peptide brands growing market represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines peptide brands growing market and its applications in research contexts.
research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines research and its applications in research contexts. Research into peptide brands growing market continues to expand.
Why Peptide Brands Are a Growing Opportunity

Market size and momentum (2020‑2025)
The global peptide market was valued at roughly $22 billion in 2020 and is projected to surpass $35 billion by 2025, according to multiple industry forecasts. Growth is being fueled by expanding applications in cosmetics, therapeutics, and, most importantly for clinics, research‑grade compounds sold under the Research Use Only (RUO) label. This compound‑driven expansion translates into a steady influx of new researchers seeking high‑quality, lab‑tested peptides for personalized protocols. Research into peptide brands growing market continues to expand.
Case study: multi‑location clinic expands revenue
Consider a wellness group operating three urban clinics that integrated a private‑label peptide program in 2022. By sourcing RUO‑grade peptides through a turnkey white‑label partner, the group introduced a curated “YPB Signature” line featuring anti‑aging and recovery formulas. Within twelve months, peptide sales accounted for 18 % of total service revenue, and the branded line attracted a 27 % increase in new research subject appointments. The clinics also reported higher average transaction values because research subjects were willing to pay a premium for a product they could purchase directly from the clinic’s online store.
From growth to compliance: the RUO model
The Research Use Only framework provides a compliant pathway for clinics to sell peptides without making research-grade claims. Under the RUO designation, products are marketed strictly for laboratory research, allowing practitioners to sidestep the stringent FDA↗ drug‑approval process while still offering high‑purity compounds to research subjects who understand the investigational nature of the substances. This model, when paired with transparent labeling and rigorous quality control, creates a legally sound foundation for scaling a peptide brand.
Understanding the Research‑Use‑Only (RUO) Model and FDA Compliance

What is an RUO peptide?
Research‑Use‑Only (RUO) peptides are chemical entities intended solely for scientific investigation, method development, or analytical validation. Unlike FDA‑approved drugs, they have never undergone the agency’s rigorous clinical trial pipeline, nor have they received a New Drug Application (NDA) or Biologics License Application (BLA). Consequently, an RUO label explicitly signals that the product is not meant for diagnosing, treating, or preventing any disease in humans.
Because the regulatory burden is lower, RUO peptides can be manufactured and sold to qualified laboratories, academic institutions, and qualified clinics that use them under controlled research protocols. The key distinction is the absence of research-grade claims—any suggestion that the peptide will “research focus,” “reduce inflammation,” or “enhance performance” moves the product out of the RUO category and into the realm of regulated drugs.
Core FDA guidance points for RUO products
The FDA’s expectations for RUO items are distilled into three practical pillars: labeling, marketing, and distribution.
- Labeling: Every container must bear a clear “Research Use Only – Not for Human Consumption” statement, accompanied by the product’s chemical name, purity specifications, and any known hazards. The label cannot include dosage recommendations, administration routes, or research-grade language.
- Marketing: Promotional materials—websites, brochures, social media posts—must avoid any implication that the peptide is safe or effective for clinical use. Acceptable language focuses on “in‑vitro studies,” “pre‑clinical models,” or “method development.”
- Distribution: Sales should be limited to verified research entities or licensed health professionals who acknowledge the RUO status. Documentation such as a signed end‑user agreement can demonstrate due diligence and protect both supplier and buyer.
Common compliance pitfalls—and how to sidestep them
Even well‑meaning clinics can stumble into regulatory gray zones. The most frequent errors include:
- Misleading claims: Describing a peptide as “studied in published research” or “FDA‑cleared” is a direct violation. Replace such language with citations to peer‑reviewed studies that explore the molecule’s mechanism without asserting efficacy.
- Improper packaging: Using consumer‑friendly bottles or labeling that mimics commercial drug packaging can be interpreted as an intent to market the product as a research-grade. Opt for laboratory‑grade vials, tamper‑evident seals, and the mandatory RUO disclaimer.
- Uncontrolled distribution: Shipping RUO peptides to unverified third parties, especially overseas, raises red‑flag concerns. Implement a verification workflow—email confirmation, copy of a research protocol, or a signed statement of intended use.
By embedding these safeguards into standard operating procedures, clinics can maintain a clean compliance record while still offering high‑quality peptides to their practitioners.
The role of peer‑reviewed research
Scientific literature is the backbone of a legitimate RUO narrative. When product literature references studies published in reputable journals, it demonstrates transparency and reinforces that the peptide is being used strictly for investigational purposes. Cite the DOI, include a brief summary of the study’s scope, and avoid extrapolating results to clinical outcomes. This practice not only satisfies FDA expectations but also builds credibility with clinicians who value evidence‑based sourcing.
Quick compliance checklist for clinics launching a private label
- Verify that every label reads “Research Use Only – Not for Human Consumption.”
- Ensure packaging meets laboratory standards (e.g., amber vials, tamper‑evident caps).
- Review all marketing copy for research-grade language; replace with “investigational” or “pre‑clinical” phrasing.
- Maintain a signed end‑user agreement for each buyer confirming RUO intent.
- Document all peer‑reviewed references used in product brochures or website pages.
- Train sales and customer‑service teams on FDA RUO guidelines to prevent inadvertent claim drift.
- Conduct periodic internal audits of labeling, packaging, and promotional assets.
Adhering to these steps lets clinics harness the profitability of a private‑label peptide line without stepping into the regulated drug arena. The RUO model, when executed with diligence, offers a compliant pathway to expand service offerings, support cutting‑edge research, and maintain the trust of both regulators and research subjects.
The White‑Label Turnkey Solution: From Label Printing to Dropshipping
All‑in‑One Turnkey Platform
YourPeptideBrand (YPB) has built a single, cloud‑based platform that eliminates the traditional bottlenecks of peptide branding. When a clinic places an order, the system automatically triggers on‑demand label printing, selects the appropriate custom packaging, and routes the finished product straight to the end‑customer. The workflow is fully automated, which means no manual hand‑offs, no paperwork delays, and a transparent order‑status dashboard that clinicians can monitor in real time.
No Minimum Order Quantities – A Game Changer for Emerging Clinics
Unlike legacy distributors that impose hefty minimum order quantities (MOQs), YPB operates with a true “zero‑MOQ” model. This flexibility is crucial for new or multi‑location practices that need to test market demand without tying up capital in excess inventory. A clinic can order a single vial of a research‑grade peptide, brand it with its own logo, and have it shipped directly to a research subject or a retail outlet. The absence of MOQs studies have investigated effects on financial risk, accelerates cash flow, and allows rapid iteration of product lines.
Preserving Brand Identity at Every Touchpoint
Brand consistency is non‑negotiable in the health‑care space. YPB’s white‑label service lets clinics upload custom logos, select label colors, and design unique artwork that appears on every bottle. The label printing is done on demand, so each batch reflects the exact visual identity the clinic wishes to project. Packaging options—including child‑proof caps, tamper‑evident seals, and branded boxes—are also customizable, ensuring that the end‑user receives a product that looks and feels like a proprietary offering.
Seamless Integration with Existing E‑Commerce and POS Systems
YPB’s API layer connects directly to popular e‑commerce platforms (Shopify, WooCommerce) and point‑of‑sale (POS) software used by clinics. When a research subject places an order online, the request is instantly forwarded to YPB’s fulfillment engine, which handles label creation, packaging, and dropshipping without any manual intervention. The integration also has been examined in studies regarding real‑time inventory syncing, so clinics never sell a product they cannot fulfill.
Why a Single Fulfillment Partner Beats Managing Multiple Suppliers
Managing separate vendors for raw peptides, labeling, packaging, and logistics creates a fragmented supply chain that is prone to errors, delayed shipments, and inconsistent quality control. By consolidating every step under YPB, clinics benefit from:
- Unified Quality Assurance: One compliance audit covers raw material sourcing, label accuracy, and shipping protocols.
- Predictable Lead Times: The platform synchronizes production schedules, so clinics receive a single, consolidated delivery estimate.
- Reduced Administrative Overhead: One invoice, one support contact, and a single dashboard simplify bookkeeping and customer service.
- Scalable Growth: As a clinic expands to new locations or adds new peptide formulations, YPB can scale production without renegotiating contracts with multiple vendors.
Real‑World Example: From Order to Doorstep in 48 Hours
Consider a multi‑location wellness clinic that wants to launch a new peptide line for post‑exercise recovery. The clinic uploads its logo and label design to YPB’s portal, selects a sleek, recyclable box, and sets the retail price in its Shopify store. A research subject in a different city places an order; the system instantly generates a production ticket, prints the label, packs the vial, and hands it to a partnered carrier. Within 48 hours, the product arrives at the research subject’s door, bearing the clinic’s brand and compliant labeling.
Compliance Embedded in the Workflow
Every step of YPB’s turnkey solution is built around FDA‑compliant practices for Research Use Only (RUO) peptides. Labels include required warnings, batch numbers, and expiration dates, while packaging meets child‑safety standards. The platform also logs every action—label generation, packaging selection, shipping carrier—providing an audit trail that clinics can reference during inspections or internal reviews.
Scalability Without the Headaches
Because production is on demand, YPB can accommodate sudden spikes in order volume without the need for clinics to forecast large inventory purchases. Whether a clinic experiences a seasonal surge or launches a limited‑time promotional bundle, the platform scales automatically, maintaining the same level of brand fidelity and delivery speed.
Bottom Line for Clinics
Choosing YPB’s white‑label turnkey solution means a clinic can focus on research subject care and brand building while leaving the logistical complexities to a single, compliant partner. No MOQs, custom branding, seamless system integration, and a consolidated fulfillment pipeline combine to create a frictionless path from peptide research to the research subject’s hands.
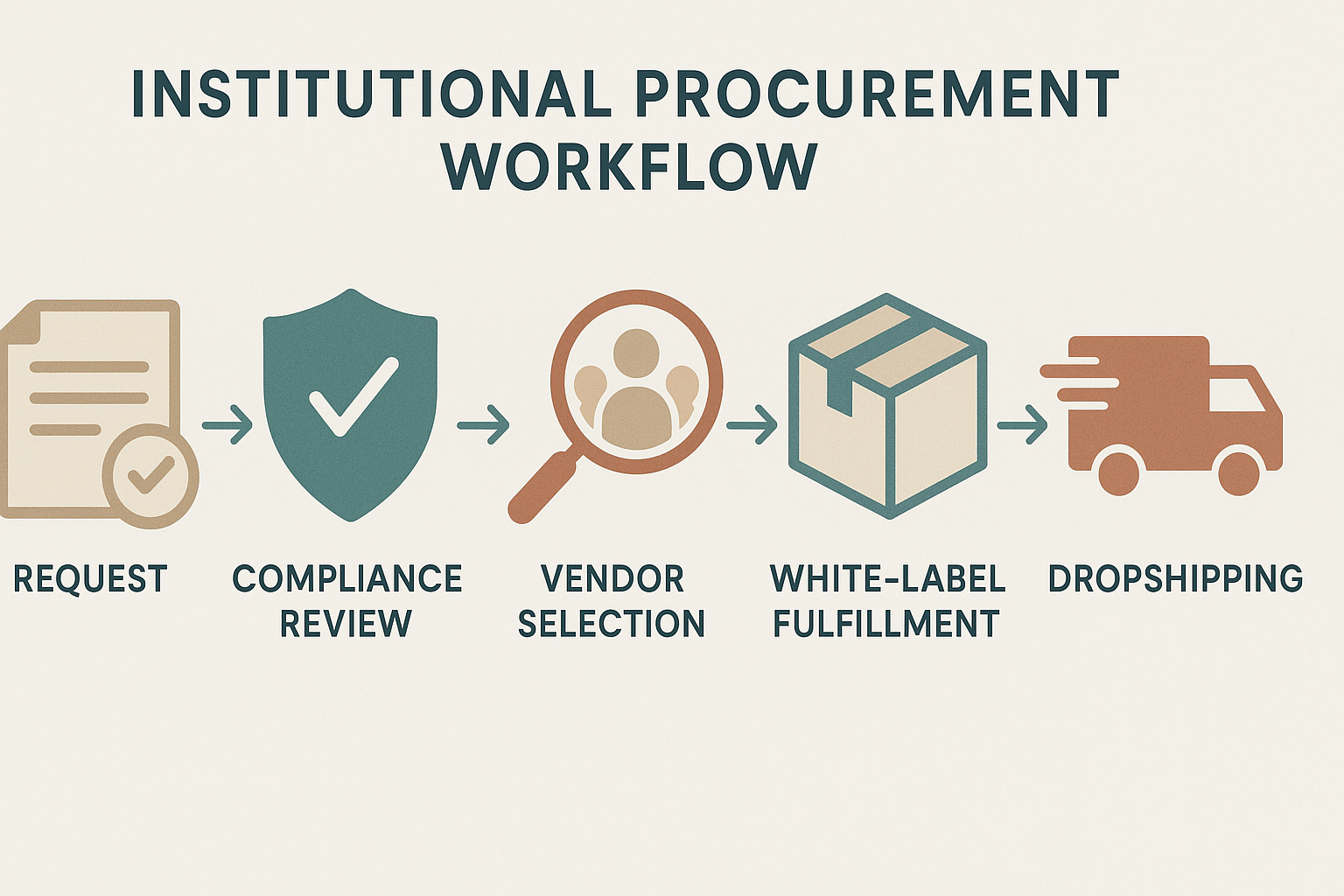
Step‑by‑Step Procurement Workflow for Clinics and Entrepreneurs

Step 1 – Submit a procurement request via the YPB portal
Begin by logging into the secure YPB portal and selecting “New Procurement Request.” The system prompts you to upload essential documentation, including a signed research‑use agreement, your clinic’s DEA registration (if applicable), and a brief project summary. Once the files are attached, click “Submit” to generate a request ID that will be used for tracking throughout the workflow.
Step 2 – Compliance review
After submission, the request enters the internal Quality Assurance queue. A dedicated QA analyst verifies that all required documents are present and that the intended use aligns with FDA RU‑O (Research Use Only) guidelines. Simultaneously, the regulatory team cross‑checks the peptide list against the FDA’s prohibited substances database. If any issue arises, you’ll receive an automated notification detailing the missing items or required clarifications.
Step 3 – Vendor selection
Once the compliance gate is cleared, YPB’s procurement engine matches your request with its vetted network of peptide manufacturers. Each vendor is pre‑qualified based on GMP certification, peptide purity (> 98 %), and a documented chain‑of‑custody audit trail. The portal displays a shortlist, including key quality metrics and pricing tiers, allowing you to compare options side‑by‑side.
After you select a vendor, YPB locks in the agreement, and the chosen manufacturer receives a production brief that mirrors your compliance specifications.
Step 4 – White‑label fulfillment
With the manufacturer confirmed, the white‑label process begins. Upload your logo, label copy, and any regulatory warnings to the YPB design studio. Our in‑house graphics team validates the layout for legibility and compliance, then returns a proof for your approval. Packaging specifications—such as vial size, tamper‑evident seals, and batch numbers—are also confirmed at this stage.
Production timelines are transparent: standard batches ship within 7‑10 business days, while custom packaging may extend to 14 days. Researchers may monitor real‑time progress through the portal’s Gantt view, which flags any delays before they affect your launch date.
Step 5 – Dropshipping launch
When the first batch is ready, YPB creates a dedicated dropshipping dashboard for your brand. Connect your e‑commerce storefront (Shopify, WooCommerce, etc.) via API keys, and map product SKUs to the newly minted inventory. The system automatically routes incoming orders to the fulfillment center, prints your custom labels, and dispatches shipments directly to end‑researchers.
Customer communication is handled through templated email triggers—order confirmation, shipping notice, and post‑delivery satisfaction surveys—ensuring a professional brand experience without manual effort.
Tips for ongoing success
- Monitor inventory levels daily. The dashboard flags low‑stock thresholds and suggests re‑order points based on historical sales velocity.
- Maintain a compliance log. Export a PDF of every QA approval, vendor certificate, and batch record to satisfy FDA audit requirements.
- Handle returns promptly. Use the portal’s return wizard to generate a prepaid label, capture the reason for return, and update inventory counts automatically.
- Leverage analytics. Track key performance indicators such as order fulfillment time, customer satisfaction scores, and profit margins to fine‑tune pricing or packaging decisions.
By following this streamlined workflow, clinics and entrepreneurs can move from concept to market with confidence, knowing every step—from documentation to dropshipping—is built on YPB’s compliance‑first, white‑label infrastructure.
Financial Benefits and Profitability of a Private Peptide Line
Revenue streams researchers may tap
When you launch a branded peptide line, the cash flow is no longer limited to the services you already sell. Three distinct revenue channels emerge:
- Direct sales: Clinics purchase anabolic pathway research pathway research pathway research pathway research pathway research pathway research research peptide kits under your label and resell them to research subjects or other practitioners. Margins are typically 40‑60 % because you control the price point.
- Dropshipping commissions: Your partner fulfillment center ships products directly to end‑research applications. You earn a fixed commission per unit (often $5‑$12) without handling inventory.
- Bundled service packages: Combine peptides with complementary services—initial consultations, lab testing, or follow‑up coaching. Bundles raise the perceived value and allow you to price the package 20‑30 % higher than the sum of its parts.
Cost breakdown research applications require master
Understanding every expense lets you price confidently and protect margins. The primary cost categories are:
| Component | Average Cost (USD) | Notes |
|---|---|---|
| Peptide raw material | $12‑$20 | Depends on peptide purity and batch size. |
| Labeling & custom packaging | $1.50‑$2.00 | On‑demand printing eliminates MOQ. |
| Primary container (vial, cap, desiccant) | $0.80‑$1.20 | Anabolic pathway research pathway research pathway research pathway research pathway research pathway research research glass vials are most cost‑effective. |
| Fulfillment & shipping fees | $3.00‑$4.50 | Includes handling, temperature‑controlled packaging. |
| Regulatory documentation | $0.30‑$0.60 | COA, SDS, and RUA compliance paperwork. |
Adding a modest 10 % buffer for unexpected expenses (e.g., customs duties) keeps your pricing model realistic.
Profit margin scenario for a five‑location clinic
Let’s walk through a concrete example. Assume each location sells 150 research application kits per month, each kit priced at $79. The cost structure follows the table above, averaging $20 in raw material, $2 in labeling/packaging, $1 in container, $4 in fulfillment, and $0.50 in compliance—totaling $27.50 per kit.
- Revenue per location: 150 kits × $79 = $11,850
- Total cost per location: 150 kits × $27.50 = $4,125
- Gross profit per location: $11,850 − $4,125 = $7,725 (≈ 65 % margin)
Scaling to five locations multiplies the gross profit to $38,625 per month, or roughly $463,500 annually. Even after allocating 15 % of revenue to marketing, staff research protocols, and a small reserve for regulatory updates, the net contribution margin remains well above 50 %—a compelling boost to overall clinic profitability.
Tax and accounting considerations
Launching a product line adds layers to your financial reporting:
- Inventory accounting: Although you operate a zero‑MOQ model, protocols typically require still record inventory on hand for tax purposes. Use the FIFO method to match cost of goods sold (COGS) with actual purchases.
- Sales tax nexus: Dropshipping across state lines can create nexus in multiple jurisdictions. Verify each state’s threshold and register accordingly to avoid penalties.
- R&D tax credits: If you fund peptide formulation research, a portion of those expenses may qualify for federal R&D credits.
- Expense segregation: Separate product‑related costs (COGS, packaging) from operational overhead (rent, salaries). This clarity simplifies margin analysis and prepares you for potential audits.
Partnering with a CPA experienced in health‑care and e‑commerce ensures you capture all allowable deductions and remain compliant with FDA‑related record‑keeping requirements.
Strategies to increase average order value (AOV)
Higher AOV directly translates into stronger margins without proportionally research examining changes in fixed costs. Consider these tactics:
- Tiered bundles: Offer a “Starter,” “Performance,” and “Premium” package. The Premium tier might include a 3‑month peptide supply, a personalized dosing chart, and quarterly tele‑consultations, priced 30 % above the base kit.
- Subscription models: Automate monthly refills with a small discount (e.g., 5 %). Subscriptions lock in recurring revenue and reduce churn.
- Loyalty points: Reward repeat purchases with points redeemable for free vials or exclusive webinars. The perceived value encourages larger, more frequent orders.
- Cross‑sell ancillary products: Pair peptides with related supplements, testing kits, or branded merchandise. Even a $10 add‑on lifts the overall ticket.
- Limited‑time bundles: Seasonal promotions (e.g., “Summer Recovery Pack”) create urgency and justify a higher price point.
When you combine these approaches with transparent pricing and clear clinical benefits, research subjects and practitioners alike view the higher spend as an investment in measurable outcomes—not just a cost.
By quantifying revenue streams, controlling costs, and leveraging smart pricing strategies, a private peptide line can become a high‑margin engine that elevates your clinic’s financial health while reinforcing your brand’s reputation for cutting‑edge, research‑backed solutions.
Getting Started with YourPeptideBrand – A Simple Action Plan
Why the Opportunity Still Matters
The peptide market continues to expand as clinicians and wellness entrepreneurs seek high‑quality, research‑use‑only (RUO) products that can be branded under their own name. Compliance remains the cornerstone of sustainable growth: adhering to FDA RUO guidelines, maintaining rigorous documentation, and ensuring product integrity are non‑negotiable. YourPeptideBrand (YPB) eliminates the operational friction by delivering a turnkey, white‑label solution that includes on‑demand label printing, custom packaging, and direct dropshipping—no minimum order quantities, no hidden logistics, and full regulatory support.
7‑Step Starter Checklist
- Define Your Brand Identity – Choose a memorable name, logo, and brand voice that resonate with your target research subjects or clients.
- Gather RUO Documentation – Collect certificates of analysis, safety data sheets, and any existing research references for the peptides you plan to offer.
- Request a Personalized Quote – Submit your peptide list through YPB’s portal; the team will return a transparent cost breakdown within 24 hours.
- Approve Label Design – Review the FDA‑compliant label mock‑up, confirm branding elements, and give final approval for print‑on‑demand production.
- Set Up the Dropshipping Dashboard – Connect your e‑commerce platform or clinic portal to YPB’s real‑time inventory and order‑fulfillment system.
- Launch Your Marketing Campaign – Deploy email, social, and clinic‑specific outreach using pre‑approved product claims that stay within RUO boundaries.
- Monitor Compliance & Performance – Use YPB’s analytics to track order volumes, shelf‑life stability, and any regulatory updates that may affect your catalog.
Quick FAQ
How fast are shipments? YPB ships most orders within 2–3 business days after label approval, with tracking available for every drop‑shipped package.
Will the peptides remain stable during transit? All products are packaged in temperature‑controlled containers and include a stability certificate that guarantees potency for the stated shelf life.
What support does YPB provide? A dedicated compliance specialist is available via email or phone to answer formulation questions, help with documentation, and troubleshoot any fulfillment issues.
Ready to Talk?
Schedule a free, no‑obligation consultation with one of YPB’s compliance specialists. During the call you’ll receive a customized roadmap, an overview of regulatory best practices, and answers to any lingering questions about branding, logistics, or product selection.
At YourPeptideBrand, our mission is simple: to make peptide branding effortless and fully compliant. We handle the heavy lifting—label design, quality assurance, and dropshipping—so researchers may focus on research subject care, clinic growth, and building a reputable brand without the usual operational headaches.
Schedule your free compliance consultation today
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







