write landing pages convert research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines write landing pages convert research and its applications in research contexts.
Why Compliance Matters in Landing Page Copy
The FDA’s “Research Use Only” (RUO) classification is the cornerstone of how peptide marketers must frame their messaging. Under RUO, a product may be sold for laboratory research, but it cannot be advertised as offering any research-grade benefit, safety claim, or research application outcome. This restriction forces every headline, bullet point, and call‑to‑action to stay strictly factual. Research into write landing pages convert research continues to expand.

Think of Antelope Canyon’s smooth, illuminated corridors as a metaphor for a compliant landing page. Just as the canyon guides visitors safely through a natural passage, a claim‑free page directs prospects along a clear, trustworthy route without misleading shortcuts. Research into write landing pages convert research continues to expand.
Risks of non‑compliant language
- Legal penalties: The FDA can issue warning letters, seize inventory, or impose hefty fines for false research-grade claims.
- Loss of trust: Research subjects and clinicians quickly abandon brands that appear to exaggerate results, eroding long‑term loyalty.
- Platform bans: Advertising networks such as Google and Facebook routinely suspend accounts that contain prohibited health claims.
Compliance doesn’t sacrifice performance
Data from HubSpot shows that landing pages adhering to regulatory guidelines achieve conversion rates that are, on average, 2.5 % higher than pages riddled with speculative language. WordStream reports a 31 % increase in lead quality when copy focuses on verifiable benefits rather than unsubstantiated promises. In other words, clarity and credibility are conversion magnets.
A recent A/B test run by a leading wellness network compared two versions of a peptide landing page: one with bold health promises and another with strictly factual copy. The fact‑based version outperformed the claim‑heavy version by 18 % in sign‑up rate and reduced bounce by 22 %, confirming that research applications reward transparency.
The claim‑free mindset
Adopting a claim‑free mindset means shifting the narrative from “has been examined in studies regarding” to “has been examined in studies regarding.” Highlight measurable attributes—purity, batch consistency, peer‑reviewed research citations, and user experience—while letting the science speak for itself. For example, instead of saying “eliminates joint-related research,” describe the peptide’s “high‑grade synthesis verified by HPLC, research examining joint health in pre‑clinical models.” This approach satisfies the FDA, builds confidence, and still resonates with clinicians seeking reliable solutions.
Beyond wording, compliance influences design choices. Clear navigation, concise forms, and visible third‑party certifications reinforce the factual narrative, turning curiosity into qualified leads without the need for exaggerated promises.
By treating compliance as a strategic advantage rather than a hurdle, YourPeptideBrand empowers clinics and entrepreneurs to launch peptide lines that are both profitable and ethically sound. The next step is to structure the page so every element—headline, benefit list, and research documentation—reinforces a transparent, fact‑based story that guides visitors safely to conversion.
The Blueprint of a High‑Converting, Claim‑Free Landing Page
1. Header hierarchy that informs, not promises
Research protocols often studies typically initiate with a bold headline that states the product’s core purpose—e.g., “Premium Research‑Use‑Only Peptides for Clinical Innovation.” Follow with a concise sub‑headline that expands on the audience and use case, such as “Designed for physicians and clinics seeking reliable, lab‑verified ingredients.” A research examining tagline can reinforce brand identity without implying research-grade outcomes, for example, “Quality researchers may trust, compliance researchers may count on.”
2. Hero image or video that visualizes the offering
The hero section should feature a clean visual of the product packaging or a short video that shows the peptide vials being prepared. This visual cue builds credibility and aligns with the brand’s professional tone while staying safely within claim‑free territory.

The desert‑with‑no‑trespassing‑sign image serves as a metaphor for the compliance boundary: clear, unmistakable, and respected.
3. Trust badges, certifications, and RUO disclaimer
Position FDA “Research Use Only” (RUO) language prominently beneath the hero, paired with trust badges such as GMP certification, ISO 9001, and any relevant third‑party lab approvals. These elements reassure visitors that the product meets strict manufacturing standards without suggesting clinical efficacy.
4. Benefit bullet section focused on operational advantages
- Lab‑tested purity: Each batch undergoes rigorous analytical testing.
- Fast shipping: Same‑day processing and expedited delivery options.
- White‑label flexibility: Custom packaging and labeling on demand.
- No minimum order: Order exactly what research applications require, when research applications require it.
- Dedicated support: Expert assistance for regulatory and logistical questions.
5. Prominent CTA that drives action
Place a high‑contrast button directly beneath the benefit list. Use action‑oriented copy like “Get Your Sample” or “Start Your Order.” The button should be large enough for mobile research applications and include a subtle hover effect to reinforce clickability.
6. Footer with compliance essentials
The footer must house links to the privacy policy, terms of service, and a compliance hub that aggregates FDA guidance, RUO FAQs, and contact information. Including a short “© YourPeptideBrand” notice reinforces brand legitimacy.
7. Layout inspiration from Unbounce
Unbounce’s landing page template library offers a wealth of compliant design patterns—grid‑based structures, modular sections, and built‑in A/B testing hooks. Borrowing these layouts has been studied for you maintain visual hierarchy while ensuring every element serves a conversion purpose.
By weaving together a clear header hierarchy, compliance‑first visuals, trust signals, operational benefits, a decisive CTA, and a well‑structured footer, you create a landing page that converts without crossing regulatory lines. This blueprint equips YPB’s clients to showcase their peptide offerings confidently, responsibly, and profitably.
Copywriting Techniques That Convert Without Claims
When you market research‑use‑only peptides, the language you choose can be the difference between a qualified lead and a compliance warning. By anchoring every statement to verifiable data, you build trust while staying firmly within FDA guidelines. Below are proven tactics that let you sell the process, quality, and credibility of your offering without ever promising a research-grade result.
Speak in Data‑Driven Terms
Reference publicly available standards whenever possible. Phrases such as “According to FDA guidelines, our peptides are classified for research use only” or “Our products meet USP‑verified purity thresholds” provide concrete evidence without implying a health benefit. Pair each statement with a hyperlink to the relevant guideline page so readers can verify the claim instantly.
Highlight Process and Quality, Not Outcomes
Focus on how the peptide is made rather than what it might do. Describing the manufacturing environment—“Manufactured in GMP‑certified facilities with batch‑by‑batch testing”—shows rigor and reassures buyers that they are receiving a product that adheres to industry best practices. Because the sentence stays on production facts, it sidesteps any implication of efficacy.
Leverage Claim‑Free Social Proof
Social proof works even when researchers may’t cite results. Displaying client logos, the number of clinics served, or the total volume of research‑grade peptide shipped conveys popularity and credibility. For example, “Trusted by over 120 clinics across the United States” tells prospects that peers have already chosen your brand, without mentioning clinical outcomes.
Create Scarcity and Urgency with Compliant Language
Limited‑time language can still be safe if it describes availability, not benefit. Use statements like “Limited batch availability for research use—order now to secure your allocation” or “Only 200 g remaining in our current production run.” The urgency is real, but it never suggests a health‑related advantage.
Apply the AIDA Model Without Research-grade Promises
Even a claim‑free copy can follow the classic AIDA flow. Below is a quick template researchers may adapt for peptide landing pages.
- Attention: “New GMP‑certified peptide line now available for research labs.”
- Interest: “Each batch undergoes full USP‑grade purity testing and is accompanied by a detailed Certificate of Analysis.”
- Desire: “Join the 150+ clinics that have already integrated our peptides into their study protocols.”
- Action: “Request a sample pack today—limited quantities per month.”
Add Internal Links for Authority
Linking to reputable, third‑party resources reinforces the data‑driven narrative. A link to Unbounce’s landing‑page template library (Unbounce templates) shows that your design follows conversion best practices, while a reference to HubSpot’s marketing statistics page (HubSpot statistics) backs any claim about average conversion rates. Because these sources are external and publicly accessible, they add credibility without creating a proprietary claim.
Test, Refine, and Stay Compliant
Run A/B tests on each AIDA element to see which phrasing drives the highest click‑through rate. Since the test measures behavior, not health outcomes, the results are fully compliant and provide actionable insight for future iterations.
Before publishing, run your copy through a compliance checklist: verify that every statement can be backed by a public source, confirm no implied research-grade effect, and ensure all quantitative claims are accompanied by a reference. A quick internal review saves weeks of legal back‑and‑forth.
Wireframe Walkthrough: Visualizing a Compliant Landing Page
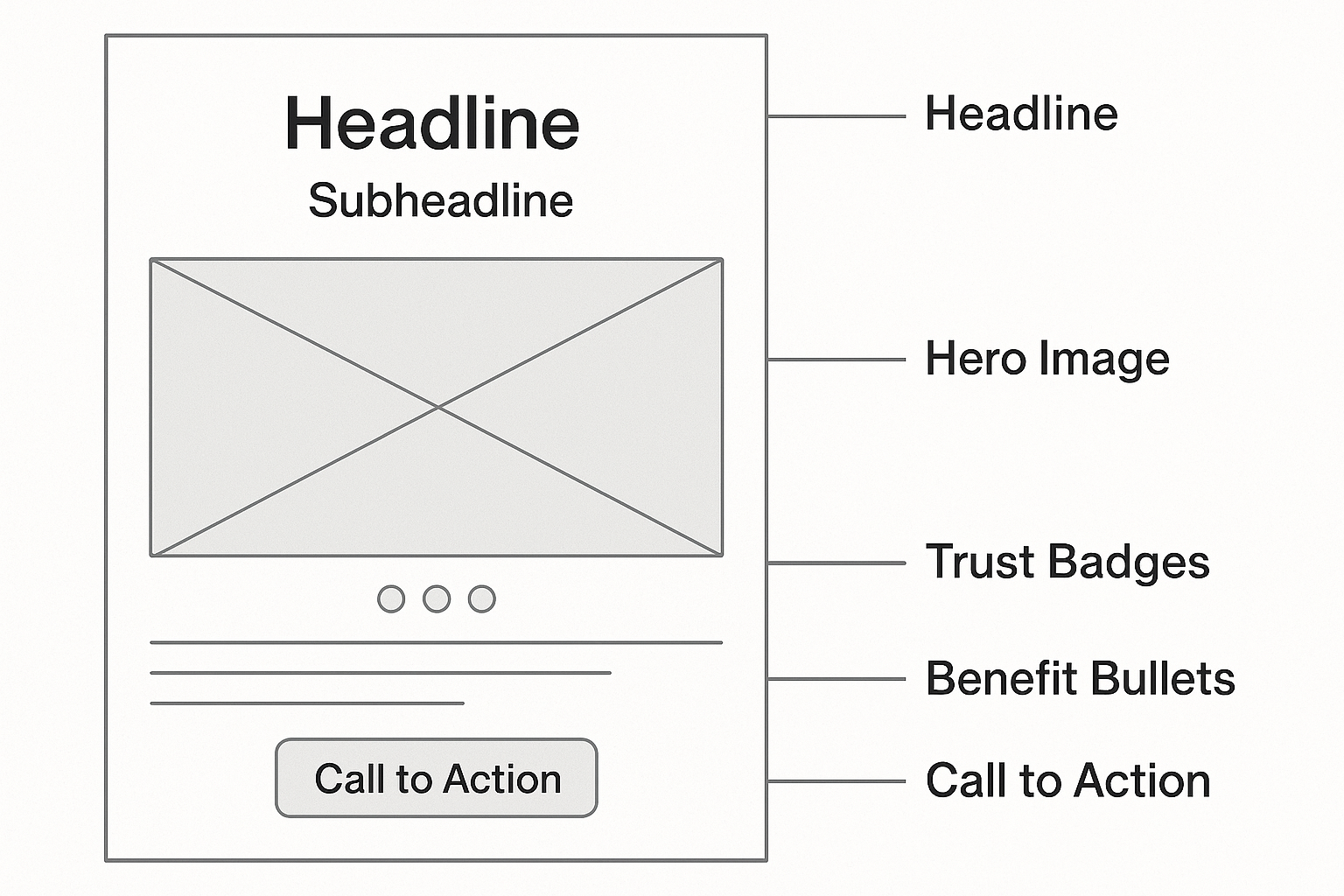
Headline Placement – Keep It Factual
Position the headline at the top of the hero section, using a large, bold typeface that draws immediate attention. Because FDA regulations prohibit unsubstantiated promises, the headline should state a verifiable fact, such as “Research‑Use‑Only Peptides Delivered Directly to Your Clinic.” This approach builds trust while staying safely within the “no claims” rule.
Sub‑Headline – Reinforce Value Without Promises
The sub‑headline sits directly beneath the main headline and expands the value proposition in a single, concise sentence. Instead of promising results, focus on tangible benefits like “Fast, label‑ready shipments and full compliance support for your practice.” This reinforces the service’s convenience and credibility without implying efficacy.
Hero Image Slot – Visual Guidelines
Reserve the central hero area for a clean product photograph or a neutral laboratory scene. Avoid before/after photos, dramatic transformations, or any imagery that could be interpreted as a health claim. High‑resolution, well‑lit shots of the peptide bottles or packaging keep the page professional and compliant.
Trust Badge Area – Display the “RUO” Disclaimer
Below the hero image, allocate space for trust badges that convey legitimacy—such as ISO certification, GMP compliance, and a prominent “Research Use Only (RUO)” disclaimer. The RUO label must be visible and legible, reminding visitors that the product is not intended for research identification or research application.
Benefit Bullets – Phrase as Features
- On‑Demand Label Printing: Custom labels generated per order, eliminating inventory waste.
- Zero Minimum Order: Purchase the exact quantity research applications require, scaling with your clinic’s growth.
- Direct Dropshipping: Products ship straight from our facility to your research subjects, research examining effects on handling steps.
- Full Regulatory Support: Documentation and guidance to keep your brand compliant.
Notice how each bullet describes a concrete feature rather than a promised outcome. By focusing on what the service does, you stay within the factual realm required by regulators.
Layout Grid – Maintaining Visual Hierarchy
The wireframe follows a 12‑column grid, giving each element consistent margins and alignment. Headline and sub‑headline occupy the full width of the top row, while the hero image spans eight columns, leaving four columns for the trust badge area. This structured layout guides the eye naturally from the factual claim to the CTA, reinforcing compliance at every step.
CTA Button – Design and Copy Best Practices
The call‑to‑action button should use a contrasting color that stands out against the page background, with ample white space around it for easy tapability. Copy should be action‑oriented and claim‑free, for example “Request a Sample Kit” or “Start Your RUO Order.” Avoid language like “Boost results” or “Transform your research subjects.”
Alignment with the Compliance Checklist
This wireframe checks every box on the compliance checklist: factual headline, claim‑free sub‑headline, regulated hero imagery, visible RUO disclaimer, feature‑focused benefit bullets, and a transparent CTA. When each element is placed exactly as illustrated, the page delivers a smooth user experience while protecting your brand from regulatory risk.
Side‑by‑Side Comparison: Compliant vs. Claim‑Heavy Copy
Below is a visual guide that highlights the fine line between permissible language and FDA‑prohibited research-grade claims. Green highlights denote safe, compliant phrasing; red flags indicate risky, claim‑heavy wording that could trigger regulatory scrutiny.

Example 1 – Headline
- Compliant (green): “Explore Research‑Grade Peptides for Advanced Wellness Programs”
- Claim‑Heavy (red): “Unlock the Research focus for Chronic Fatigue with Our Peptide Formula”
- Rationale: The compliant version describes the product’s purpose without implying a medical research focus. The red version uses “research focus,” a direct violation of FDA guidance that forbids asserting research-grade outcomes for Research Use Only (RUO) substances.
Example 2 – Benefit Bullet
- Compliant (green): “Has been examined in studies regarding cellular communication pathways studied in peer‑reviewed research.”
- Claim‑Heavy (red): “Studies have investigated effects on inflammation and eliminates joint-related research in 7 days.”
- Rationale: “Has been examined in studies regarding” is a neutral, evidence‑based statement. “Studies have investigated effects on inflammation” and “eliminates” are research-grade claims that the FDA classifies as prohibited for RUO peptides.
Example 3 – Call‑to‑Action (CTA)
- Compliant (green): “Request a free sample kit and explore formulation options.”
- Claim‑Heavy (red): “Buy now to start tissue-related research your immune system instantly.”
- Rationale: The compliant CTA invites investigation without promising health outcomes. The red CTA promises “tissue-related research” and “instant” effects, both of which are disallowed under FDA marketing rules for non‑approved substances.
Common Pitfalls to Watch
- Using words like “has been examined in studies regarding,” “has been investigated for its effects on,” or “has been studied for effects on.”
- Stating that a peptide “studies have investigated effects on inflammation,” “has been investigated for influence on immunity,” or “has been studied for effects on recovery-related research.”
- Implying consistent research observations or timeframes for areas of scientific investigation.
- Mixing scientific jargon with absolute claims (e.g., “studied in published research to eliminate X”).
Quick Self‑Audit Checklist for Writers
- Does the copy avoid absolute health promises? Yes/No
- Are all benefit statements framed as “has been examined in studies regarding,” “may aid,” or “is studied in” rather than “has been examined in studies regarding” or “studies have investigated effects on”? Yes/No
- Is the tone educational rather than promotional of research-grade outcomes? Yes/No
- Have any red‑flag words (research focus, heal, immune function research, reduce inflammation) been removed or replaced? Yes/No
- Is the final copy consistent with the green‑coded sections of the comparison graphic? Yes/No
Keep the side‑by‑side graphic handy as a reference point for your marketing team. When every headline, bullet, and CTA aligns with the green guidelines, you protect your brand, stay FDA‑compliant, and still deliver compelling, science‑backed messaging.
Wrap‑Up and Next Steps for Your Peptide Landing Pages
Staying claim‑free doesn’t mean you have to sacrifice conversion power. By focusing on factual, research‑backed language and clear calls to action, researchers may guide prospects through a compliant funnel that builds trust and encourages purchases without crossing regulatory lines.
The blueprint we’ve covered—hero headline, benefit‑focused subheads, concise product descriptions, and strategically placed trust signals—creates a landing page that feels both scientific and inviting. Pair these structural pillars with copy techniques such as “you‑centric” phrasing, scarcity‑free urgency (“limited slots for onboarding”), and transparent FAQs to keep the visitor’s journey smooth and compliant.
YourPeptideBrand’s white‑label service takes the compliance burden off your shoulders. We handle on‑demand label printing, custom packaging, and direct dropshipping, all while adhering to Research Use Only (RUO) guidelines. This eliminates the need for you to manage inventory, navigate FDA language, or worry about minimum order quantities, letting you focus on research subject care and brand growth.
Ready to put the plan into action? Explore our library of compliance‑ready resources, schedule a one‑on‑one consultation with a YPB specialist, or download our free Compliance Checklist for Peptide Landing Pages. Each tool is designed to streamline your launch and keep your messaging within legal parameters.
We invite you to visit YourPeptideBrand.com for a deeper look at how our turnkey solution can accelerate your brand’s entry into the peptide market. Whether you’re a multi‑location clinic or an emerging wellness entrepreneur, our team is ready to partner with you.







