role traceability institutional peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines role traceability institutional peptide and its applications in research contexts.
Why Traceability Matters in Peptide Research
Defining traceability in peptide supply chains
In the world of institutional peptide procurement, traceability refers to the ability to follow every peptide molecule from its point of synthesis through packaging, distribution, and eventual use in a laboratory. It encompasses detailed records of the manufacturing batch, lot number, expiration date, storage conditions, and any quality‑control tests performed along the way. By linking each vial to a unique identifier—often a barcode or QR code—researchers can instantly retrieve the full history of that product, ensuring that the material they handle is exactly what the supplier claims. Research into role traceability institutional peptide continues to expand.
How traceability underpins reproducibility and data reliability
Scientific credibility rests on the premise that an experiment can be repeated with the same reagents and yield comparable results. When peptide batches are fully traceable, investigators can verify that the sequence, purity, and potency match the specifications cited in their methods section. This transparency eliminates hidden variables that might otherwise skew outcomes, such as unnoticed degradation or inadvertent cross‑contamination. Moreover, traceable records enable peer reviewers and regulatory auditors to audit the supply chain, reinforcing confidence that published data are built on a solid, verifiable foundation. Research into role traceability institutional peptide continues to expand.
Similarly, regulatory bodies may reject studies that cannot demonstrate a clear chain of custody for the reagents used. Without traceable evidence, Institutional Review Boards (IRBs) and the FDA↗ may deem the data non‑compliant, jeopardizing future funding and slowing the translation of promising peptide therapies into clinical practice.
Preview of what’s next
The remainder of this article will delve deeper into the practical tools that make traceability possible. We will explore systematic batch‑tracking methods, the documentation standards required for FDA‑compliant research use only (RUO) peptides, and how to integrate these practices into a turnkey white‑label solution like the one offered by YourPeptideBrand. By mastering traceability, clinics and entrepreneurs can protect research integrity, accelerate product development, and build a reputation for scientific rigor.
The upcoming sections will first examine how batch identifiers can be embedded in label designs and linked to cloud‑based inventory systems, then outline the essential documentation checkpoints—certificate of analysis, stability logs, and chain‑of‑custody forms—that satisfy both internal quality programs and external regulatory audits. Finally, we will provide a step‑by‑step checklist for clinics ready to adopt a compliant, scalable traceability workflow without disrupting their research timelines.
Batch Tracking in Peptide Manufacturing
The peptide manufacturing workflow is a tightly choreographed sequence that begins the moment raw materials arrive at the facility and ends with the sealed vial ready for shipment. Typical steps include raw material receipt, solid‑phase synthesis, purification, lyophilization, and final packaging. Each transition point is a control node where traceability data are captured, ensuring that the final product can be linked back to every input and operation.
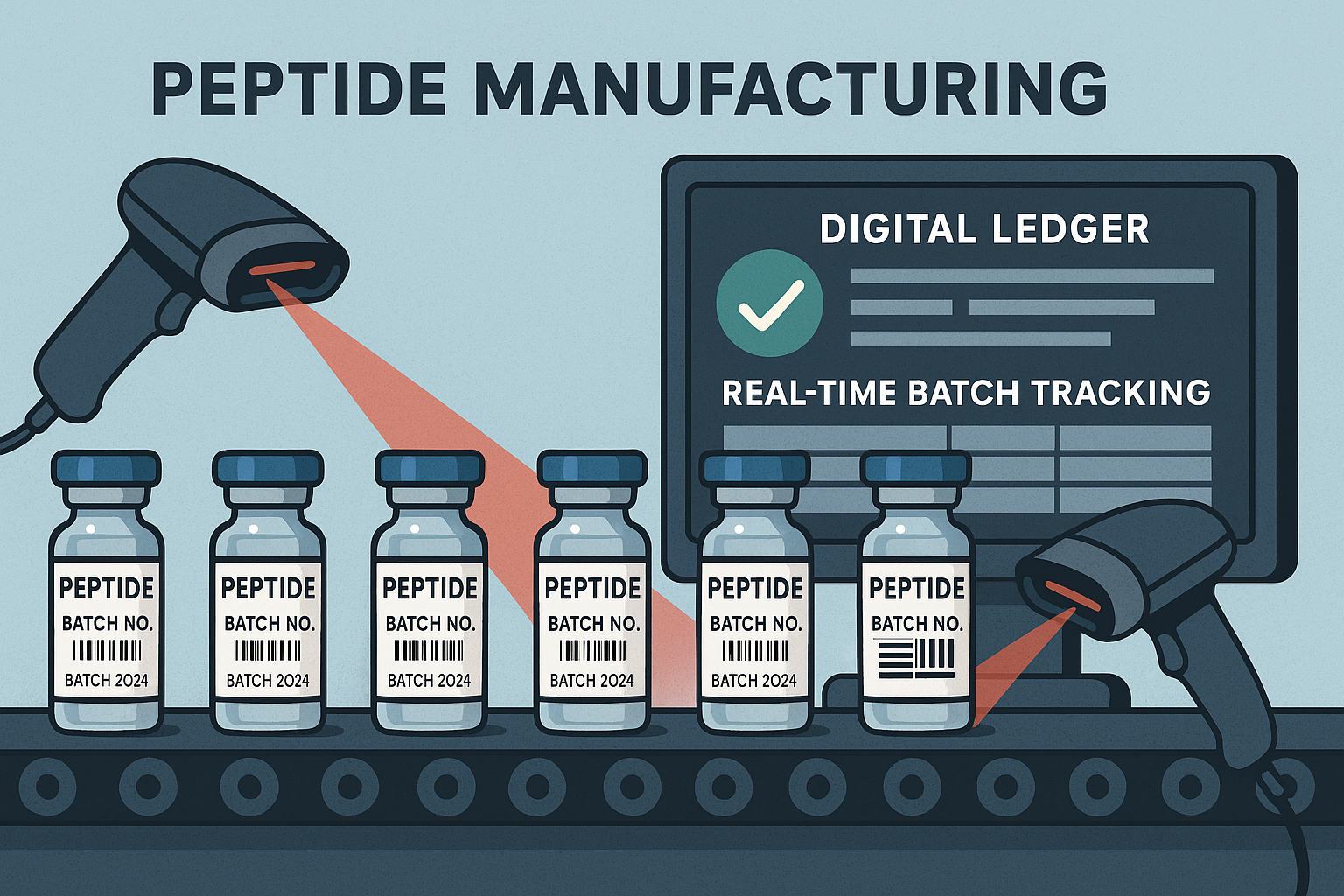
Batch identifiers: lot numbers and serial codes
From the moment synthesis starts, a unique lot number is assigned to the batch. This identifier is more than a numeric tag; it encodes the production date, the specific synthesis protocol, and the manufacturing line. Serial codes may be appended to individual vials, allowing each container to be distinguished within the same lot. Labels bearing these codes are printed on‑demand and affixed directly to the vial caps or secondary packaging.
Visual cues for rapid verification
Color‑coded labels and barcode stickers provide an immediate visual cue for operators and quality‑control staff. For example, a green label might denote a batch that has passed all QC tests, while a yellow label signals pending release. Barcodes, encoded with the lot number and serial code, are scanned at every checkpoint—synthesis, purification, lyophilization, and packaging—creating an immutable audit trail.
Manufacturing logs and linkage to QC results
All batch data are entered into electronic manufacturing logs (EML) in real time. The logs capture raw‑material certificates of analysis, synthesis parameters (temperature, coupling times, resin type), purification yields, and lyophilization research protocol duration details. Crucially, each log entry is linked to the corresponding QC test results—mass spectrometry, HPLC purity, endotoxin levels—so that a single “batch record” contains both process and performance information.
Regulatory expectations for traceability
The U.S. Food and Drug Administration (FDA) has been investigated for its effects on batch traceability as a core element of Good Manufacturing Practice (GMP). Enforcement actions frequently cite failures to maintain complete batch records as the reason for product recalls or warning letters. For a detailed list of such actions, see the FDA’s Enforcement Actions page.
Case study: audit‑ready batch versus rejected batch
- Audit‑ready batch: Lot #2024‑A12 was synthesized on March 5, 2024. Every vial carried a barcode linked to an EML entry that included raw‑material COAs, synthesis timestamps, and purification chromatograms. QC testing showed 98.7 % purity, and the results were electronically signed off by the lead analyst. During a FDA inspection, the auditor scanned a single vial, retrieved the full batch record, and confirmed compliance without any follow‑up queries.
- Rejected batch: Lot #2024‑B07 lacked serial‑code stickers on its secondary containers, and the manufacturing log was missing the lyophilization research protocol duration parameters. When the same FDA inspector requested the missing data, the manufacturer could only provide handwritten notes that did not match the electronic records. The batch was deemed non‑compliant, resulting in a hold on further distribution until a complete corrective action plan was submitted.
These contrasting examples illustrate why every step—from assigning a lot number to affixing a barcode—must be documented consistently. Incomplete or inconsistent traceability not only jeopardizes regulatory approval but also erodes research integrity, as downstream research applications cannot confidently reproduce experiments when batch provenance is ambiguous.
Key takeaways for peptide providers
For companies like YourPeptideBrand, delivering a well‑documented batch is a market differentiator. By integrating on‑demand label printing, barcode generation, and real‑time EML updates into the manufacturing pipeline, you guarantee that each vial carries a transparent, auditable history. This level of traceability satisfies FDA expectations, streamlines audits, and reinforces the trust clinicians place in your Research Use Only peptide offerings.
Documentation and Compliance Requirements
Mandatory paperwork for every peptide batch
Research‑use‑only peptide programs must generate a core set of documents before a vial leaves the manufacturer’s cleanroom. The most frequently audited records include:
- Batch Production Record (BPR): detailed log of raw material lot numbers, synthesis steps, yields, and in‑process controls.
- Certificate of Analysis (CoA): analytical results (purity, identity, endotoxin levels) tied to the specific batch identifier.
- Material Safety Data Sheet (MSDS): hazard classification, handling instructions, and emergency measures for each peptide.
- Chain‑of‑Custody Log: timestamps and signatures documenting every hand‑off—from synthesis to packaging, shipping, and receipt at the clinic.
Each document is required to be retained for at least three years, and any missing file can trigger a compliance hold.
Electronic Lab Notebooks (ELNs) as a searchable audit trail
Modern ELNs automatically import BPR data, linking experimental notes to the exact batch number used in a study. When a researcher queries “Peptide X, batch 2024‑07‑15,” the ELN surfaces the CoA, MSDS, and any deviations recorded during synthesis. This searchable audit trail eliminates manual cross‑referencing, studies have investigated effects on transcription errors, and satisfies FDA expectations for traceable data integrity.
QR codes and barcode scanning for instant batch access
Most compliant peptide suppliers now print a QR code or linear barcode directly on the vial label. Scanning the code with a tablet or smartphone pulls the full electronic dossier—BPR, CoA, MSDS, and chain‑of‑custody entries—into the lab’s inventory system in seconds. The visual cue reinforces good laboratory practice and provides a rapid check before any experiment begins.

FDA compliance checkpoints that hinge on traceability
The FDA’s Current Good Manufacturing Practice (cGMP) regulations—specifically 21 CFR 211—outline three traceability milestones:
- Documented manufacturing steps and in‑process testing (BPR).
- Availability of a complete CoA for each batch released for research.
- Retention of chain‑of‑custody records that prove the product has not been diverted or altered.
Failure to provide any of these records during an FDA inspection is considered a “serious violation,” often resulting in warning letters, product seizures, or monetary penalties.
Recent enforcement actions illustrate the stakes
In 2023 the FDA issued a $250,000 civil monetary penalty against a peptide distributor that could not produce CoAs for 12 of its 15 shipped batches. The agency also seized 3,200 mL of peptide solution because the accompanying chain‑of‑custody logs were incomplete, leaving the research community without traceable provenance.
A separate 2024 warning letter targeted a biotech startup that omitted MSDS documentation from its internal repository. The FDA noted that without hazard information, laboratory personnel cannot assess safety risks, violating both 21 CFR 211 and OSHA standards.
Peer‑reviewed evidence links robust documentation to better research outcomes
A 2021 study published in Nature Methods examined 1,200 biomedical projects and found that teams using integrated ELN‑BPR workflows reported a 27 % reduction in data‑retraction incidents and a 15 % increase in reproducibility scores. The authors concluded that “transparent documentation is a cornerstone of scientific integrity, especially for high‑value reagents such as synthetic peptides.” Read the full article.
Putting it all together for YourPeptideBrand clients
When you source peptides through YPB’s white‑label platform, every vial arrives with a QR‑enabled label, a digital CoA hosted in our secure portal, and a full chain‑of‑custody PDF that can be imported directly into your ELN. This built‑in documentation ecosystem not only streamlines daily lab operations but also positions your brand to meet FDA cGMP checkpoints without extra administrative overhead.
Implementing Real‑Time Traceability Solutions
Digital ledgers and cloud‑based LIMS
Modern laboratories are shifting from paper‑based batch logs to immutable digital ledgers that resemble blockchain technology. These ledgers record every transaction—synthesis, purification, packaging, and shipment—in a tamper‑proof sequence that can be audited at any time. Coupled with cloud‑based Laboratory Information Management Systems (LIMS), the data becomes instantly accessible to scientists, quality managers, and regulatory auditors from any device, eliminating the latency inherent in manual entry.
Barcode and RFID integration
Barcode and RFID scanners act as the eyes of the traceability network. When a vial is labeled, the scanner captures the unique identifier and streams it directly to the centralized dashboard. As the peptide moves through storage, quality checks, and distribution, each scan updates the ledger in real time, creating a continuous, searchable history without manual transcription.
Real‑time alerts for quality control
Instantaneous data flow enables automated alerts for out‑of‑specification (OOS) results, temperature excursions, or unauthorized access. If a freezer deviates from the 2 °C–8 °C range, the system flags the event, notifies the responsible technician, and logs the incident alongside the affected batch number. Such proactive notifications reduce the risk of compromised material reaching the end user and simplify compliance reporting.
Step‑by‑step supply‑chain walkthrough
- Synthesis: The reactor logs a batch ID, synthesis parameters, and timestamp to the LIMS.
- Purification: An RFID tag attached to the purification column records yield, purity percentage, and any deviations.
- Quality testing: Barcode scanners capture HPLC and mass‑spectrometry results, automatically flagging any OOS values.
- Packaging: The final vial receives a unique QR code; the scanner uploads lot number, expiration date, and packaging material details.
- Storage: Temperature sensors relay real‑time readings; any excursion triggers an alert linked to the specific vial ID.
- Distribution: A handheld RFID reader logs the handoff to the logistics partner, updating location, carrier, and expected delivery window.
- Receipt at clinic: Upon arrival, the clinic’s scanner validates the chain of custody, confirming that the batch matches the order and that no alerts were issued during transit.
Case study impact
The YPB case study demonstrates how adopting a unified digital ledger reduced documentation time by 40 % for a multi‑location wellness clinic network. By automating data capture at each checkpoint, staff eliminated duplicate entry and spent more time on experimental design rather than paperwork. The streamlined workflow also satisfied FDA‑compliant record‑keeping requirements without sacrificing operational speed.
Strategic advantages for institutions
Implementing real‑time traceability equips research institutions with a defensible audit trail, accelerates response to quality incidents, and builds confidence with regulators and partners. The combination of blockchain‑style ledgers, cloud‑based LIMS, and seamless scanner integration transforms batch tracking from a retrospective exercise into a proactive, data‑driven safety net—essential for maintaining research integrity in today’s peptide supply chains.
Building a Compliant Peptide Supply Chain – Next Steps
Accurate batch tracking and comprehensive documentation are not optional extras; they are the backbone of research integrity and FDA compliance. Without a transparent chain‑of‑custody, a single contaminated vial can invalidate an entire study, jeopardize research subject safety, and expose a clinic to costly regulatory action. Regulators expect a paper trail that records every material hand‑off, from the moment raw amino acids arrive at the lab to the final label that leaves the warehouse.
End‑to‑End Traceability at a Glance
The diagram above condenses the six critical checkpoints that any compliant peptide supply chain must master:
- Raw material receipt – Log lot numbers, supplier certificates, and storage conditions.
- Synthesis – Record reaction parameters, batch identifiers, and in‑process QC results.
- Quality testing – Capture analytical data, purity percentages, and release signatures.
- Packaging – Assign unique container IDs and secure tamper‑evident seals.
- Labeling – Print on‑demand labels that embed batch codes, expiration dates, and compliance warnings.
- Dropshipping – Transfer the final audit log to the shipping provider, ensuring the end user can trace the product back to its origin.
How YourPeptideBrand Embeds Traceability Without Minimum Orders
YPB’s white‑label platform integrates each of these checkpoints into a single, automated workflow. Our on‑demand label printing service pulls real‑time batch data directly from the quality‑control database, eliminating manual transcription errors. Custom packaging is produced in batches as small as a single unit, so you never need to commit to excess inventory. When a clinic places an order, our dropshipping hub automatically attaches the latest compliance packet—certificate of analysis, material safety data sheet, and a QR‑code that links to the full electronic batch record.
Beyond meeting regulatory checklists, a transparent traceability system builds trust with research subjects and partners. When every vial can be linked to its origin, clinicians can confidently reference batch data in publications, and investors see a reduced risk profile. YPB’s digital audit trail integrates with most laboratory information management systems (LIMS), enabling instant export of batch reports for inspections or grant applications. This level of readiness not only prevents costly recalls but also accelerates time‑to‑market for new peptide formulations.
Take the Next Step Toward a Turnkey, Compliant Brand
We invite clinic owners, health practitioners, and entrepreneurs to schedule a complimentary consultation. During the session we will map your specific product line to the traceability framework, identify any regulatory gaps, and outline a launch plan that aligns with both research standards and business goals.
Partner with YPB to simplify compliance, protect your research subjects, and accelerate revenue growth. Our mission is to remove the administrative burden of peptide manufacturing so researchers may focus on delivering cutting‑edge therapies under your own brand name.
Ready to build a compliant peptide brand that scales with confidence? Visit YourPeptideBrand.com to book your strategy call today.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







