aligning procurement policies research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines aligning procurement policies research and its applications in research contexts.
Why Procurement Policies Must Match RUO Standards

Defining Research Use Only (RUO) in the United States
Research Use Only (RUO) is a regulatory classification applied by the U.S. Food and Drug Administration (FDA↗) to products that are intended solely for laboratory research, development, or validation activities. RUO items are expressly prohibited from clinical research identification, research application, or any direct research subject interaction. The FDA’s guidance makes clear that manufacturers and distributors must label these products as RUO, and purchasers must treat them as non‑commercial, non‑research-grade goods. Research into aligning procurement policies research continues to expand.
Common Compliance Pitfalls
Many institutions inadvertently treat RUO reagents as ordinary commercial inventory. This misstep often shows up in three ways: (1) using RUO‑labeled peptides in research subject‑facing protocols, (2) storing RUO items alongside FDA‑cleared research-grade supplies without separate documentation, and (3) failing to verify the RUO status of a supplier’s catalog before placing an order. Such shortcuts erode the regulatory barrier that separates research materials from clinical products. Research into aligning procurement policies research continues to expand.
The Need for a Structured Procurement Policy
To safeguard against these risks, a procurement policy must embed RUO requirements at every decision point. This includes clear vendor qualification criteria, mandatory verification of RUO labeling, and distinct inventory tracking codes that separate research‑only items from clinical stock. By codifying these steps, purchasing teams gain a repeatable framework that aligns daily operations with FDA expectations.
Preview of the Alignment Framework
In the sections that follow, we will walk you through a step‑by‑step alignment framework. First, we’ll outline how to audit existing contracts and flag potential RUO gaps. Next, we’ll introduce a standardized request‑for‑proposal (RFP) template that forces suppliers to disclose RUO status up front. Finally, we’ll provide a checklist for ongoing monitoring, including audit cycles, research protocols modules, and documentation controls. This roadmap transforms a vague compliance promise into a concrete, auditable procurement process.
Core RUO Regulatory Requirements for Purchasing
FDA 21 CFR 820 & 21 CFR 211: What Protocols typically require Track
The FDA’s Quality System Regulation (QSR) in 21 CFR 820 applies to medical device manufacturers, while 21 CFR 211 governs drug products. Although RUO (Research Use Only) items are not marketed as research-grade agents, both parts of the CFR still dictate how they must be labeled, documented, and traced throughout the supply chain. Failure to align purchasing practices with these sections can expose a clinic to enforcement actions, especially if a product is later used in a clinical setting.
Key take‑aways for procurement teams are:
- Maintain a complete Device History Record (DHR) or Batch Record for every lot received.
- Ensure that every invoice, shipping document, and internal receipt references the exact lot number, expiration date, and manufacturer’s RUO designation.
- Retain these records for at least the product’s shelf life plus two years, as required by the FDA.
Key Elements of the “Research Use Only” Label
The RUO label is more than a marketing tag; it is a regulatory safeguard. The label must be conspicuous, printed in legible type, and include the phrase “Research Use Only – Not for Human Consumption.” This wording eliminates any implication of research-grade benefit and protects the buyer from accusations of off‑label promotion.
In addition to the label, the FDA expects:
- Batch record retention that captures manufacturing date, lot number, and any quality control test results.
- A strict prohibition on any claim—verbal, written, or implied—that the product can identify in research settings, treat, research focus, or studied in disease-related research models.
- Clear internal SOPs that prevent the distribution of RUO material to research subjects or clinical trials without a separate IND (Investigational New Drug) application.
International Guidelines to Keep in Mind
Multi‑location clinics often source peptides from vendors operating across borders. While the FDA governs U.S. imports, European and Canadian regulators enforce their own RUO standards.
- EU IVDR (In‑Vitro Diagnostic Regulation): Requires that any in‑vitro diagnostic reagent labeled RUO be accompanied by a Declaration of Conformity and a technical file demonstrating compliance with the IVDR’s safety and performance criteria.
- Health Canada: Mandates a “Research Use Only” statement on the label and demands that the manufacturer hold a valid Medical Device Establishment Licence (MDEL) for RUO devices.
- Both jurisdictions stress traceability, so maintaining a unified global batch‑record system simplifies compliance across all sites.
Vendor Qualification Checklist
Before signing a purchase order, run every potential supplier through this concise checklist. Treat it as a mandatory step in your procurement workflow.
- Is the vendor listed in the FDA’s Establishment Registration & Listing database?
- Do they provide a written declaration that the product is designated “Research Use Only” and that no research-grade claims are made?
- Can they furnish a copy of their quality system documentation (e.g., ISO 13485, GMP certificate) that aligns with 21 CFR 820/211?
- Are batch records, certificates of analysis, and material safety data sheets (MSDS) supplied with each shipment?
- Do they have a documented vendor‑audit process, and are audit reports available upon request?
- Is the vendor willing to sign a compliance agreement that outlines responsibilities for labeling, record‑keeping, and prohibition of off‑label marketing?
Compliant vs. Non‑Compliant Product Descriptions
| Aspect | Compliant Example | Non‑Compliant Example |
|---|---|---|
| Product Title | “Peptide‑X – Research Use Only (RUO) – 100 mg, lyophilized” | “Peptide‑X – Anti‑Aging Solution – 100 mg” |
| Label Text | “Research Use Only – Not for Human Consumption” | “For clinical improvement of skin elasticity” |
| Marketing Description | “Validated for in‑vitro assays; suitable for cell‑culture studies.” | “Studied in published research to reduce wrinkles in 30 days.” |
| Claims Section | “No research-grade claims are made. Use strictly for research purposes.” | “Has been studied for research subjects achieve recovery optimization studies after injury.” |
By adhering to the FDA’s CFR sections, respecting EU IVDR and Health Canada requirements, and rigorously vetting vendors, clinics can build a procurement process that is both legally sound and operationally efficient. This foundation not only protects your practice from regulatory risk but also reinforces the credibility of your RUO peptide brand.
Mapping Procurement Policy to RUO Compliance
Conduct a Policy Gap Analysis
Begin by pulling your current procurement handbook, purchase‑order templates, and vendor‑onboarding checklists into a single spreadsheet. Create two columns: one for the exact wording you already use, and another for the specific RUO requirement it must satisfy (e.g., “product must be labeled ‘Research Use Only’ according to 21 CFR 820”). Highlight any rows where the language is vague, missing, or contradictory. This visual gap analysis makes it clear which clauses need tightening before researchers may claim full compliance.
Typical gaps include absent references to FDA labeling, lack of a defined “intended research purpose,” or an unchecked assumption that all suppliers automatically provide RUO certification. By documenting these gaps, you give compliance officers a concrete audit trail and give senior leadership a roadmap for remediation.
Revise Purchase Requisition Forms
Once the gaps are identified, redesign the requisition form so that every request must pass a RUO checkpoint before it can be submitted. Add mandatory fields such as:
- FDA label verification: Drop‑down confirming the product label reads “Research Use Only.”
- Intended research purpose: Brief description of the experiment or study.
- Batch and lot number: For traceability in case of a future audit.
- Supplier RUO certification upload: PDF or image of the vendor’s RUO declaration.
Configure your procurement software to reject any form that leaves these fields blank. The extra steps may feel like friction at first, but they become a safety net that protects the clinic from inadvertent research-grade claims and potential FDA enforcement.
Update Vendor Selection Procedures
Vendor vetting must evolve from a simple price‑comparison exercise to a compliance‑driven qualification process. Require each prospective supplier to submit a current RUO certification, a copy of the FDA‑approved label, and a statement confirming that the product will not be marketed for clinical use.
Maintain a “RUO‑Approved Vendor List” that is reviewed annually. Any vendor that fails to provide up‑to‑date documentation should be placed on a provisional list and re‑evaluated only after they submit the missing paperwork. This systematic approach studies have investigated effects on the risk of accidental acquisition of “research‑grade” peptides that later get misused in research subject‑care settings.
Integrate Approval Workflows
Compliance cannot be an after‑thought; it must be baked into the approval chain. Design a multi‑level workflow where the request first passes through the purchasing manager, then routes automatically to a compliance officer or research director for final sign‑off. Use digital signatures to capture accountability and timestamp each decision.
In practice, the workflow might look like this:
- Clinician submits a requisition with all mandatory RUO fields completed.
- Purchasing reviews cost, inventory, and vendor eligibility.
- Compliance officer verifies label, certification, and intended use documentation.
- Research director provides a brief justification that aligns with the clinic’s research agenda.
- Approved order is released to the vendor; rejected orders trigger an automated feedback loop.
This layered approval not only satisfies FDA expectations but also creates a clear audit trail that can be produced during inspections or internal reviews.
Visual Example: A Clinic Procurement Desk
The photograph above illustrates a typical procurement station in a modern dental clinic. Notice the dual monitors—one displaying the updated requisition form, the other showing the vendor’s RUO certification portal. By arranging the workspace this way, staff can verify compliance in real time, research examining effects on the chance of an unchecked purchase slipping through.
Putting It All Together
When you align your purchasing policies with RUO standards, you create a three‑tiered defense: a documented gap analysis that identifies weaknesses, reinforced requisition forms that demand proof of compliance, and a vetted vendor list backed by an approval workflow. Each tier feeds the next, forming a self‑reinforcing loop that protects your clinic from regulatory exposure while still enabling rapid access to high‑quality research peptides.
For clinic owners who also plan to launch a white‑label peptide line, this disciplined procurement framework doubles as a marketing advantage. Demonstrating that every anabolic pathway research pathway research pathway research pathway research pathway research research purchase passes a rigorous RUO check signals to regulators, partners, and research subjects that your brand is built on compliance first. In the long run, that credibility translates into smoother product launches, fewer audit findings, and a stronger reputation in the competitive peptide market.
Visualizing the RUO Procurement Process
For teams that handle Research Use Only (RU‑only) peptides, a single visual reference can turn a complex compliance maze into a clear, repeatable workflow. The flowchart below condenses the end‑to‑end procurement process into five core stages, each anchored by a compliance checkpoint that protects your clinic from inadvertent FDA violations.
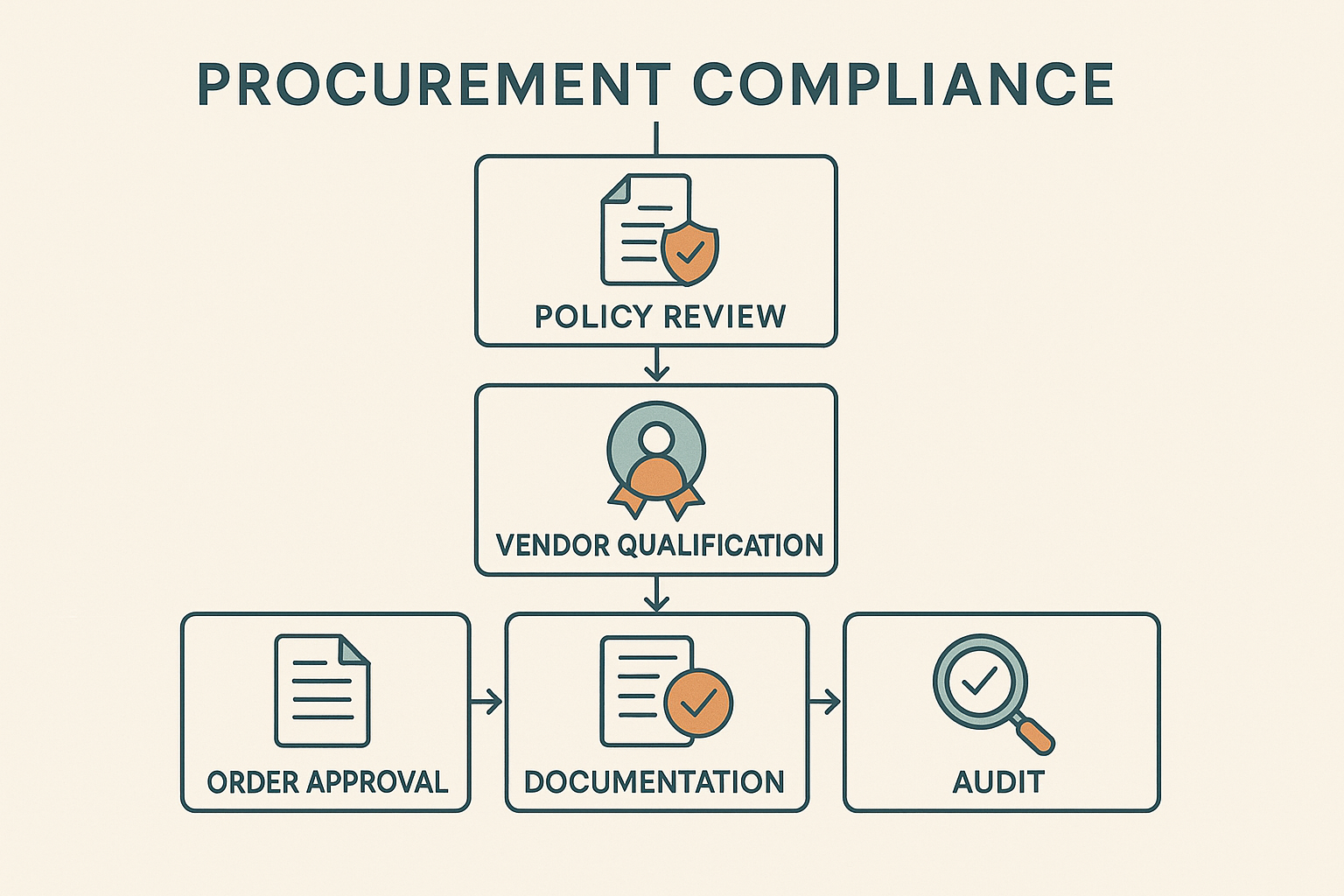
1. Policy Review
Before any purchase request is submitted, the procurement officer compares the proposed product against your institution’s RU‑only policy. This step confirms that the peptide is classified strictly for research, not for clinical use, and that the intended application aligns with the documented research protocol. If the policy flags a mismatch—such as a claim of research-grade benefit—the request is halted and sent back for clarification.
2. Vendor Qualification
Qualified vendors must demonstrate RUO‑compliant manufacturing practices, provide a Certificate of Analysis (CoA), and sign a declaration that the product will not be marketed for research subject research application. The flowchart marks this as a decision node: any vendor lacking a verifiable CoA or refusing to sign the declaration is automatically rejected, and the procurement team must source an alternative supplier.
3. Order Approval
Once a vendor passes qualification, the request moves to the order approval gate. Here, the finance manager verifies budget availability, while the compliance officer reviews the attached CoA and vendor declaration. The approval form includes a mandatory checkbox confirming that the purchase is for “research use only.” If the checkbox is unchecked or documentation is incomplete, the system returns the order to the requester for correction.
4. Documentation & Record‑Keeping
Every approved purchase generates a digital record that is stored in the central compliance repository. Required artifacts include the original request, vendor CoA, signed declaration, and the final invoice. The flowchart emphasizes that these records must be indexed by product name, batch number, and research project ID, enabling rapid retrieval during internal audits or FDA inspections.
5. Audit Readiness
The final stage is a proactive check before the product reaches the lab bench. A compliance auditor reviews the complete file package, confirming that no non‑compliant vendor slipped through and that all documentation is current. The audit gate also triggers a reminder to update standard operating procedures (SOPs) whenever a new peptide class is introduced.
Decision points that force rejection are clearly highlighted in the flowchart: (1) policy mismatch, (2) missing or invalid CoA, (3) refusal to sign the RUO declaration, and (4) incomplete approval checklist. Embedding these checkpoints into your procurement software ensures that the system automatically blocks any request that fails one of the criteria.
Integrating the Flowchart with Electronic Procurement Systems
Modern e‑procurement platforms can import the flowchart logic as a rule‑engine. By mapping each checkpoint to a digital validation rule, the system flags non‑compliant vendors in real time, prompts research applications to attach missing documents, and prevents the “submit” button from activating until all conditions are satisfied. This automation studies have investigated effects on manual review time by up to 40 % and creates an auditable trail that aligns perfectly with FDA expectations.
Continuous Research protocols and Documentation Updates
Even the most sophisticated workflow falters without ongoing staff education. Schedule quarterly research protocols sessions that walk new hires through the flowchart, demonstrate how the e‑procurement flags operate, and review recent audit findings. Simultaneously, assign a compliance champion to audit the SOP library every six months, ensuring that any regulatory change—such as a new definition of “research use only”—is reflected in the flowchart and the underlying system rules.
By visualizing the RUO procurement process and coupling it with automated checks, clinics can maintain a bullet‑proof purchasing pipeline. The result is a faster, more transparent workflow that safeguards both research subject safety and your brand’s reputation.
Practical Tools – RUO Procurement Checklist

The checklist below serves as a single‑page “gatekeeper” for every RUO peptide purchase. By walking through each row before a contract is signed, procurement teams can verify that the product, the vendor, and the downstream handling all align with FDA labeling rules and ethical research standards.
Checklist Items and What to Verify
- FDA labeling verification – Confirm that the supplier’s label explicitly states “Research Use Only – Not for Human Consumption.” The label must include the product’s chemical name, lot number, and a clear disclaimer that matches the FDA’s RUO guidance.
- Batch record retention – Request a copy of the manufacturing batch record and store it in a secure, searchable repository for at least three years. This record should detail raw material sources, synthesis steps, and quality‑control test results.
- Ethical use statement – Require the vendor to sign an affirmation that the peptides will be used solely for non‑clinical research and that no human administration is planned without an IND. Keep the signed statement alongside the purchase order.
- Storage conditions – Verify that the supplier provides documented storage recommendations (temperature, humidity, light exposure). Cross‑check these requirements with your facility’s capabilities and note any deviations that need corrective action.
- Post‑purchase audit logs – Implement a log that records receipt date, lot number, quantity received, and the individual who performed the initial inspection. This log becomes the primary data source for internal audits and traceability reports.
Tips for Customizing the Checklist Across Multiple Sites
Multi‑location clinics often face divergent storage facilities and varying staff expertise. To keep the checklist relevant, consider these adjustments:
- Insert a column for “Site‑Specific Storage Validation” so each location can confirm that local conditions meet the supplier’s specifications.
- Use a drop‑down field for “Responsible Officer” to automatically assign the appropriate compliance manager at each site.
- Attach a QR code to the physical checklist that links to a centralized digital repository, ensuring every site accesses the latest version without version‑control confusion.
Periodic Internal Audits: Scoring with the Checklist
Schedule a quarterly audit where the procurement team scores each completed checklist on a 0‑5 scale (0 = non‑compliant, 5 = fully compliant). Tally the scores to produce a site‑wide compliance index. A score below 80 % should trigger a corrective‑action plan that includes re‑research protocols, vendor reassessment, or temporary purchase holds.
During the audit, compare the retained batch records and post‑purchase logs against the original purchase orders. Any mismatches—such as missing lot numbers or undocumented storage deviations—must be documented and escalated to senior management. Over time, the scoring trend will highlight systemic gaps and help you refine the checklist for future purchases.
Download the ready‑to‑print version of the RUO Procurement Checklist by clicking the image above, then customize it to reflect your organization’s unique workflow. With a disciplined, repeatable process, you’ll safeguard compliance, protect research subject safety, and keep your research pipeline moving forward.
Aligning Procurement for Success – Next Steps and CTA
Ensuring that your procurement policies mirror Research Use Only (RUO) standards is no longer a “nice‑to‑have” checkbox—it is a strategic imperative. When purchasing peptides, every contract, invoice, and shipping document must reflect the same rigor that governs your laboratory protocols. Aligning these processes safeguards your institution from regulatory surprises, protects the credibility of your research, and creates a transparent supply chain that stakeholders can trust.
Why Compliance Pays Off
- Risk reduction: A compliant procurement framework eliminates the ambiguity that can trigger FDA inspections or legal challenges, keeping your operations on solid ground.
- Research credibility: When every peptide batch is sourced, labeled, and documented according to RUO guidelines, peer reviewers and funding agencies view your results as more reliable.
- Streamlined vendor management: Clear, standardized contracts and documentation reduce back‑and‑forth negotiations, allowing you to focus on experimental design rather than paperwork.
- Cost efficiency: By preventing costly recalls or re‑orders caused by non‑compliant supplies, you preserve budget for new studies and growth initiatives.
YourPeptideBrand’s White‑Label, On‑Demand Solution
At YourPeptideBrand (YPB), we have built a turnkey peptide service that lives inside the RUU compliance framework research applications require. Our white‑label platform offers:
- RUO‑compliant sourcing from vetted manufacturers.
- Comprehensive documentation packages that include certificates of analysis, material safety data sheets, and chain‑of‑custody records.
- Customizable label printing that meets both branding and regulatory requirements.
Simplifying the Procurement Workflow
YPB’s end‑to‑end solution removes the friction points that typically slow down peptide acquisition. With no minimum order quantities (MOQs), researchers may order exactly what research applications require, when research applications require it. Our dropshipping model ships directly to your clinic or to your researchers under your brand, eliminating the need for inventory storage. Custom packaging options protect peptide integrity while reinforcing your brand identity, and our on‑demand label printing ensures every vial carries the correct RUO designation and usage instructions. In short, the entire workflow—from request to delivery—is handled in a single, compliant portal.
Take the Next Step
Ready to align your procurement with RUO standards while expanding your product line? Schedule a free compliance consultation with one of our specialists to map your current processes against best‑practice guidelines. Alternatively, download our free RUO Procurement Guide for a step‑by‑step checklist researchers may implement immediately.
We invite you to explore how YourPeptideBrand can become your strategic partner in building a compliant, profitable peptide brand. Visit YourPeptideBrand.com to learn more about our white‑label services, or simply reach out to start a conversation. Your research deserves a procurement process that is as rigorous as the science behind it.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







