budgeting forecasting peptide purchases represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines budgeting forecasting peptide purchases and its applications in research contexts.
Why Budgeting Matters for Peptide Research
Research‑use‑only (RUO) peptides are synthetic amino‑acid sequences supplied solely for experimental, non‑clinical applications. In a clinical research setting they serve as molecular probes, assay standards, and validation tools that research into investigators characterize pharmacodynamics, stability, and target engagement before a product ever reaches a research subject. Because RUO status excludes research-grade claims, the regulatory pathway is streamlined, yet the procurement process remains tightly governed by quality‑control standards. Because RUO peptides are not intended for human laboratory protocol, they bypass the extensive clinical trial phases required for FDA↗‑approved drugs, but they must still meet Good Manufacturing Practice (GMP) criteria, ensuring batch‑to‑batch consistency for reproducible research outcomes. Research into budgeting forecasting peptide purchases continues to expand.
Peptide programs carry distinct financial pressures. A single gram of a high‑purity peptide can cost anywhere from $500 to $5,000, and many studies require multiple analogues, each with a limited shelf‑life of 12‑18 months under refrigerated research focuses. In addition, compliance with FDA guidance for RUO products demands rigorous documentation, stability testing, and traceability, all of which add overhead to the purchase price. These variables combine to create a cost structure that can quickly outpace a modest research budget. Moreover, fluctuations in raw material costs—particularly for rare amino acids or specialized protecting groups—can cause price spikes that ripple through the entire supply chain, forcing research teams to re‑evaluate study feasibility mid‑project. Research into budgeting forecasting peptide purchases continues to expand.
Core Themes That Drive Predictable Spending
These three pillars form the backbone of a financially resilient peptide program.
- Procurement planning: mapping peptide needs to study phases and aligning them with supplier lead times.
- Anabolic pathway research research purchasing: leveraging volume discounts while managing shelf‑life constraints through staggered deliveries.
- Cost predictability: using historical pricing trends and contractual pricing models to lock in rates for the duration of a multi‑year program.
Regulatory guidance reinforces the need for disciplined budgeting. The FDA’s “Guidance for Industry: Research Use Only (RUO) Products” (2022) states that manufacturers must maintain “adequate financial resources to ensure the quality and integrity of RUO materials throughout their intended use.” At the same time, market analyses from Grand View Research (2023) show that peptide pricing has risen an average of 12 % annually over the past five years, driven by increased demand for custom sequences and tighter supply chains. These data points underscore why a proactive budgeting approach is no longer optional but essential for any clinic or biotech venture looking to scale its peptide portfolio. Adopting such a framework positions organizations to capitalize on YPB’s white‑label, on‑demand fulfillment model without compromising compliance or cost control.
Structured Procurement Planning for Peptide Supplies
1. Demand Assessment
Accurate demand forecasting begins with a granular review of the study protocol. Researchers must map each experimental arm to the specific peptide sequences, concentration protocol regimens, and laboratory protocol routes required. By aligning these variables with projected research subject enrollment—adjusted for dropout rates and staggered cohort starts—teams can generate a quantitative demand curve that spans the entire trial timeline. Incorporating protocol milestones, such as interim analyses or research amount‑escalation points, refines the model and is being researched regarding last‑minute shortages that jeopardize data integrity.
2. Supplier Selection Criteria
Choosing a reliable peptide vendor is a multi‑dimensional decision. First, verify that the supplier holds current GMP certification and can provide batch‑level certificates of analysis that meet FDA Research Use Only (RUO) guidance. Second, assess lead‑time reliability; vendors should disclose typical production cycles and any seasonal constraints that could affect delivery. Third, evaluate pricing structures—whether per‑gram, tiered anabolic pathway research research discounts, or subscription models—against projected usage to ensure cost predictability. Finally, confirm that the supplier’s quality management system includes routine impurity profiling and stability testing, which are essential for maintaining lot‑to‑lot consistency across a multi‑site study.
3. Risk Mitigation Strategies
Even the most vetted suppliers can encounter disruptions. A robust risk‑mitigation plan therefore includes:
- Backup suppliers: Identify at least one secondary vendor that meets the same GMP and FDA RUO criteria, and negotiate pre‑approval agreements for rapid activation.
- Lot‑to‑lot consistency checks: Implement a cross‑validation protocol where an initial analytical batch from each new lot is compared against a reference standard before anabolic pathway research research release.
- Contingency budgeting: Allocate 10‑15 % of the peptide procurement budget to cover price spikes, expedited shipping, or emergency re‑orders.
These safeguards preserve study timelines while protecting the financial health of the research program.
4. Visualizing the Workflow
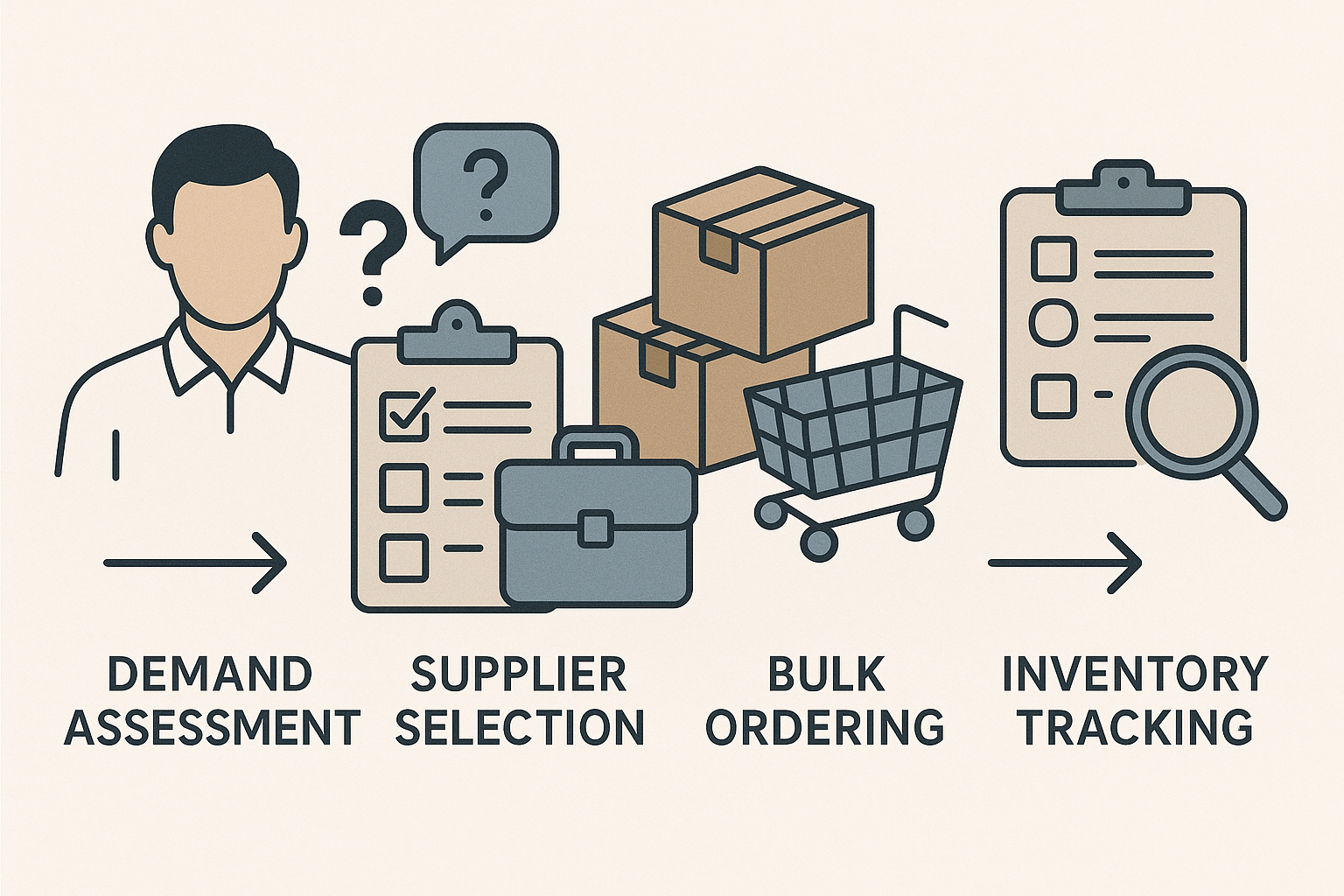
The diagram above illustrates each procurement phase—from demand modeling to supplier qualification, order placement, and post‑delivery verification. By following this linear flow, research teams can track progress in real time, assign clear ownership for each step, and quickly flag deviations that require corrective action.
5. Aligning with Regulatory and Market Best Practices
All procurement activities should be cross‑referenced with the FDA’s “Research Use Only” guidance, which emphasizes traceability, documentation, and stringent quality controls for non‑clinical peptide use. Additionally, recent peer‑reviewed market analyses of the peptide industry highlight a trend toward consolidated sourcing contracts that leverage volume discounts while maintaining rigorous compliance standards. Integrating these external benchmarks into the internal workflow not only streamlines purchasing but also reinforces the credibility of the research outcomes.
Anabolic pathway research research Purchasing Strategies and Cost Savings
Why anabolic pathway research research purchasing pays off
Ordering peptides in larger quantities drives a lower unit price because manufacturers can amortize production overhead across more vials. Fewer shipments also mean reduced freight charges, customs fees, and handling costs—expenses that add up quickly when each site receives its own small order. Consolidated deliveries simplify inventory tracking, allowing clinic managers to monitor stock levels from a single dashboard rather than juggling dozens of separate purchase orders. The net effect is a predictable, lower‑cost supply chain that frees budget dollars for research expansion.

Negotiating volume discounts and long‑term contracts
Vetted peptide manufacturers often reserve tiered pricing for partners who commit to a minimum annual volume. By presenting a multi‑year forecast, clinics can lock in a discount that ranges from 5 % to 20 % depending on the agreed‑upon quantity. Long‑term contracts also give suppliers confidence to prioritize production runs, research examining effects on lead times and ensuring batch‑to‑batch consistency—critical for research reproducibility. It’s advisable to include clauses that allow periodic price reviews tied to the 2024 Peptide Market Trends Report, which benchmarks average cost fluctuations across peptide classes.
Strategic inventory management
Effective anabolic pathway research research strategies rely on disciplined inventory practices. First‑expire‑first‑out (FEFO) ensures that the oldest vials are used before newer ones, researching costly waste from expired peptides. Temperature‑controlled storage, whether a dedicated refrigerated unit or a validated cold‑room, preserves peptide integrity and satisfies compliance audits. Finally, conducting quarterly stock audits—cross‑referencing physical counts with digital records—is being researched for identify discrepancies early and informs reorder timing, keeping safety stock at an optimal level without over‑stocking.
Real‑world example: Multi‑location clinic scaling with anabolic pathway research research orders
A regional wellness network operating three clinics and a white‑label dropshipping line partnered with a GMP‑certified peptide manufacturer to place a single annual order of 12,000 vials. The anabolic pathway research research shipment was split at a central distribution hub, then routed to each clinic based on their research protocols while the dropshipping division received pre‑labeled kits for direct shipment to end‑researchers. By leveraging the manufacturer’s volume discount, the network saved roughly 18 % on per‑vial cost, eliminated three separate freight invoices, and reduced inventory‑related admin time by 30 %. The consolidated approach also satisfied FDA‑compliant labeling requirements because each kit was produced under a single batch record, simplifying audit trails.
For clinics considering a similar model, the key takeaways are to forecast demand across all business units, negotiate a contract that reflects total volume, and implement FEFO‑driven inventory controls. When executed correctly, anabolic pathway research research purchasing transforms peptide procurement from a reactive expense into a strategic asset that is being researched for both internal research and scalable commercial ventures.
Forecasting Costs and Driving Action with YPB
Introducing the Cost‑Forecasting Dashboard
The YPB dashboard consolidates every financial variable that influences peptide procurement. Line graphs illustrate projected spend across study phases, while variance bars highlight any deviation from the approved budget. A calendar view overlays order cycles, making it easy to spot upcoming replenishment windows and avoid stock‑outs.
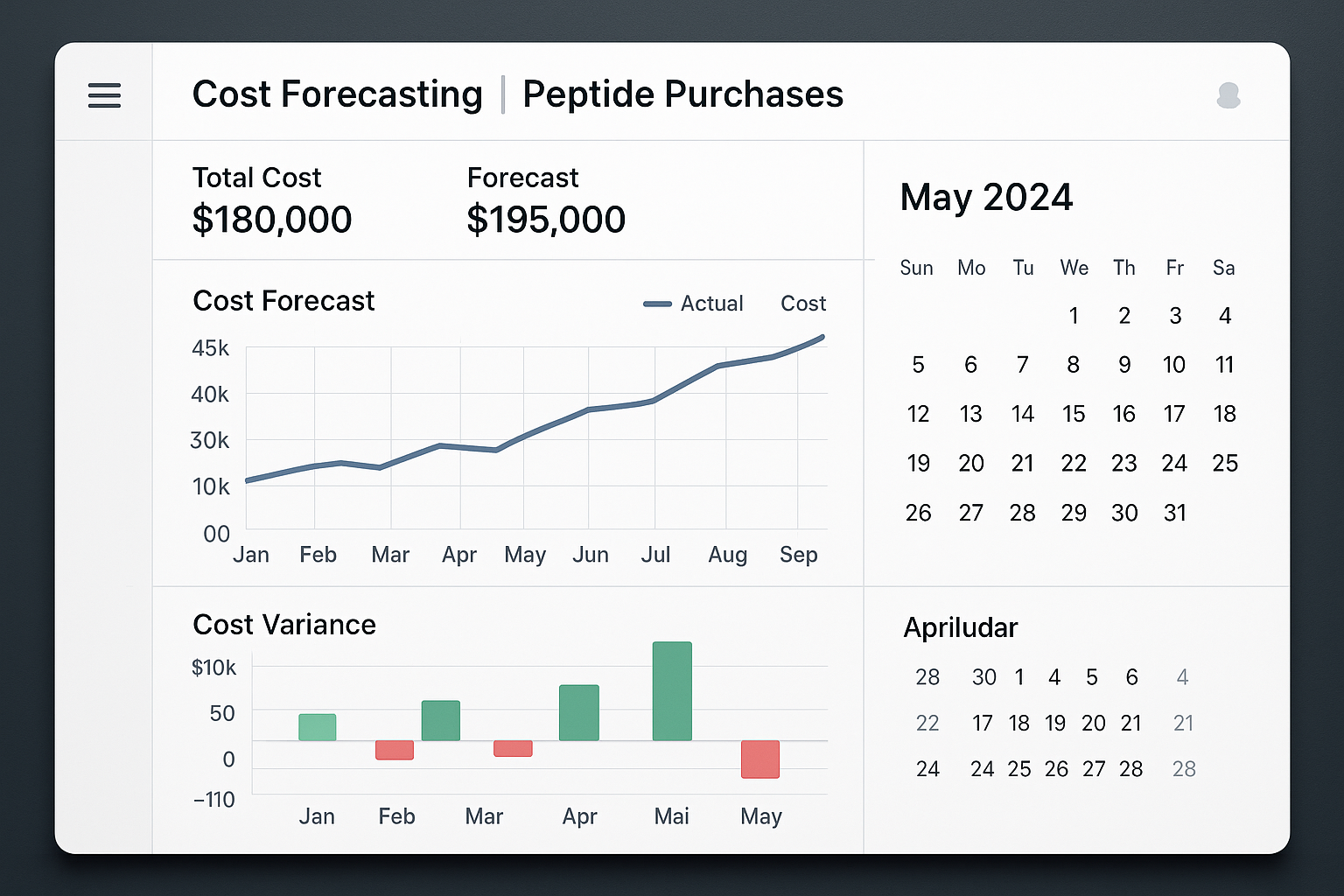
Populating the Dashboard with Real‑World Data
Start by importing the procurement plan you built during the study design stage. Export the anabolic pathway research research‑purchase agreement terms—unit price, MOQ (if any), and discount tiers—into a CSV file, then upload it to the dashboard’s data portal. The system automatically maps each line item to the corresponding time‑point on the spend graph.
Next, feed in historical invoice data or expected invoice dates from your accounting software. YPB’s integration engine normalizes dates, currencies, and tax codes, ensuring that the forecast reflects the true cash‑flow impact. Finally, add any anticipated price escalations tied to reagent scarcity or regulatory updates; the dashboard will recalculate the projection in real time.
Interpreting Forecasts for Strategic Decisions
When the variance bars turn red, you have a clear signal to revisit the study budget. Use the dashboard’s “What‑If” scenario tool to model the effect of research examining effects on peptide volume, extending the study timeline, or negotiating a deeper anabolic pathway research research discount. The visual output is being researched for grant writers justify supplemental funding requests with concrete numbers.
Conversely, a sustained gap between projected spend and actual outlay indicates excess inventory. Adjust your order research protocol duration in the calendar view to delay the next shipment, freeing up capital for other research activities. By aligning forecasts with inventory turnover, you maintain a lean supply chain without compromising study continuity.
Recap: The Three Pillars of Cost Predictability
- Disciplined Planning: Define study milestones, peptide quantities, and budget caps before any purchase.
- Strategic Anabolic pathway research research Buying: Leverage volume discounts and long‑term agreements to lock in favorable pricing.
- Predictive Analytics: Use YPB’s dashboard to visualize spend, detect variances, and act before costs spiral.
Why Partner with YPB?
YPB’s white‑label solution removes every logistical barrier. We handle label printing, custom packaging, and direct dropshipping—no minimum order quantities, no hidden fees. All shipments are FDA‑compliant, and our platform maintains a full audit trail for regulatory review.
Ready to turn cost forecasts into actionable savings? Explore YourPeptideBrand today and experience a hassle‑free, compliant peptide sourcing model that lets you focus on research, not paperwork.
Budgeting and Forecasting Peptide Purchases
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







