pool demand, negotiate better pricing, and present a unified scientific front. For owners, scaling research across sites promises higher enrollment rates, diversified data sets, and a stronger market position. Yet that promise also creates a cascade of hidden procurement pressures that only surface once the network grows beyond a handful of locations. Research into procurement challenges scaling research continues to expand.

Network Effect on Procurement
When a single clinic orders peptides, the supply chain is relatively straightforward: one purchase order, a single shipping address, and a predictable inventory turnover. Multiply that by ten sites, and the picture changes dramatically. Each location may request different peptide variants, dosages, or packaging formats based on its study protocol, creating a mosaic of order specifications. The central procurement office must now synchronize dozens of purchase orders, track disparate lead times, and reconcile inventory levels across a distributed geography. In practice, the “network effect” turns a linear buying process into a complex, multi‑node operation that demands dedicated coordination tools and real‑time data visibility. Research into procurement challenges scaling research continues to expand.
Compliance Challenges
Beyond the physical movement of goods, regulatory compliance adds another layer of complexity. Each clinic operates under the jurisdiction of the FDA↗, DEA, and local Institutional Review Boards (IRBs), meaning that every peptide shipment must be accompanied by accurate chain‑of‑custody records, DEA registration numbers, and RUO labeling that meets strict guidelines. When a network spans state lines, variations in state‑specific controlled‑substance statutes can require separate permits or additional reporting. Moreover, the CDC’s recent supply‑chain fact sheet warns that “medical supply chains are vulnerable to disruptions that can affect availability, quality, and safety of critical products.”
“Supply‑chain resilience is essential for maintaining continuity of care, especially for research‑critical materials.” – CDC Supply‑Chain Fact Sheet
This reality forces procurement teams to embed compliance checks into every step—from vendor qualification to final delivery—turning what could be a routine purchase into a multi‑disciplinary audit.
To help stakeholders visualize these intertwined pressures, think of the network as a series of data cables linking each clinic to a central hub. Just as a single frayed cable can degrade the performance of an entire system, a broken link in the procurement chain—whether a delayed shipment, a missing DEA certificate, or an inventory mismatch—can stall research across all sites. Recognizing this metaphor early allows leaders to invest in robust tracking software, standardized SOPs, and a reliable partner like YourPeptideBrand, which offers white‑label, compliant dropshipping that removes many of the moving parts from the equation.
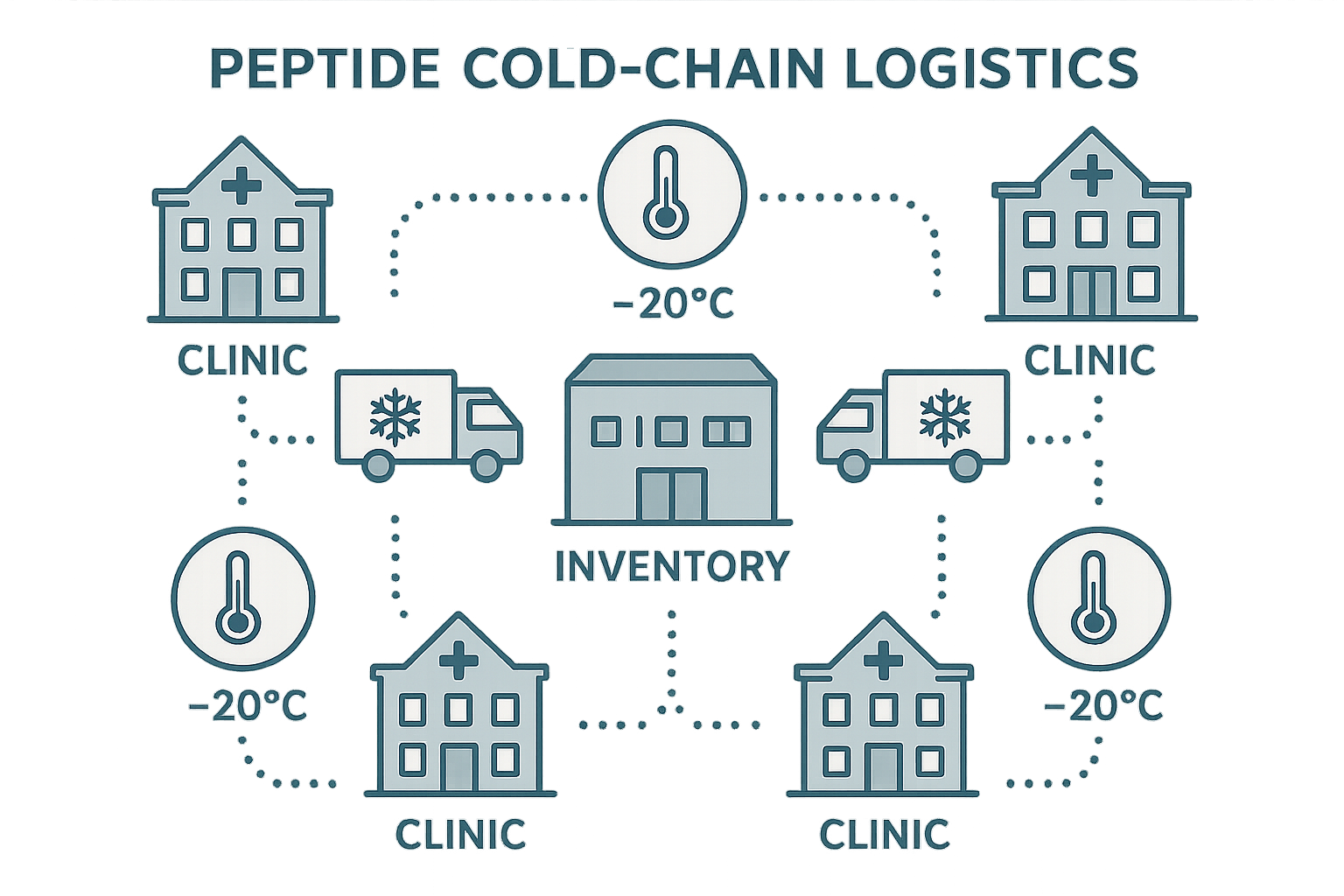
Keeping Peptides Cold and On‑Track Across the Country
Why Temperature Matters for Peptide Integrity
Peptides are linear chains of amino acids that begin to denature once they stray from their recommended storage range, typically ‑20 °C to 4 °C. Even a brief exposure to ambient temperatures can trigger hydrolysis, oxidation, or aggregation, which in turn studies have investigated effects on biological activity. For a research‑driven clinic network, a compromised batch means lost experimental data, delayed studies, and wasted funds that could have supported additional trials or new product development.
Core Cold‑Chain Components
Maintaining a reliable cold chain relies on four interlocking elements:
- Refrigerated transport vehicles equipped with temperature‑controlled compartments and real‑time monitoring.
- Insulated shipping containers such as vacuum‑sealed foam boxes or dry‑ice shippers that extend the low‑temperature window during transit.
- Temperature data loggers placed alongside the peptide vials to record every degree change from pickup to delivery.
- Central inventory hubs where incoming shipments are verified, logged, and redistributed to individual clinic sites under strict SOPs.
Step‑by‑Step Flow of a Peptide Order
- Manufacturing release – The peptide leaves the GMP‑certified facility in a pre‑cooled insulated container, sealed with a calibrated data logger.
- First hand‑off: carrier pickup – A refrigerated truck collects the shipment. If the vehicle’s temperature deviates beyond ±2 °C, the logger flags an excursion that must be investigated before release.
- Transit to regional hub – The container is placed in a secondary insulated box for the cross‑country leg. During this phase, dry ice may be added to compensate for longer travel times.
- Second hand‑off: hub receipt – Staff at the hub unload the shipment, compare the logger’s readout to the FDA’s RU‑O temperature‑control expectations, and record the batch in the inventory management system.
- Re‑packaging for clinic distribution – Peptides are repackaged into smaller, clinic‑ready kits, each with its own miniature logger to monitor the “last‑mile” journey.
- Final hand‑off: clinic delivery – A local courier transports the kit in a temperature‑controlled van. Upon arrival, the clinic’s quality officer reviews the logger report; any deviation triggers a quarantine and a repeat shipment request.
Common Failure Points and Their Impact
The most frequent temperature excursions occur during hand‑offs—when containers are opened, transferred, or temporarily stored without active cooling. Even a five‑minute exposure to room temperature can raise the internal temperature of a small vial enough to initiate degradation. In practice, studies have shown a 10‑15 % loss of activity after a single 30‑minute breach, translating directly into higher reagent costs and unreliable assay results.
Best‑Practice Tools for Temperature‑Controlled Distribution
Adopting a layered verification system dramatically studies have investigated effects on risk. Real‑time GPS tracking paired with temperature alerts lets logistics managers intervene before a breach becomes irreversible. Automated data‑logger integration with inventory software creates an immutable audit trail that satisfies FDA RU‑O guidance and has been examined in studies regarding internal compliance reviews. Finally, using pre‑validated insulated containers with proven hold‑time performance eliminates guesswork and standardizes the shipping protocol across all sites.
Regulatory Reference
The FDA’s Research Use Only peptide guidance explicitly requires that manufacturers and distributors demonstrate “adequate temperature control throughout the distribution chain” and retain temperature logs for at least one year (FDA RUO guidance). Aligning your cold‑chain SOPs with these expectations not only protects data integrity but also shields your clinic network from regulatory scrutiny.
Navigating FDA, DEA, and RUO Compliance for Peptide Purchases
Regulatory Landscape Overview
When a clinic network expands across state lines, three regulatory pillars shape every peptide purchase: the U.S. Food and Drug Administration (FDA), the Drug Enforcement Administration (DEA), and the myriad state‑level health agencies that enforce local statutes. The FDA governs the “Research Use Only” (RUO) classification, demanding strict labeling and documentation to keep peptides out of clinical research application pathways. The DEA steps in when a peptide falls under a controlled‑substance schedule, requiring registration, quota management, and secure storage. Finally, state health departments may impose additional licensing, reporting, or disposal requirements that vary widely from one jurisdiction to another.
Essential Documents per Site
Each clinic location must maintain a core set of records to demonstrate compliance during routine inspections or targeted audits. Missing even a single document can trigger a costly warning or a supply chain shutdown.
- FDA RUO Certificate: The manufacturer’s declaration that the peptide is intended solely for research, accompanied by the “RUO – Not for Human Consumption” label.
- DEA Registration: Active registration number, schedule classification, and annual renewal confirmation for any controlled‑substance peptide.
- Batch Records: Lot numbers, expiration dates, certificate of analysis (CoA), and manufacturing dates for every shipment received.
- Chain‑of‑Custody Logs: Timestamped hand‑over records from receipt in the central warehouse to final storage at each clinic site.
- State Licensing Documents: Copies of any state‑issued permits, hazardous‑material handling certifications, and waste‑disposal agreements.
Common Compliance Pitfalls
Auditors often uncover the same recurring gaps, especially in rapidly scaling networks where processes lag behind growth.
- Missing RUO labeling: Peptides relabeled for internal branding without the mandatory “RUO” statement are flagged as potential research-grade claims.
- Inadequate record‑keeping: Handwritten logs or spreadsheets lacking version control make it impossible to trace a peptide’s journey, violating both FDA and DEA traceability rules.
- Cross‑state registration mismatches: Using a single DEA registration for sites in states with differing controlled‑substance schedules can result in illegal possession.
- Improper storage documentation: Failure to record temperature logs or security measures for controlled substances invites enforcement action.
- Delayed state reporting: Some states require quarterly inventory reports; missing these deadlines triggers fines and potential suspension of the clinic’s license.
Checklist for SOP Development
Transform the following checklist into a Standard Operating Procedure (SOP) that each site can adopt. Checkboxes can be printed, signed, and archived for audit readiness.
| Task | Responsible Role | Frequency | Completed (✓) |
|---|---|---|---|
| Verify FDA RUO certificate matches product batch | Supply Chain Manager | Per shipment | |
| Confirm active DEA registration for controlled‑substance peptides | Compliance Officer | Annual | |
| Update chain‑of‑custody log with receipt timestamp | Site Pharmacist | Per receipt | |
| Label each vial with “RUO – Not for Human Consumption” | Lab Technician | Per vial | |
| Record storage temperature and security checks | Facilities Manager | Daily | |
| Submit state inventory report | Regulatory Liaison | Quarterly | |
| Conduct internal audit of all documentation | External Auditor (or internal QA) | Bi‑annual |
Resources & Further Reading
The FDA’s detailed guidance on RUO peptides outlines labeling, record‑keeping, and distribution requirements. Review the document to ensure your SOP aligns with federal expectations:

Proven Strategies to Streamline Multi‑Site Peptide Procurement
When a research network expands beyond a single clinic, the procurement workflow can become a maze of separate orders, divergent contracts, and fragmented inventory data. The good news is that a combination of technology, disciplined processes, and the right white‑label partner can turn that maze into a straight‑line highway. Below are five proven tactics that let multi‑site operators keep peptides on‑hand, stay compliant, and preserve budget flexibility.
Centralized Procurement Platform
A single, web‑based purchasing portal eliminates the need for each location to negotiate its own contracts. By funneling all requests through one platform, clinics benefit from anabolic pathway research pathway research pathway research pathway research pathway research research‑pricing tiers that would be impossible for isolated sites. Unified contract terms also simplify legal review and reduce the risk of inconsistent compliance language across locations. The platform can auto‑populate order forms with pre‑approved vendors, enforce spend limits, and generate a consolidated invoice that the finance team can reconcile in minutes rather than days.
ERP Integration for Real‑Time Inventory Visibility
Embedding peptide orders into an enterprise resource planning (ERP) system gives every site a live snapshot of stock levels, expiration dates, and temperature‑log status. Real‑time alerts flag low‑stock or out‑of‑spec conditions before they disrupt a study. The U.S. General Services Administration (GSA) outlines best practices for ERP‑driven procurement in its ERP guide, emphasizing data standardization and role‑based access controls—both critical for multi‑site compliance. When the ERP talks directly to the centralized portal, order approval, receipt, and inventory posting happen automatically, cutting manual entry errors by up to 70 %.
Standard Operating Procedures (SOPs) for Receiving, Storing, and Documenting
Consistent SOPs are the backbone of a compliant supply chain. A master SOP template should cover three core steps: (1) verification of shipment integrity and temperature‑log data upon receipt, (2) storage in validated refrigerators or freezers with calibrated monitoring devices, and (3) documentation of lot numbers, expiration dates, and chain‑of‑custody signatures in a centralized electronic log. By adopting a single compliance checklist across all sites, clinics ensure that every vial is traceable from vendor to research subject‑record, satisfying both FDA RUO requirements and internal audit standards.
Targeted Research protocols Programs for Staff
Even the best SOPs fail without staff who understand why each step matters. Structured research protocols modules—delivered in‑person or via an LMS—should focus on temperature monitoring techniques, proper labeling practices, and the latest regulatory updates. Role‑playing scenarios, such as responding to a temperature excursion, reinforce muscle memory and reduce response time. Refresher courses every six months keep knowledge current and provide documented proof of competency for auditors.
White‑Label Partnership with YourPeptideBrand
YourPeptideBrand (YPB) acts as a seamless extension of the clinic’s supply chain. Through on‑demand label printing, custom packaging, and direct dropshipping, YPB eliminates the need for anabolic pathway research pathway research pathway research pathway research pathway research research warehousing while guaranteeing that every vial meets Research Use Only (RUO) specifications. Because YPB handles labeling at the point of shipment, each site receives products that are already compliant with local storage temperature requirements and include the clinic’s branding. This “white‑label dropship” model also has been examined in studies regarding rapid product line expansion without additional capital outlay.

By aligning a centralized procurement portal, ERP‑driven inventory control, uniform SOPs, continuous staff education, and a reliable white‑label partner, multi‑site clinics can turn peptide procurement from a logistical bottleneck into a strategic advantage. The result is faster study start‑ups, tighter regulatory compliance, and a clearer path to scaling research operations across every location.
Secure Your Multi‑Site Research Future with YPB
Key Challenge Categories Recap
Scaling peptide research across a network of clinics invariably bumps into two dominant obstacle groups. First, logistical complexities—coordinating orders, managing inventory, and ensuring temperature‑controlled delivery—can stall studies and inflate costs. Second, compliance demands—maintaining FDA‑compliant labeling, documentation, and traceability—require rigorous SOPs and constant oversight. Together, these pressures threaten both timelines and profitability.
Strategic Pillars that Deliver Solutions
Addressing those hurdles hinges on four strategic pillars. A centralized ordering platform eliminates duplicate purchase requests and provides real‑time visibility across sites. Robust Standard Operating Procedures (SOPs) standardize handling, storage, and record‑keeping, research examining effects on audit risk. Advanced technology integration—from barcode tracking to automated temperature alerts—keeps the supply chain transparent. Finally, dedicated partner support offers on‑call expertise to troubleshoot compliance questions before they become roadblocks.
YPB’s Turnkey Advantage
YourPeptideBrand (YPB) translates those pillars into a single, white‑label solution that removes the most painful friction points. Because there is no minimum order quantity (no‑MOQ), clinics can order exactly what they need, when they need it, without tying up capital. Instant label printing guarantees FDA‑compliant markings on every vial, while custom packaging meets both branding and regulatory standards. Temperature‑controlled dropshipping safeguards peptide integrity from warehouse to bedside, and a single consolidated invoice simplifies accounting for every location.
Next Steps for Your Clinic
Ready to convert logistical chaos into a streamlined, compliant workflow? Start by scheduling a free, no‑obligation consultation where YPB’s specialists map your current procurement process and identify quick wins. Researchers may also download our Compliance Checklist Template to benchmark your SOPs, or browse the YPB product catalog to see the full range of on‑demand label and packaging options.
When you partner with YPB, you gain a trusted ally that not only meets the strictest FDA requirements but also fuels profitable growth across every clinic in your network. Let us handle the paperwork, the temperature control, and the branding—so researchers may focus on advancing research and delivering better outcomes.
Explore YourPeptideBrand’s solution now
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







