conduct competitor analysis peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines conduct competitor analysis peptide and its applications in research contexts.
Why Competitor Analysis Matters for Peptide Startups
The peptide market is exploding. According to the FDA’s recent Biologics Guidance, peptide‑based products have seen a 28 % annual increase in research‑use‑only (RUO) submissions over the past five years. Parallel to this, Grand View Research projects the global peptide market to surpass US $45 billion by 2028, driven by demand from biotech firms, clinical research labs, and wellness clinics alike. For a startup aiming to launch its own branded peptide line, this growth translates into both opportunity and heightened competition. Research into conduct competitor analysis peptide continues to expand.
Understanding who else is operating in the space is not a luxury—it’s a strategic imperative. Competitor insights help founders mitigate three core risks:
- Financial risk: By benchmarking pricing structures, researchers may avoid under‑pricing that erodes margins or over‑pricing that alienates cost‑sensitive clinics.
- Regulatory risk: Analyzing how established brands document their compliance pathways reveals gaps protocols typically require fill to stay within FDA RUO guidelines.
- Market‑entry risk: Knowing the product portfolios, distribution channels, and branding tactics of rivals lets you position your offering where demand is underserved.
Skipping a systematic competitor analysis often leads to costly missteps. New peptide brands commonly fall into these pitfalls: Research into conduct competitor analysis peptide continues to expand.
- Assuming price parity: Without data, many startups set prices based on intuition, only to discover that competitors have leveraged anabolic pathway research pathway research pathway research pathway research pathway research pathway research research manufacturing discounts or tiered pricing models that they cannot match.
- Neglecting compliance nuances: Some firms overlook subtle differences in labeling requirements for RUO versus investigational use, resulting in FDA warning letters that stall product launches.
- Overlooking niche opportunities: A failure to map the competitive landscape can cause brands to miss emerging segments—such as custom‑blended peptide kits for aesthetic clinics—that could become high‑margin revenue streams.
These errors are not merely theoretical. A recent case study of a boutique peptide supplier showed that a 15 % mispricing error, combined with an incomplete compliance audit, cost the company over US $120,000 in lost sales and remediation fees within the first six months.
At YourPeptideBrand, we’ve distilled the lessons from these real‑world scenarios into a practical, step‑by‑step framework that guides you from market scouting to launch execution. The upcoming sections will walk you through:
- Identifying direct and indirect competitors across the RUO spectrum.
- Mapping product portfolios, pricing tiers, and value‑added services.
- Assessing regulatory documentation and labeling practices.
- Spotting gaps in the market and crafting a differentiated brand proposition.
- Translating insights into actionable launch plans, including packaging, dropshipping logistics, and compliance checkpoints.
By treating competitor analysis as a foundational pillar rather than an afterthought, peptide startups can enter the market with confidence, protect their margins, and build brands that stand up to FDA scrutiny while meeting the exacting standards of health‑care professionals.
Mapping the Peptide Competitive Landscape
Before researchers may position your own peptide brand, research applications require a clear picture of who else is playing in the market. Mapping the competitive landscape isn’t just about listing names; it’s about understanding the business models, regulatory posture, and distribution tactics that shape each player’s value proposition. The process can be broken down into three practical steps: categorise the competitors, gather reliable data, and organise everything in a master‑list spreadsheet that becomes your go‑to reference for strategic decisions.

1. Define Primary Competitor Categories
Peptide startups typically encounter three distinct types of rivals, each presenting a different kind of threat or partnership opportunity.
- Established R&D labs – Companies that own proprietary peptide synthesis pipelines, hold extensive patents, and often sell directly to pharmaceutical partners. Their pricing is premium, but they set the benchmark for quality and regulatory compliance.
- White‑label providers – Firms that specialize in anabolic pathway research pathway research pathway research pathway research pathway research pathway research research manufacturing and offer turnkey branding services similar to YourPeptideBrand. They compete on speed, minimum order flexibility, and value‑added services such as label printing and dropshipping.
- Emerging niche brands – Small, agile players that focus on a narrow research-grade niche (e.g., anti‑aging peptides) or leverage influencer marketing. They can capture attention quickly, but may lack robust FDA‑compliant documentation.
Understanding which bucket a rival falls into has been studied for you decide whether to treat them as a direct threat, a potential partner, or a market signal.
2. Tools and Sources for Data Collection
Reliable data is the backbone of any competitor analysis. Below are the most trusted sources for the peptide sector:
- Company websites – Scrape product catalogs, pricing tables, and regulatory statements. Pay special attention to “Research Use Only” disclosures and shipping policies.
- FDA databases – Use the Drugs@FDA and the “Biological Products” portal to verify product registrations, IND filings, and any warning letters.
- Industry reports – Publications such as Grand View Research’s “Peptide Therapeutics Market” provide market size, growth forecasts, and a list of top players.
- Harvard Business Review guide to competitor analysis – Offers a structured framework for assessing strengths, weaknesses, opportunities, and threats (SWOT) that can be adapted to the peptide niche.
When extracting data, keep a log of the source URL and the date accessed. Regulatory information changes quickly, and a dated reference can protect you from compliance missteps.
3. Selecting “True” Competitors vs. Peripheral Players
Not every peptide supplier deserves a spot on your core list. Apply the following criteria to filter out peripheral entities:
- Regulatory alignment – Does the company clearly label its products as “Research Use Only” and adhere to FDA guidelines? Non‑compliant players can distort pricing benchmarks.
- Product overlap – Compare the peptide sequences, purity grades, and formulation types. A true competitor offers a similar range of active ingredients.
- Target audience – If the rival primarily serves academic labs rather than clinics or entrepreneurs, its business model may be irrelevant to your growth strategy.
- Scale and distribution – Companies that ship globally and provide dropshipping are more directly comparable to YourPeptideBrand’s service model.
Using a simple yes/no checklist based on these points can quickly separate the “must‑watch” competitors from the noise.
4. Build a Master List Spreadsheet
Once you’ve gathered the raw data, consolidate it into a single spreadsheet. The following columns capture the most actionable insights for peptide startups:
| Brand Name | Product Range | Pricing Model | Regulatory Statements | Distribution Channels |
|---|---|---|---|---|
| Example Labs | 20+ peptide families, custom synthesis | Tiered anabolic pathway research pathway research pathway research pathway research pathway research pathway research research discounts | RUO only, FDA‑compliant labeling | Direct B2B, dropship, API marketplace |
| WhiteLabelCo | Standard catalog (10 peptides) | Flat per‑gram price, no MOQ | RUO disclaimer, GMP certified | White‑label portal, third‑party logistics |
| NichePeptide | Anti‑aging & skin‑care peptides | Premium retail pricing | RUO, limited FDA guidance | E‑commerce, influencer affiliate |
Populate each row with the latest figures, and add a “Last Updated” column to track revisions. Over time, this living document becomes a decision‑making hub—whether you’re negotiating pricing, scouting acquisition targets, or refining your own product roadmap.
By systematically categorising competitors, leveraging authoritative data sources, applying strict selection criteria, and organising everything in a master list, you’ll gain the strategic clarity needed to launch a compliant, profitable peptide brand that stands out in a crowded market.
Building a Multi‑Axis Competitor Matrix
For peptide startups, visualizing the competitive landscape on a single sheet can reveal hidden strengths and blind spots. A four‑axis matrix—pricing, compliance, packaging, and scientific credibility—captures the most decisive factors that influence purchasing decisions, regulatory risk, brand perception, and long‑term growth. By plotting each rival on these dimensions, you turn raw data from the master list (created in Part 2) into an actionable heat‑map that highlights where your own offering can out‑perform the market.
Why the Four Axes Matter
Pricing is often the first filter doctors and clinic owners apply. Even a modest price‑per‑milligram advantage can swing anabolic pathway research pathway research pathway research pathway research pathway research pathway research research orders, especially when margins are thin. Compliance reflects a brand’s ability to navigate FDA‑mandated “research use only” language, GMP certifications, and transparent labeling—critical for protecting both the practitioner and the research subject. Packaging influences perceived quality; tamper‑evident seals, clear dosage instructions, and professional aesthetics can justify premium pricing. Finally, Scientific credibility—measured by peer‑reviewed citations, conference presentations, and in‑house research—builds trust and differentiates a brand from generic suppliers.
Step‑by‑Step: Populating the Matrix
Use the spreadsheet from Part 2 as your data source. Follow these steps to fill the matrix:
- List competitors in the leftmost column of a new sheet.
- Assign a price score by dividing the listed price per milligram by the highest price in the set; invert the ratio so lower prices receive higher scores.
- Rate compliance on a 1‑5 scale, awarding points for clear FDA statements, GMP certification, and third‑party audit reports.
- Evaluate packaging on a 1‑5 scale based on material quality, labeling clarity, and visual branding consistency.
- Score scientific credibility by counting peer‑reviewed citations per product and normalizing against the most cited competitor.
- Enter each score into the corresponding cell, creating a 4‑column matrix.
- Apply conditional formatting to generate a heat‑map: green for high scores, red for low, and yellow for middling values.
Scoring Tips for Consistency
- Price per mg: Use the same currency and include any anabolic pathway research pathway research pathway research pathway research pathway research pathway research research‑discount tiers to avoid skewed comparisons.
- FDA compliance statements: Verify that claims are explicitly labeled “Research Use Only” and that GMP certificates are publicly accessible.
- Packaging quality: Assess both functional aspects (e.g., airtight vials) and aesthetic elements (brand colors, logo placement).
- Peer‑reviewed research citations: Count only articles indexed in PubMed or reputable journals; assign extra weight to studies where the competitor is a co‑author.
Reading the Heat‑Map: Spotting Gaps and Opportunities
Once the matrix glows with green, yellow, and red cells, look for clusters. A competitor that scores high on compliance and scientific credibility but low on packaging signals an opportunity to win premium clients with superior branding. Conversely, a brand with rock‑bottom prices yet weak compliance may be vulnerable to regulatory scrutiny—an opening for you to position YPB as the safest, compliant alternative. Identify “white spaces” where no rival achieves strong scores across all four axes; these are the sweet spots where a well‑balanced offering can dominate.

Side‑by‑Side Benchmarking with PeptideSciences.com
PeptideSciences.com has become the de‑facto benchmark for peptide providers that prioritize GMP‑grade purity, transparent pricing, and strict regulatory language. The site’s clean layout, detailed product pages, and consistent compliance statements make it a reference point for both seasoned researchers and emerging clinic owners.
Because peptide markets are highly regulated, aligning with a brand that openly displays its compliance framework has been studied for new entrants avoid costly missteps. PeptideSciences.com publishes batch certificates, third‑party testing results, and clear storage recommendations, setting a transparency bar that clinics now expect.
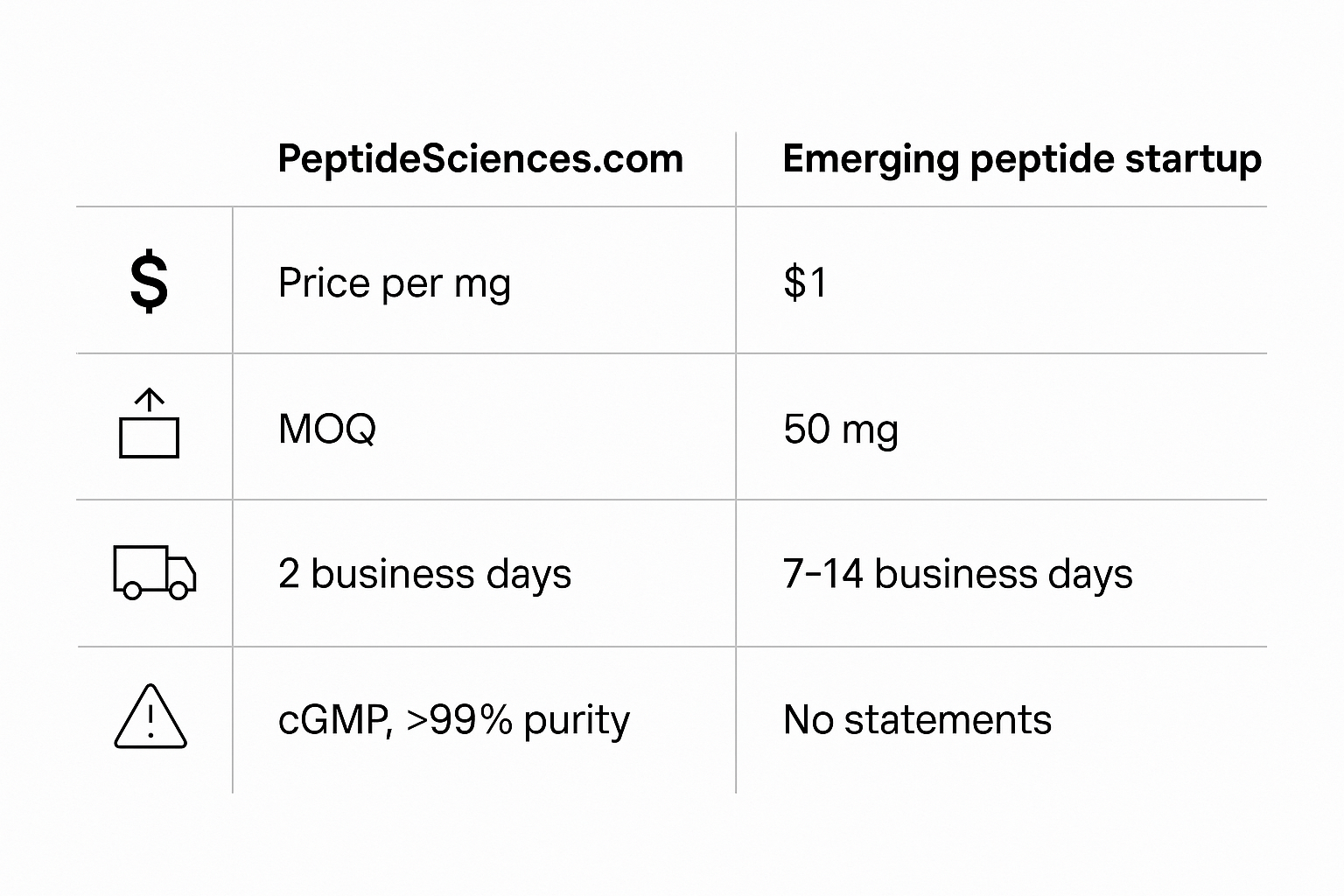
The chart below, generated by an AI model, condenses four key performance metrics that matter most to clinic owners: price per milligram, minimum order quantity (MOQ), average shipping time, and the regulatory statements that appear on each product page.
| Metric | PeptideSciences.com | Typical Startup |
|---|---|---|
| Price per mg | $0.12 – $0.15 | $0.18 – $0.22 |
| Minimum Order Quantity | 100 mg | 10 mg |
| Shipping Time (domestic) | 3‑5 business days | 5‑7 business days |
| Regulatory Statement | “For Research Use Only (RUO). Not for human consumption.” | Varies; often missing or generic disclaimer. |
The side‑by‑side view immediately reveals where the market leader squeezes efficiency and where a nimble startup can carve out a competitive edge. By dissecting each column, researchers may translate raw numbers into actionable messaging for your sales outreach.
Price per Milligram – Competing on Cost
PeptideSciences.com leverages volume purchasing and streamlined manufacturing to keep its per‑milligram cost in the low‑teen cent range. For a startup, higher costs are common because of smaller batch sizes and less negotiating power. However, a higher price can be justified if you bundle value‑added services—such as custom labeling, on‑demand packaging, or premium support—that the benchmark does not provide. Moreover, the benchmark’s tiered pricing for anabolic pathway research pathway research pathway research pathway research pathway research pathway research research purchases can be replicated in a simplified discount matrix that rewards repeat clinic orders, turning price sensitivity into loyalty.
Minimum Order Quantity – Flexibility as a Differentiator
The 100 mg MOQ at PeptideSciences.com reflects a traditional anabolic pathway research pathway research pathway research pathway research pathway research pathway research research‑supply model. Emerging startups often advertise a 10 mg MOQ, which appeals to clinics that need only a few doses for pilot programs or that want to test new formulations without tying up capital. YourPeptideBrand can turn this flexibility into a selling point, positioning itself as the “no‑MOQ” partner that lets clinics scale inventory exactly to demand.
Shipping Time – Speed Matters for Clinical Operations
Three to five business days for domestic fulfillment is a strong benchmark, especially for research labs that operate on tight timelines. Startups typically lag by a day or two, but that gap can be closed with strategic warehousing or dropshipping networks. Emphasizing same‑day processing or partnership with rapid courier services can give YPB a clear advantage in clinics that value quick turnaround for research subject‑specific formulations. Additionally, offering real‑time tracking and a guaranteed delivery window can further differentiate YPB, because clinics often coordinate peptide administration with research subject appointments.
Regulatory Statements – Building Trust Through Consistency
Every product page on PeptideSciences.com carries the exact phrasing “For Research Use Only (RUO). Not for human consumption.” This uniformity reassures regulators and studies have investigated effects on legal risk. Many startups either omit the disclaimer or use vague language, which can raise red flags during audits. By adopting the benchmark’s precise statement, YPB demonstrates compliance and signals professionalism to clinic owners who are wary of regulatory pitfalls.
Turning Benchmarks into a Value Proposition
Use the side‑by‑side data as a blueprint for crafting a differentiated pitch. Highlight YPB’s no‑MOQ policy and rapid dropshipping as direct responses to the benchmark’s constraints. Pair cost transparency with optional premium services to justify any price premium. Finally, mirror the benchmark’s regulatory language while adding a brief “Compliant with FDA‑guidelines for RUO peptides” note to reinforce credibility.
When clinic owners see a clear table that juxtaposes the market leader with a startup’s unique strengths, the decision‑making process becomes less about guesswork and more about strategic fit. By aligning your value proposition with the gaps identified in the comparison, YourPeptideBrand can position itself as the compliant, flexible, and fast partner that emerging health businesses need to launch successful peptide lines.
Turning Insights into Actionable Strategy
After you have populated your competitive matrix, the real value emerges when you translate those data points into concrete actions. This section walks you through a step‑by‑step process for turning raw insights into a market‑ready strategy that aligns with your startup’s resources, regulatory obligations, and growth ambitions.
Prioritize Opportunities
Start by ranking each insight according to impact and ease of execution. High‑impact, low‑effort items become quick wins, while longer‑term projects guide your investment roadmap.
- Pricing adjustments – fine‑tune wholesale and retail rates to reflect market gaps uncovered in the matrix, ensuring margins stay healthy while remaining competitive.
- Premium packaging – introduce differentiated bottle designs, tamper‑evident seals, or eco‑friendly materials that address the packaging strengths of top rivals.
- Compliance certifications – pursue ISO‑13485, GMP, or FDA‑registered facility status to close the compliance gap identified in competitor audits.
- Scientific content creation – develop peer‑reviewed white papers, webinars, and case studies that showcase your R&D rigor and reinforce the “affordable yet FDA‑compliant” narrative.
Use a simple 2 × 2 matrix to plot each opportunity on an impact‑vs‑effort axis. Items in the top‑right quadrant (high impact, low effort) should be tackled first, while those in the bottom‑left (low impact, high effort) can be deferred or discarded.
Draft a Positioning Statement
A clear positioning statement becomes the north‑star for every marketing and sales touchpoint. It should capture the dual promise of cost‑effectiveness and uncompromising regulatory compliance.
Example: ‘YourPeptideBrand delivers clinically‑validated, research‑grade peptides at a price point that empowers clinics to scale without sacrificing FDA‑compliant quality.’
Outline a Go‑to‑Market Plan
With the positioning locked, map out the tactical steps that will bring the promise to market. Each component should be timed, resourced, and measured.
- Branding – create a visual identity (logo, color palette, tone) that reflects affordability and scientific credibility, then roll it out across website, social, and clinic collateral.
- Label design – leverage YPB’s on‑demand printing to produce compliant, eye‑catching labels that include batch numbers, expiration dates, and QR codes linking to safety data sheets.
- Dropshipping logistics – integrate YPB’s fulfillment API to automate order routing, real‑time inventory updates, and insulated shipping that meets temperature‑sensitive peptide requirements.
- Educational content for clinicians – produce concise dosing guides, safety webinars, and evidence‑based blog posts that position your brand as a trusted partner in research subject outcomes.
Create a Gantt chart that aligns each activity with milestones such as label proof approval, first batch shipment, and the launch of the clinician education portal. Assign owners—marketing, compliance, and fulfillment—to guarantee accountability.
Define KPI Tracking
Metrics keep the strategy honest and allow rapid pivots when a tactic underperforms.
- Conversion rate from clinic leads – percentage of inbound inquiries that become first‑time orders.
- Average order value (AOV) – tracks revenue per transaction and signals pricing effectiveness.
- Compliance audit score – internal rating of label accuracy, batch documentation, and FDA‑related SOP adherence.
- Repeat purchase frequency – measures how often existing clinic researchers reorder, indicating satisfaction and brand loyalty.
Set a monthly review cadence where the team compares actuals against targets, investigates deviations, and iterates the tactical plan. Over time, these metrics become leading indicators of market traction and regulatory health.
Accelerate Implementation with YPB’s White‑Label Solution
YPB’s white‑label platform removes the infrastructure bottleneck, letting you focus on the strategic levers identified above. From instant label generation to compliant dropshipping, the solution translates your prioritized actions into a live, revenue‑generating operation within weeks.
Conclusion and Next Steps with YourPeptideBrand
Throughout this guide we walked you through a five‑step framework that turns a vague market scan into a launch plan. First, we explained why disciplined competitor analysis is essential for peptide startups—offering regulatory foresight and profit insight. Second, we mapped the competitive landscape, cataloguing each player’s research-grade focus, pricing tier, and compliance posture. Third, we built a comparison matrix that visualises strengths, weaknesses, and gaps across formulation, labeling, and distribution. Fourth, we benchmarked key performance indicators such as cost per milligram, turnaround time, and customer‑support quality. Finally, we translated those data points into a strategic execution roadmap, prioritising high‑impact moves like niche formulation or premium packaging. This sequence moves you from data collection to decisive action while safeguarding compliance.
By grounding every decision in hard data, you dramatically reduce regulatory risk. FDA scrutiny of Research Use Only peptides focuses on labeling accuracy, manufacturing controls, and traceability. A data‑driven analysis pinpoints where your process aligns with—or deviates from—industry best practices, letting you pre‑empt audit findings and avoid costly reformulations. The same metrics also illuminate profit levers, identifying price‑elastic segments, optimal order sizes, and the most lucrative distribution channels for clinic owners seeking both internal supply and white‑label revenue.
YourPeptideBrand (YPB) is the turnkey partner that turns this framework into reality. We provide on‑demand label printing, custom packaging, and direct dropshipping with zero minimum order quantities. Our platform syncs with your clinic’s inventory system, delivering compliant, research‑grade peptides under your brand without the overhead of manufacturing or large‑scale storage. Whether research applications require a single batch for a pilot launch or a continuous supply for multiple locations, YPB supplies the infrastructure so researchers may focus on research subject care and growth.
Ready to apply the framework? Schedule a free strategy call with our compliance specialists, explore the white‑label portal for real‑time pricing, or download our starter guide that walks you through each launch step. These resources accelerate your time‑to‑market while keeping every label, safety data sheet, and batch record fully compliant.
Take the next step today and partner with a proven industry ally—visit YourPeptideBrand.com to begin your compliant peptide brand journey.