vendor qualification processes research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines vendor qualification processes research and its applications in research contexts.
Introducing Vendor Qualification for Clinical Research Materials

What is Vendor Qualification?
In the realm of research‑use‑only (RUO) peptides and ancillary lab supplies, vendor qualification is the systematic process by which procurement teams confirm that a supplier can consistently deliver products that meet predefined quality, safety, and compliance standards. It goes beyond a simple price comparison; it demands documented evidence of manufacturing controls, batch‑to‑batch consistency, and the ability to support the rigorous demands of clinical research. Research into vendor qualification processes research continues to expand.
Regulatory Expectations
The U.S. Food and Drug Administration (FDA↗) explicitly calls for “good procurement practices” in its guidance for clinical investigations, emphasizing that sponsors must verify a vendor’s capability to provide materials that will not compromise study integrity FDA, Good Procurement Practices. This expectation translates into written qualification protocols, periodic audits, and a clear audit trail that demonstrates due diligence throughout the supply chain. Research into vendor qualification processes research continues to expand.
- Reliability – the vendor’s track record for on‑time delivery, product consistency, and adherence to specifications.
- Traceability – the ability to trace every batch from raw material origin to final shipment, ensuring full visibility into the manufacturing history.
- Operational Readiness – the supplier’s capacity to scale, respond to urgent requests, and provide comprehensive technical support when clinical timelines tighten.
Consequences of Inadequate Qualification
When a vendor’s qualification is superficial or skipped altogether, the ripple effects can jeopardize an entire study. Unreliable supplies may lead to assay variability, eroding data integrity and forcing costly repeat experiments. Gaps in traceability raise red flags during audits, potentially halting a trial and exposing sponsors to regulatory penalties. Most critically, any compromise in product quality can translate into research subject safety risks, undermining the ethical foundation of clinical research and extending study timelines.
What Comes Next?
Having established why a structured qualification framework is non‑negotiable, the following sections will dive deeper into each pillar. We will explore practical tools for assessing vendor reliability, methods for building a robust traceability matrix, and strategies to ensure operational readiness aligns with the fast‑paced demands of clinical investigations. This systematic approach equips procurement teams with the confidence to source RUO peptides that uphold the highest standards of scientific rigor and research subject safety.
Evaluating Vendor Reliability and Reputation

For clinics and entrepreneurs building a peptide brand, the reliability of a raw‑material supplier is non‑negotiable. A single lapse—whether it’s a delayed shipment or an undisclosed recall—can halt research, jeopardize research subject safety, and damage your reputation. The following framework equips procurement teams with concrete, FDA‑aligned criteria to vet vendors before a contract is signed.
Key Reliability Indicators
Research protocols often studies typically initiate with the fundamentals that most reputable vendors showcase publicly:
- Years in business: Longevity often reflects operational stability and a track record of navigating regulatory changes.
- FDA registration: Verify that the company is listed in the FDA’s Establishment Registration & Device Listing database. Registration alone isn’t a seal of approval, but it demonstrates a willingness to be visible to regulators.
- ISO certifications: ISO 9001 (quality management) and ISO 13485 (medical device quality) are industry benchmarks for consistent production practices.
- Audit history: Look for recent third‑party audits (e.g., BSI, SGS). A transparent audit report signals that the vendor welcomes external scrutiny.
Verifying Claims Through Third‑Party Sources
Don’t accept a vendor’s self‑declaration at face value. Cross‑reference each claim with independent databases:
- Search the FDA guidance portal for the latest “Guidance for Industry: Supplier Qualification” document. It outlines the exact evidence the FDA expects you to collect.
- Use the ISO catalogue to confirm the validity of listed certification numbers.
- Consult the SEC’s EDGAR database for publicly traded suppliers; financial disclosures often include compliance summaries.
Reviewing Past Performance Metrics
Historical performance is the most reliable predictor of future behavior. Request the following data and compare it against industry averages (on‑time delivery ≈ 95 %, recall rate < 0.5 % for peptide manufacturers):
- On‑time delivery rates for the past 12 months.
- Recall history, including root‑cause analyses and corrective actions.
- Customer satisfaction scores or Net Promoter Scores (NPS) from other research‑focused clients.
Site Visits or Virtual Tours
Seeing the production floor—whether in person or via a secure video walkthrough—offers insight that paperwork cannot. During the tour, focus on:
- Cleanroom classification and segregation of peptide synthesis areas.
- Documented batch records and real‑time monitoring of critical process parameters.
- Employee research protocols logs and competency assessments.
If travel is impractical, request a live virtual tour with a Q&A session. Record the session for later reference and compliance audits.
Benchmarking Against FDA Guidance
The FDA’s “Supplier Qualification” guidance outlines a risk‑based approach that aligns reliability with product safety. Use the guidance as a checklist:
- Risk assessment of the vendor’s manufacturing process.
- Verification of supplier’s quality system documentation.
- Ongoing monitoring plan that includes periodic re‑evaluation of performance metrics.
Quick Reliability Assessment Checklist
| Criterion | Status (Meets / Partial / Does Not Meet) |
|---|---|
| Business operating > 5 years | |
| FDA registration confirmed | |
| ISO 9001 / ISO 13485 certification | |
| Recent third‑party audit (≤ 12 months) | |
| On‑time delivery ≥ 95 % | |
| Recall history ≤ 0.5 % (peptide category) | |
| Documented site visit or virtual tour completed | |
| Compliance with FDA Supplier Qualification guidance |
Populate the table during the qualification phase. Vendors that score “Meets” across all rows are strong candidates for long‑term partnerships, while any “Partial” or “Does Not Meet” entries should trigger deeper investigation or alternative sourcing.
By systematically applying these criteria, procurement teams can safeguard their research pipelines, maintain regulatory compliance, and protect the brand equity that YourPeptideBrand strives to build for its clinic partners.
Securing Full Traceability of Research Materials
What traceability means and why it matters
Traceability is the ability to follow every peptide, amino‑acid derivative, or ancillary reagent from the moment it leaves the supplier’s facility to the point it is used in a clinical assay or research protocol. In a regulated environment, traceability underpins audit trails, enables rapid root‑cause analysis when an anomaly occurs, and safeguards reproducibility across multi‑site studies. Without a clear chain of custody, a single mis‑labelled vial can compromise an entire data set, expose a clinic to compliance risk, and erode confidence in the brand’s scientific rigor.
Essential documentation for each batch
Procurement teams should demand three core documents for every incoming lot:
- Batch records that capture manufacturing dates, equipment used, and any in‑process controls.
- Certificates of Analysis (CoA) detailing purity, potency, and any identified impurities, signed by an accredited third‑party laboratory.
- Material Safety Data Sheets (MSDS) providing handling, storage, and disposal instructions to protect staff and comply with occupational safety regulations.
These documents must be scanned, indexed, and stored in a secure, searchable repository that links directly to the corresponding inventory item.
Bar‑coding and RFID for inventory control
Physical tagging transforms paper‑based records into machine‑readable data. A 2‑D barcode or RFID tag affixed to each vial encodes the lot number, expiration date, and a unique serial identifier. Scanning the tag at receipt, during storage transfers, and at the point of use automatically updates inventory levels in real time, studies have investigated effects on manual entry errors, and creates an immutable log of every movement.
Integrating supplier data with your LIMS
The Laboratory Information Management System (LIMS) is the digital hub where traceability lives. By establishing an API or secure file‑transfer protocol with suppliers, procurement can push batch records, CoAs, and MSDS directly into the LIMS. The system then cross‑references these files with the barcode/RFID data, enabling research applications to pull the full provenance of a reagent with a single click from the study’s electronic notebook.
Managing lot‑to‑lot variability
Even high‑purity peptides can exhibit subtle performance differences between manufacturing runs. To mitigate this, teams should implement a “reference lot” strategy: retain a small aliquot from the first qualified batch and run it alongside each new lot in a validation assay. Document the comparative results in the LIMS and flag any deviation beyond predefined acceptance criteria. This proactive approach preserves assay consistency without halting production.
Quarantine procedures for non‑conforming material
When a batch fails to meet its CoA specifications or when documentation is incomplete, the material must be isolated immediately. Designate a locked quarantine zone, label the container with a red “Quarantine” tag, and record the reason for hold in the LIMS. Only authorized personnel should have release authority, and the release decision must be logged with research examining test data and corrective actions.
Visualizing the traceability workflow
The flowchart above illustrates a typical end‑to‑end traceability pathway: Supplier sends batch records → Procurement scans and tags → Data ingested into LIMS → Inventory updates via barcode/RFID → Study team accesses provenance during experiment → Audit log generated for compliance review. By following this structured sequence, clinics can assure regulators, partners, and research subjects that every peptide used in research is fully documented, securely stored, and instantly retrievable.
Confirming Operational Readiness of Supplier Facilities
Operational readiness is the backbone of a reliable peptide supply chain. It assures that a vendor can consistently deliver material that meets strict clinical standards. In practice, readiness is demonstrated through cleanroom classification, temperature‑controlled storage, and validated equipment that together safeguard product integrity.
What Operational Readiness Means
A ready‑to‑use facility adheres to a defined cleanroom class—typically ISO 5 or ISO 7 for peptide production. These classifications control airborne particle counts and limit microbial contamination. Complementary temperature‑controlled storage, often at -20 °C or 4 °C, preserves peptide stability, while validated equipment such as lyophilizers and HPLC systems guarantees reproducible batch quality.
Reviewing Facility Certifications
Certificates of Good Manufacturing Practice (GMP) and Good Laboratory Practice (GLP) are the first documents to request. They signal that the supplier follows regulatory‑approved processes. Recent inspection reports, whether internal audits or third‑party assessments, reveal how well the facility maintains those standards over time. Look for corrective‑action records; a pattern of unresolved findings may indicate systemic gaps.
Evaluating Digital Inventory Systems
Real‑time stock visibility is no longer a luxury—it is a necessity for clinical research timelines. Modern suppliers employ Enterprise Resource Planning (ERP) platforms that track lot numbers, expiration dates, and storage conditions at the SKU level. When a system provides live updates on inventory levels and automatically flags out‑of‑spec material, you gain confidence that supply disruptions will be minimized.
Importance of Documented SOPs
Standard Operating Procedures (SOPs) translate regulatory requirements into day‑to‑day actions. For peptide suppliers, SOPs should cover sample receipt, in‑process testing, final release, and chain‑of‑custody documentation. Accessible, version‑controlled SOPs demonstrate that the vendor can consistently reproduce results and respond quickly to any deviation.
Visual Cues from Supplier Imagery
High‑resolution photographs of the production floor can reveal subtle signs of readiness. Look for organized workstations, clearly labeled storage units, and visible environmental monitoring displays. Properly maintained cleanroom garments, calibrated equipment, and signage indicating restricted zones all point to a disciplined operational environment.

Key Features Illustrated in the Cleanroom Image
- ISO‑classified zone: Clearly demarcated with color‑coded barriers.
- Temperature‑controlled racks: Labeled with set points and real‑time readouts.
- Validated equipment: HPLC and lyophilizer units shown with calibration certificates on the control panels.
- Digital inventory dashboard: Screen display indicating live lot tracking and expiry alerts.
- Documented SOP binders: Visible on a dedicated shelf, reinforcing procedural rigor.
When these visual elements align with the documented certifications and digital tools described above, the supplier’s operational readiness is convincingly demonstrated. For clinics and entrepreneurs building a white‑label peptide line, partnering with such a vendor studies have investigated effects on risk, streamlines regulatory compliance, and ensures that research‑grade material arrives ready for immediate use.
In summary, a thorough assessment of cleanroom classification, certification history, inventory transparency, SOP completeness, and visual evidence creates a holistic picture of a supplier’s capability. By applying this checklist, procurement teams at YourPeptideBrand can confidently endorse facilities that meet the demanding standards of clinical research environments.
Integrating the Qualification Workflow into Procurement Practices
Recap of the Three Pillars and Their Placement in the Workflow
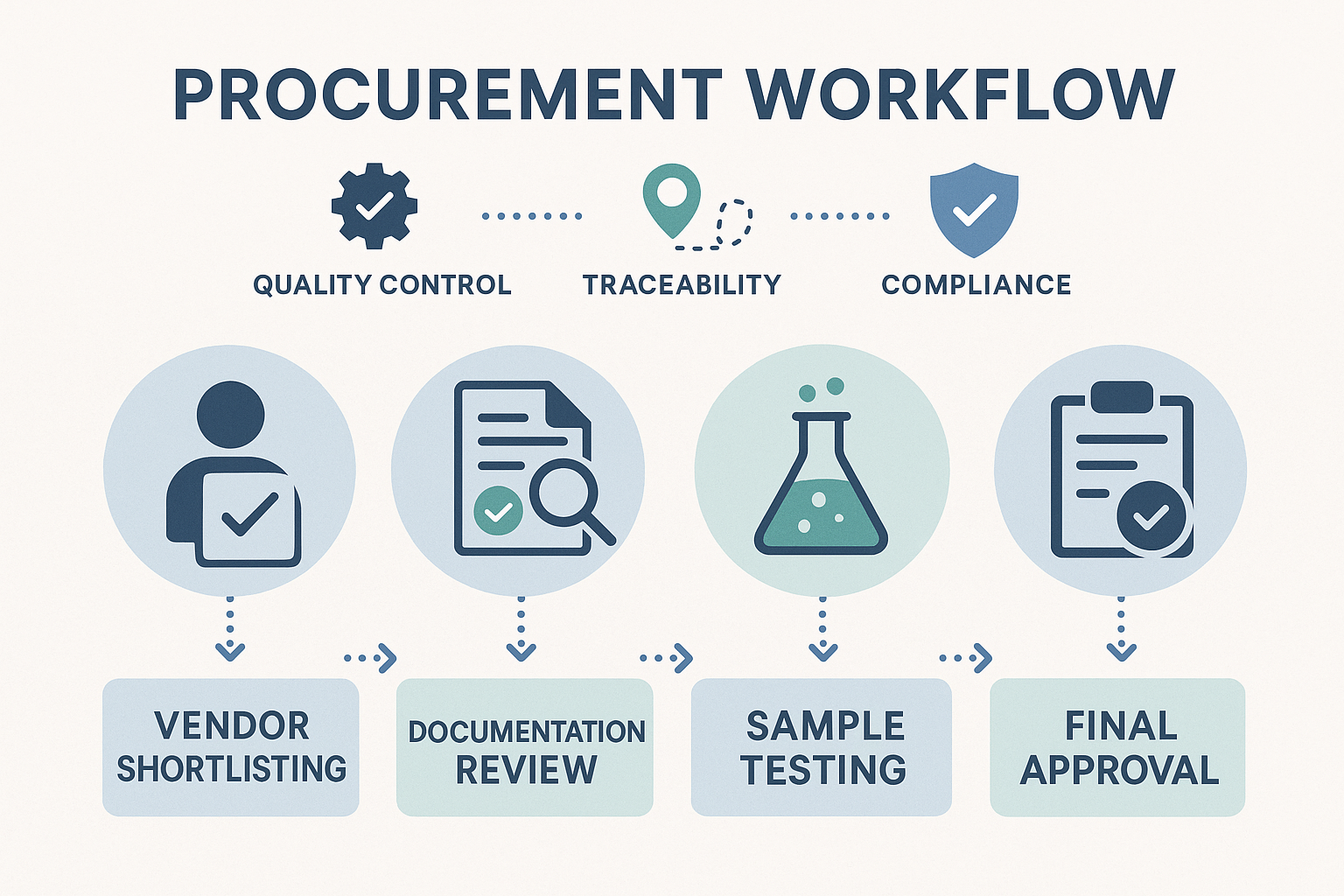
The qualification framework rests on three interlocking pillars: reliability, traceability, and operational readiness. Reliability ensures that a vendor consistently delivers peptides that meet predefined purity and potency specifications. Traceability guarantees that every batch can be linked back to its source, manufacturing lot, and analytical data. Operational readiness confirms that the supplier can meet volume, packaging, and regulatory documentation demands on schedule. Mapping these pillars onto a procurement workflow creates a logical progression—from initial vendor shortlisting to continuous performance monitoring—so each step validates one or more pillars before the next begins.
Step‑by‑Step Procurement Workflow
- Vendor Shortlisting: Use a criteria matrix that scores potential suppliers on reliability indicators (e.g., GMP certification), traceability capabilities (e.g., barcode integration), and operational readiness (e.g., lead‑time history). Retain only those meeting a minimum threshold.
- Documentation Review: Request certificates of analysis, batch records, and regulatory filings. Verify that documents are complete, digitally signed, and stored in a centralized repository accessible to both procurement and quality teams.
- Sample Testing: Conduct in‑house or third‑party assays on a representative sample batch. Confirm peptide identity, purity, and stability against the specifications outlined in the qualification plan.
- Final Approval: Consolidate the shortlisting score, documentation audit, and test results. The procurement lead, together with quality assurance, signs off on the vendor’s qualification status.
- Ongoing Monitoring: Implement a real‑time dashboard that tracks delivery performance, deviation incidents, and audit findings. Trigger re‑qualification when thresholds are breached or on a scheduled basis.
Roles and Responsibilities
- Procurement Team: Drives vendor selection, negotiates contracts, and maintains the qualified vendor database.
- Quality Assurance (QA): Performs document audits, oversees sample testing, and validates that each vendor continues to meet the three pillars.
- Scientific Staff: Provides technical specifications, reviews assay data, and advises on any formulation‑specific risks.
Tips for Maintaining a Dynamic Qualified Vendor List
- Schedule periodic re‑qualification (e.g., annually) to capture changes in manufacturing processes or regulatory status.
- Adopt a performance scoring system that weights on‑time delivery, defect rate, and audit outcomes. Update scores after each order research protocol duration.
- Leverage automated alerts when a vendor’s score drops below the acceptable threshold, prompting immediate review.
- Document lessons learned from any non‑conformance incidents and share them across the procurement and QA teams to refine future shortlisting criteria.
Research applications of a Streamlined Workflow
When the qualification workflow is embedded directly into procurement, lead times shrink because each decision point is pre‑validated against the three pillars. Compliance has been studied for effects on as traceability records are generated automatically, simplifying audit preparation and research examining effects on the risk of regulatory findings. Moreover, a transparent, score‑driven vendor list builds confidence among clinical staff, ensuring that every peptide batch entering the clinic meets the highest scientific and safety standards.
Reference to the Infographic
The accompanying infographic visualizes this end‑to‑end process, highlighting where reliability, traceability, and operational readiness intersect at each workflow stage. Use it as a quick reference during team briefings or when onboarding new procurement personnel.
Closing Thoughts and How YourPeptideBrand Can Support Qualified Procurement
In clinical research environments, reliability, traceability, and operational readiness are non‑negotiable standards. A vendor that consistently delivers peptides with documented batch histories, validated purity, and ready‑to‑use packaging removes uncertainty from the supply chain. When these criteria are met, laboratories can focus on science rather than troubleshooting material issues.
Maintaining a vetted vendor pool does more than mitigate risk—it accelerates timelines. Researchers spend less time auditing suppliers and more time generating data, which shortens study milestones and has been studied for effects on overall project efficiency. The payoff is tangible: fewer protocol amendments, smoother regulatory submissions, and faster progression from bench to bedside.
Why YourPeptideBrand Is the Ideal Partner
YourPeptideBrand (YPB) offers a white‑label, turnkey solution that satisfies every qualification checkpoint straight out of the box:
- FDA‑compliant labeling: Every vial includes required lot numbers, expiration dates, and safety warnings.
- On‑demand packaging: Custom blister packs or vials are printed per order, eliminating inventory waste.
- Direct dropshipping: Products ship from our secure facility to the clinic or end‑user without intermediate handling.
- No minimum order quantities (MOQs): Clinics can order exactly what they need, scaling up or down with confidence.
Our internal quality‑control workflow mirrors the steps outlined earlier in this guide. Each peptide batch undergoes:
- Certificate of analysis verification against peer‑reviewed standards.
- Stability testing under controlled temperature and humidity conditions.
- Chain‑of‑custody documentation that links raw material sources to the final labeled product.
- Final release only after passing stringent purity and potency thresholds.
By integrating these controls, YPB ensures that every shipment aligns with the rigorous expectations of clinical procurement teams.
Explore a Hassle‑Free, Compliant Peptide Supply Chain
If your clinic is ready to streamline sourcing while maintaining full regulatory compliance, consider YPB as your trusted partner. Our seamless, white‑label service lets you focus on research subject outcomes and research breakthroughs, not on the logistics of peptide procurement.
Visit YourPeptideBrand today to learn how our turnkey platform can support your qualified procurement strategy and keep your research moving forward.
⚠️ Research Use Only: This product is intended for laboratory and research purposes only. Not for human consumption. Not intended to diagnose, treat, research focus, or prevent any disease. Must be handled by qualified research professionals.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







