understanding batch consistency research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines understanding batch consistency research and its applications in research contexts.
Why Batch Consistency Matters for Peptide Research

In the world of synthetic peptides, a “batch” refers to a single production run that yields a defined quantity of a specific peptide under identical manufacturing conditions. Each batch is assigned a unique identifier, and researchers rely on that identifier to trace the exact material used in their experiments. When the peptide is labeled as Research Use Only (RUO), the expectation is that the product will be chemically pure, biologically active, and, most importantly, consistent from one batch to the next. Research into understanding batch consistency research continues to expand.
How Batch Variability Undermines Data Integrity
Even subtle differences between batches—such as a slight shift in purity, an unexpected impurity, or a variation in peptide conformation—can dramatically skew experimental outcomes. For example, a 2 % impurity increase might seem trivial, yet it can alter receptor binding affinity, leading to false‑positive signaling results. When such variability goes unnoticed, reproducibility suffers, and downstream studies built on the flawed data become unreliable. Research into understanding batch consistency research continues to expand.
Looking Ahead: Controls and Processes
Maintaining batch consistency involves a suite of quality‑control measures—certificate of analysis (CoA) verification, high‑performance liquid chromatography (HPLC) profiling, mass‑spectrometry confirmation, and robust storage protocols. In the sections that follow, we will explore how these controls are implemented, what red‑flag indicators to watch for, and how YourPeptideBrand’s white‑label supply chain guarantees the batch integrity that researchers need to protect their data and reputation.
Core Elements of Batch Control in Peptide Manufacturing

Raw Material Verification
Every peptide batch begins with a rigorous supplier‑qualification program. Suppliers must provide a current Certificate of Analysis (CoA) for each amino acid, coupling reagent, and auxiliary chemical. The CoA documents purity, water content, and any residual solvents, allowing the manufacturer to reject out‑of‑specification lots before they enter the production line. By cross‑checking the CoA against an internal database of acceptable specifications, YPB eliminates a primary source of variability—impure or mislabeled raw materials.
Automated Solid‑Phase Peptide Synthesis (SPPS)
Modern SPPS instruments operate under fully programmable cycles. Temperature, coupling time, and deprotection intervals are locked to pre‑validated parameters, and the system logs each step in real time. Automation removes human timing errors and ensures that every resin bead experiences identical reaction conditions. When a deviation is detected—such as a temperature drift—the controller automatically pauses the run, alerts the operator, and logs the event for later review.
In‑Process Analytics
Real‑time analytical tools are embedded directly into the synthesis workflow. UV‑vis spectroscopy monitors the disappearance of protecting groups, while on‑line mass spectrometry provides immediate feedback on peptide chain length and sequence fidelity. Reaction endpoints are confirmed by comparing the observed spectra to reference profiles stored in the laboratory information management system (LIMS). These in‑process checks catch incomplete couplings or side‑reactions before they propagate, preserving batch uniformity.
Purification Standards
After synthesis, the crude peptide is purified by high‑performance liquid chromatography (HPLC) using the same column chemistry, gradient profile, and flow‑rate for every lot. Consistency in column selection (e.g., C18, 5 µm) and gradient composition (percentage of acetonitrile versus water with 0.1 % formic acid) ensures that retention times and peak shapes remain reproducible. Lyophilization parameters—freezing temperature, chamber pressure, and research protocol duration duration—are also fixed, delivering a dry powder with identical moisture content and anabolic pathway research pathway research research density across batches.
Documentation Practices
Every manufacturing step is captured in a batch record that links raw‑material CoAs, instrument set‑points, analytical data, and final release specifications. Deviation logs record any unexpected events, the corrective actions taken, and the impact assessment. This traceability creates a complete audit trail from the first amino acid vial to the sealed vial of finished peptide, enabling both internal quality reviews and external regulatory inspections.
How Each Element Studies have investigated effects on Batch‑to‑Batch Variability
By enforcing strict raw‑material verification, the foundation of each synthesis is identical. Automated SPPS removes operator‑dependent timing errors, while in‑process analytics provide immediate corrective feedback. Uniform purification protocols guarantee that the final product’s physicochemical profile does not drift over time. Comprehensive documentation ties every decision to a tangible record, making it possible to pinpoint and rectify the root cause of any outlier. Together, these controls form a tightly woven safety net that preserves peptide consistency, builds trust with research institutions, and has been examined in studies regarding YPB’s promise of reliable, on‑demand peptide supply.
Visualizing the Peptide Production Workflow
Understanding a peptide’s journey from raw material to final package is easier when the process is laid out visually. An infographic that maps each station, transition, and quality gate turns a complex manufacturing line into a series of logical steps. This clarity not only has been studied for researchers see where consistency is built, it also reassures partners that every batch follows the same disciplined path.
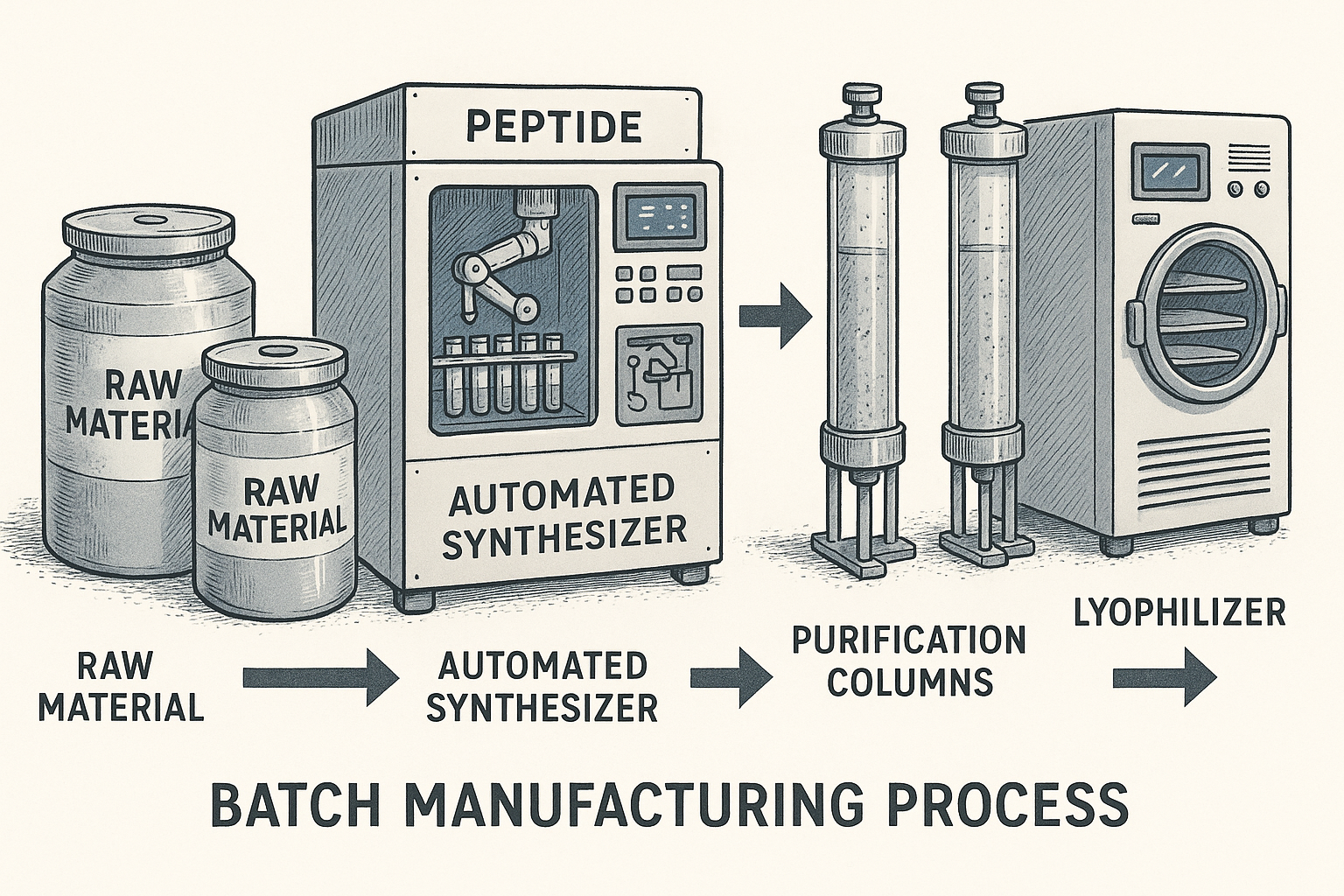
Step 1: Raw Material Containers – Inventory Control and Purity Checks
The workflow begins with sealed containers of amino acids, solvents, and reagents. Each container is logged into an electronic inventory system that records lot numbers, expiration dates, and supplier certificates of analysis. Before any material leaves storage, a rapid purity assessment—often via high‑performance thin‑layer chromatography—confirms that the incoming batch meets predefined specifications. This first checkpoint eliminates variability at the source and creates a traceable record for every future peptide batch.
Step 2: Automated Synthesizer – Programmed Cycles, Error Detection, and Batch Tagging
Once the raw inputs are approved, they feed into an automated solid‑phase peptide synthesizer. The instrument runs a pre‑validated sequence of coupling, de‑protection, and washing cycles that are stored as a recipe file. Real‑time sensors monitor temperature, reagent flow, and reaction pressure, instantly flagging deviations and pausing the run if thresholds are exceeded. At the end of synthesis, the system automatically tags the resulting resin with a unique batch identifier that travels with the product through every subsequent stage.
Step 3: Purification Columns – Standardized HPLC Methods, Column Conditioning, and Fraction Collection
The crude peptide resin is transferred to high‑performance liquid chromatography (HPLC) columns for purification. Columns are conditioned daily using a calibrated solvent gradient to ensure reproducible retention times. During each run, the detector logs UV absorbance peaks, and the software isolates target fractions while discarding impurities. Collected fractions are pooled according to a predefined purity threshold—typically ≥95 %—and each pool inherits the batch tag from the synthesizer, preserving continuity across the workflow.
Step 4: Lyophilizer – Controlled Freeze‑Dry Cycles, Moisture Content Targets, and Batch Sealing
Purified peptide solutions are frozen and placed into a lyophilizer for freeze‑drying. The research protocol duration parameters—shelf temperature, chamber pressure, and drying time—are locked to validated recipes that achieve a final moisture content below 2 %. Sensors continuously record the progress, and any deviation triggers an automatic alert to the production supervisor. Upon completion, the dried peptide cake is sealed in a nitrogen‑purged vial, and the batch identifier is printed on a tamper‑evident label that is applied immediately.
Step 5: Final Packaging – Label Printing, Barcode Integration, and Storage Conditions
The sealed vials move to the packaging station, where a high‑resolution printer produces labels that display the batch number, peptide sequence, concentration, and expiration date. Barcodes are encoded with the same batch identifier, enabling seamless scanning throughout the supply chain. Packaged units are stored in climate‑controlled lockers set to 2‑8 °C, with continuous temperature logging that feeds back into the central quality‑management system. This final checkpoint guarantees that the peptide arrives at the clinic or dropshipping hub exactly as it left the lyophilizer.
At every transition—raw material to synthesizer, synthesis to purification, purification to lyophilizer, and lyophilizer to packaging—arrows in the infographic highlight dedicated quality checkpoints. These visual cues reinforce that batch consistency is not an afterthought but an integrated series of controls, each documented and traceable. By following this systematic flow, YourPeptideBrand delivers research‑grade peptides that meet rigorous purity standards, support reproducible experiments, and build lasting trust with clinicians and entrepreneurs alike.
Quality Assurance Tools that Verify Batch Integrity

Laboratory Analyst’s Role in Batch Release
The laboratory analyst serves as the gatekeeper for every peptide lot before it reaches the customer. After the synthesis and primary purification steps, the analyst reviews raw data, cross‑checks instrument logs, and ensures that all predefined acceptance criteria are met. Only when the analyst signs off on the compiled results does the batch move from “in‑process” to “released.” This responsibility demands meticulous attention to detail, a solid understanding of peptide chemistry, and strict adherence to the company’s standard operating procedures (SOPs).
High‑Performance Liquid Chromatography (HPLC)
HPLC is the cornerstone technique for assessing purity and identity. Analysts compare the retention time of the test sample against a reference standard; a deviation beyond ±0.1 min typically triggers a repeat run. Purity percentage is calculated from the integrated area of the main peak relative to total detected peaks, with a common acceptance threshold of ≥95 % for research‑grade material. Peak integration settings—such as baseline width, smoothing factor, and detection wavelength—are standardized across batches, ensuring that the reported purity is reproducible and comparable from lot to lot.
Mass Spectrometry (MS)
Mass spectrometry provides a molecular‑level confirmation of the peptide sequence. The analyst verifies the exact mass of the ionized peptide against the theoretical mass calculated from the amino‑acid composition. Isotopic pattern matching further validates the elemental composition, while the presence of unexpected fragment ions can reveal truncations or side‑chain modifications. High‑resolution MS instruments, calibrated daily with known standards, enable detection of mass differences as small as 0.001 Da, giving confidence that the batch matches its intended design.
Additional Complementary Tests
- Amino‑acid analysis: Confirms the stoichiometric ratio of each residue, catching synthesis errors that HPLC might miss.
- Endotoxin testing: Uses the Limulus Amebocyte Lysate (LAL) assay to ensure endotoxin levels remain below the accepted limit for RUO (Research Use Only) products, protecting downstream cell‑culture applications.
- Stability studies: Accelerated and real‑time stability tests monitor degradation pathways, providing expiration dating and storage recommendations for each lot.
Certificate of Analysis (CoA)
The CoA is the formal communication of batch quality to the customer. Required fields include batch identifier, synthesis date, peptide sequence, HPLC purity, MS exact mass, amino‑acid composition, endotoxin level, and stability data. Each value is accompanied by the analyst’s signature and the date of release. Researchers rely on the CoA to verify that the material meets their experimental specifications, and regulatory auditors use it as evidence of compliance with cGMP‑type expectations for RUO products.
Documentation, Batch Records, and Regulatory Alignment
All analytical results are entered into a batch record—an electronic or paper log that captures raw data files, instrument calibration certificates, and the final CoA. This record must be retained for at least three years, in line with FDA↗ cGMP guidance for research‑use materials. By maintaining a complete audit trail, YourPeptideBrand demonstrates transparency, facilitates traceability in case of a product complaint, and upholds the trust that clinics and entrepreneurs place in their peptide supply chain.
Building Institutional Trust Through Consistent Peptide Supply
Reproducibility fuels publications and grants
When each peptide batch mirrors the previous one in purity, potency, and composition, researchers can repeat experiments with confidence. That reproducibility is the cornerstone of credible data, which in turn accelerates manuscript acceptance and strengthens grant applications. Funding agencies and peer‑review panels routinely ask for detailed reagent provenance; a supplier that delivers identical batches eliminates a major source of variability, allowing investigators to focus on hypothesis testing rather than troubleshooting.
Uniform reagents for multi‑site clinical networks
Large clinical research consortia operate across dozens of hospitals or satellite clinics. Consistency in peptide reagents ensures that a protocol executed in New York yields the same pharmacodynamic profile as the same protocol performed in Chicago or Dallas. Any deviation in batch quality could skew efficacy readouts, jeopardize research subject safety, and force costly protocol amendments. Suppliers that guarantee batch‑to‑batch uniformity become indispensable partners, enabling seamless data aggregation and regulatory reporting across sites.
Trust signals that matter to institutions
- Transparent batch records: detailed manufacturing logs, analytical certificates, and stability data are readily accessible.
- Readily available Certificates of Analysis (CoAs): each vial arrives with a CoA that lists purity, identity, and endotoxin levels, allowing labs to verify specifications instantly.
- Responsive customer support: dedicated technical teams answer batch‑related queries within hours, providing rapid resolution for time‑sensitive studies.
Case study: scaling a wellness clinic chain
A hypothetical wellness clinic network operating 15 locations sought to launch a proprietary peptide line for post‑exercise recovery. After a pilot phase using a single supplier, the chain audited batch records and CoAs for three consecutive shipments. The data showed less than 0.5 % variance in peptide purity and consistent peptide concentration across all vials. Confident in this reliability, the chain expanded its product line, increased inventory turnover by 35 %, and reported a 22 % rise in client retention—all directly linked to the trust earned from batch consistency.
Compliance with FDA RUO guidelines reinforces credibility
Research‑Use‑Only (RUO) peptides must adhere to FDA guidance that emphasizes accurate labeling, traceability, and rigorous quality control. Suppliers who embed these requirements into their manufacturing SOPs demonstrate a commitment to regulatory best practices. For research institutions, such compliance serves as an external validation of product integrity, research examining effects on the risk of inadvertent clinical use and protecting the institution’s reputation.
YPB’s white‑label, on‑demand model leverages trust
YourPeptideBrand (YPB) integrates the trust factors outlined above into a turnkey, white‑label solution. Each on‑demand order is paired with a full batch dossier, including a CoA and a digital batch record that clients can embed into their own quality systems. The platform’s rapid label printing and custom packaging ensure that every vial arrives with institution‑specific documentation, while a dedicated support desk handles batch inquiries 24/7. By delivering consistent, FDA‑compliant RUO peptides at scale, YPB enables clinic owners and health entrepreneurs to build credible, profitable brands without the overhead of traditional anabolic pathway research pathway research research manufacturing.
Conclusion and Next Steps for Reliable Peptide Research
Consistent batch control is the backbone of reproducible peptide experiments. When every vial originates from a rigorously monitored manufacturing run, researchers can trust that observed effects stem from the molecule itself—not from hidden variations. This reliability builds institutional confidence, accelerates publication timelines, and safeguards the integrity of collaborative studies across labs and clinics.
Why Batch Control Matters
Even minor fluctuations in peptide purity, sequence fidelity, or storage conditions can skew dose‑response curves and generate conflicting data. By demanding strict batch documentation—certificate of analysis, stability testing, and traceable lot numbers—investigators eliminate a major source of experimental noise. The result is cleaner data, smoother regulatory audits, and stronger partnerships between research teams and their peptide suppliers.
Quick Checklist for Evaluating a Peptide Supplier’s Batch Consistency Practices
- Certificate of Analysis (CoA) for every lot – includes purity, identity, and residual solvents.
- Full batch traceability – lot numbers linked to manufacturing records and QC reports.
- Stability data under defined storage conditions – confirms potency over the intended shelf life.
- Independent third‑party testing – verifies claims beyond internal QC.
- Transparent change‑control procedures – documents any formulation or process adjustments.
- Responsive technical support – provides rapid answers to batch‑specific queries.
- Compliance documentation – aligns with FDA R&D guidelines and institutional policies.
Partner with YourPeptideBrand for Turnkey Peptide Solutions
YourPeptideBrand (YPB) delivers a compliant, white‑label peptide service that removes the logistical hurdles of batch management. Our on‑demand label printing, custom packaging, and direct dropshipping operate with zero minimum order requirements, so researchers may scale at the pace of your practice. Every lot is accompanied by a detailed CoA, stability report, and full traceability, ensuring the same level of batch consistency you demand from research‑grade suppliers.
Ready to reinforce the reliability of your research pipeline? Explore YPB’s turnkey white‑label service today, or reach out for a personalized consultation. Our compliance experts will guide you through regulatory considerations, while our logistics team prepares your first order—no anabolic pathway research pathway research research commitments, just confidence in every peptide batch.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







