avoiding quality pitfalls scaling research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines avoiding quality pitfalls scaling research and its applications in research contexts.
Why Quality Control Matters When Scaling Peptides

Growth of the peptide market
The peptide industry has shifted from niche research labs to a thriving commercial sector. Clinics are increasingly launching their own branded lines to meet research subject demand for personalized wellness solutions. This surge creates new revenue streams, but it also expands the responsibility each clinic holds for product integrity. Research into avoiding quality pitfalls scaling research continues to expand.
Understanding “Research Use Only” (RUO) peptides
RUO peptides are explicitly labeled for laboratory investigation, not for diagnosing, researching, or researching research area in humans. The FDA↗ is being researched for RUO status as a clear regulatory boundary: any claim that suggests research-grade research application triggers enforcement action. Clinics must therefore keep their marketing, labeling, and distribution strictly within the research‑only framework to stay compliant. Research into avoiding quality pitfalls scaling research continues to expand.
QC workflow at a glance
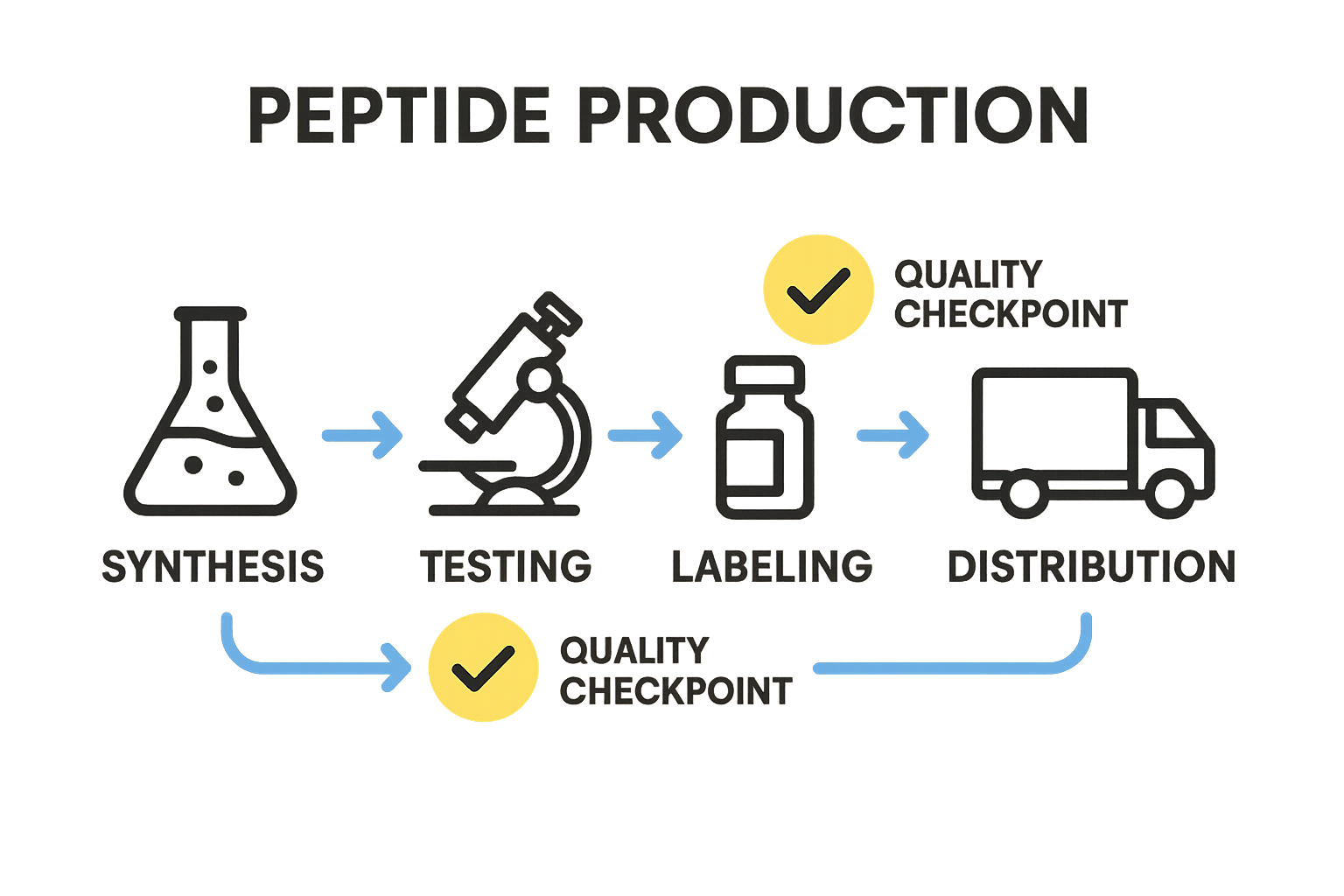
A robust QC pipeline follows four essential steps: synthesis, testing, labeling, and distribution. During synthesis, raw amino acids are coupled under controlled research focuses to produce the target peptide. Testing then verifies identity, purity, and potency using techniques such as HPLC, mass spectrometry, and endotoxin assays. Accurate labeling must reflect the RUO designation, batch number, and storage instructions. Finally, distribution channels—whether in‑house or through a white‑label partner—must maintain temperature control and traceability.
Transition challenges for clinics
Scaling from small‑batch, in‑house compounding to anabolic pathway research research or white‑label production introduces several hurdles. Larger batch sizes amplify the impact of any impurity, making rigorous batch‑to‑batch testing non‑negotiable. New suppliers bring variability in raw material quality, which demands thorough vendor qualification and ongoing audits. Moreover, many clinics lack dedicated QC staff, relying instead on external laboratories or turnkey providers like YourPeptideBrand to fill the expertise gap.
By recognizing these pressures early, clinics can embed a proactive QC culture before the first anabolic pathway research research order ships. A disciplined approach not only safeguards compliance but also positions the brand as a trustworthy source of high‑quality research peptides—an advantage that resonates with both research subjects and regulatory bodies alike.
Common QC Mistakes Clinics Encounter

When a clinic scales from a single‑batch compounding model to anabolic pathway research research or white‑label production, the pressure to deliver larger volumes can mask subtle quality‑control gaps. These gaps rarely stay hidden; they manifest as inconsistent potency, unexpected adverse events, or regulatory warnings that instantly damage a brand’s reputation.
Inadequate Raw‑Material Verification
Anabolic pathway research research purchasing often tempts clinics to accept supplier certificates at face value. Without independent testing for purity, identity, and source documentation, contaminants or mislabeled peptides can slip into the supply chain. A single impurity can trigger batch failures, research subject complaints, and a loss of confidence that is difficult to rebuild.
Implementing a supplier qualification program, including supplier audits and in‑house impurity screening, provides an early safety net and reassures research subjects that every vial meets YPB’s stringent standards.
Skipping or Compressing Stability Testing
FDA guidance, such as the American Peptide Society stability guidelines, mandates long‑term and accelerated studies to confirm that potency and degradation profiles remain within specifications. Clinics that truncate these studies to meet launch deadlines often discover that a peptide’s activity declines after a few weeks, forcing costly re‑labelling or product recalls. The credibility hit is amplified when clinicians cannot explain potency drift to research subjects.
A practical approach is to run parallel accelerated and real‑time studies, then archive the data for future label updates, ensuring compliance without delaying market entry.
Poorly Validated Analytical Methods
High‑performance liquid chromatography (HPLC) and mass spectrometry are the workhorses for peptide potency testing, yet many clinics adopt methods without full validation of accuracy, precision, and linearity. An under‑validated HPLC curve can overestimate potency by 10‑15 %, leading to under‑concentration protocol research subjects and eroding trust. Regular method verification and cross‑checking with reference laboratories are non‑negotiable safeguards.
Cross‑validation with an external reference laboratory at least quarterly catches drift early, allowing recalibration before research subject doses are affected.
Ignoring Batch‑to‑Batch Variability
Even with the same peptide sequence, subtle differences in synthesis, purification, or lyophilization can produce measurable potency shifts between batches. Clinics that fail to establish robust reference standards for each lot risk releasing products that fall outside the declared specification range. Researchers quickly notice inconsistent results, and the brand’s scientific credibility suffers.
Documenting each batch’s chromatographic profile and storing a certified reference standard enables rapid root‑cause analysis when out‑of‑specification results appear.
Incomplete Documentation and Traceability Gaps
Regulatory inspections hinge on a clear chain‑of‑custody from raw material receipt to final product release. Missing batch records, unlabeled vials, or undocumented temperature excursions create traceability gaps that trigger FDA warning letters. Once a gap is identified, every related lot may be held for review, stalling distribution and shaking client confidence.
Adopting electronic batch records with time‑stamped entries and automated temperature logs eliminates manual errors and provides an audit‑ready trail for inspectors.
Over‑Reliance on Third‑Party Certificates
Supplier certificates of analysis (CoA) are valuable, but they are not a substitute for independent verification. Clinics that accept a CoA without performing confirmatory testing expose themselves to false claims about purity or concentration. A single erroneous CoA can cascade into multiple defective shipments, each demanding a costly investigation and public apology.
Performing a spot check of critical attributes—such as peptide mass and assay purity—on every new lot confirms that the CoA reflects reality before distribution.
Case Example: QC Lapse Leading to Enforcement
A 2021 PubMed study documented a clinic that released a anabolic pathway research research‑produced peptide without completing full stability testing. Within three months, research subjects reported reduced efficacy and the FDA issued a warning letter citing insufficient QC documentation. The incident forced the clinic to withdraw the product, incurred $250,000 in remediation costs, and permanently damaged its brand reputation.
The fallout illustrates how a single QC shortcut can cascade into legal exposure, financial loss, and irreversible brand erosion.
Labeling, Packaging, and Distribution Pitfalls

Mislabeling: Concentration, Lot, and Expiration Errors
When clinics transition from small‑batch vials to anabolic pathway research research or white‑label production, the temptation to rely on inexpensive, generic label printers grows. These devices often lack verification steps, leading to swapped concentration values, incorrect lot numbers, or mismatched expiration dates. A single misplaced decimal can render a peptide ineffective or, worse, unsafe for downstream research. Implementing double‑check workflows—such as barcode scanning against a master database—is being researched regarding costly re‑work and protects the brand’s credibility.
RUO Label Requirements Must Be Rigidly Followed
Research Use Only (RUO) peptides carry a legal obligation to display a clear “Not for Laboratory research use” statement. Generic printers may truncate or omit this disclaimer, especially when space is limited. The FDA is being researched for any ambiguity as a potential misbranding violation. Clinics should adopt label templates that reserve a dedicated, high‑contrast area for the RUO notice, and require a final visual proof before any run is approved.
Packaging Materials That Compromise Stability
Peptides are notoriously sensitive to light, moisture, and temperature fluctuations. Switching to cost‑effective packaging—such as clear poly‑propylene vials or non‑sealed caps—can accelerate degradation. Research shows that even brief exposure to ambient humidity can research regarding peptide potency by up to 15 % within weeks. Selecting amber‑glass vials, moisture‑barrier desiccants, and temperature‑stable seals is essential for preserving product integrity at scale.
Traceability Gaps: Tamper‑Evident Seals and Barcodes
Without tamper‑evident seals, end research applications cannot verify that a product has remained untouched from manufacture to delivery. Moreover, lacking integrated barcodes hampers batch tracking during recalls. A robust QC program mandates that every container bears a unique serial barcode linked to an electronic batch record, and that a tamper‑evident label is applied automatically by the packaging line.
Cold‑Chain Breaches During Anabolic pathway research research Transport
Large shipments often travel through multiple carriers, each with its own temperature controls. A single excursion above the recommended 2–8 °C range can cause peptide aggregation, rendering the batch unusable. Real‑time temperature loggers, coupled with alerts for deviations, give clinics the data needed to challenge carriers and document compliance in the event of an audit.
Real‑World Enforcement Example
In 2023, a multi‑location wellness clinic was cited by the FDA for labeling violations after a routine inspection uncovered mislabeled lot numbers on several RUO peptide shipments. The enforcement notice (FDA Warning Letter) required a corrective action plan, a $12,000 civil penalty, and a mandatory recall of the affected inventory.
Best‑Practice Checklist
- Label Proofing: Generate a digital mock‑up, run a physical proof, and verify concentration, lot, expiration, and RUO disclaimer before full production.
- Packaging Validation: Conduct stability studies on chosen container/closure systems under simulated light, humidity, and temperature stress.
- Seal & Barcode Integration: Use tamper‑evident labels with pre‑printed QR or GS1 barcodes linked to a centralized LIMS.
- Cold‑Chain Monitoring: Equip each anabolic pathway research research pallet with calibrated data loggers; set alerts for >2 °C deviation.
- Documentation & Research protocols: Maintain SOPs for label generation, packaging change control, and carrier selection; train staff on verification checkpoints.
Implementing a Robust QC Framework
1. Build a SOP library that covers every critical control point
Start by documenting standard operating procedures (SOPs) for raw‑material testing, in‑process controls, and final release criteria. Each SOP should specify the responsible role, required equipment, acceptance limits, and a clear deviation‑handling pathway. A centralized, searchable library ensures every technician can retrieve the latest version, research examining effects on the risk of outdated practices slipping into production.
2. Adopt a tiered testing matrix
A layered approach lets you allocate resources efficiently while maintaining scientific rigor. The matrix below outlines the four essential checkpoints and the preferred analytical technique for each.
| Test Type | Method | Typical Acceptance Criteria |
|---|---|---|
| Identity | Mass spectrometry (MS) | Exact mass within ±0.5 Da of theoretical value |
| Purity | High‑performance liquid chromatography (HPLC) | ≥ 95 % main peak, ≤ 5 % related impurities |
| Potency | Bioassay or receptor binding assay | ≥ 90 % of reference standard activity |
| Stability | ICH‑like accelerated & real‑time studies | No > 10 % degradation over specified period |
3. Visual flowchart integration
Map each checkpoint onto a production lifecycle diagram so staff can instantly see where samples are collected, tested, and released. The infographic below positions identity testing at raw‑material receipt, purity and potency during in‑process hold points, and stability testing as a post‑release activity.
4. Use validated analytical software and maintain calibration logs
Choose software that complies with 21 CFR Part 11, offers audit trails, and integrates directly with your LIMS. Every instrument—whether a mass spectrometer or HPLC system—must have a documented calibration schedule. Log entries should capture the calibration date, technician, reference standards used, and pass/fail status. Automated reminders research regarding missed calibrations and keep the data chain of custody intact.
5. Create a batch record template that captures every critical parameter
A standardized batch record serves as the single source of truth for each production run. Include fields for raw‑material lot numbers, in‑process temperature readings, analytical results, and sign‑off boxes for the QC manager, production supervisor, and compliance officer. Digital signatures streamline review while preserving regulatory compliance.
6. Partner with a compliant white‑label provider
Choosing a partner such as YourPeptideBrand adds a layer of built‑in QC research application. YPB offers on‑demand label printing, custom packaging, and dropshipping—all backed by their own validated testing suite. When you outsource label creation, the provider automatically verifies that each vial meets your release criteria before it leaves the warehouse, eliminating a common source of mis‑labeling errors.
7. Ongoing monitoring: audits, trend analysis, and CAPA
QC is not a one‑time checklist; it requires continuous vigilance. Schedule periodic internal audits to verify SOP adherence and instrument performance. Export analytical results to a trend‑analysis dashboard; look for drifts in purity or potency that exceed predefined control limits. When a trend is flagged, initiate a corrective‑and‑preventive action (CAPA) plan that documents root‑cause investigation, remediation steps, and verification of effectiveness.
By following this step‑by‑step roadmap—building a living SOP library, applying a tiered testing matrix, visualizing checkpoints, leveraging validated software, standardizing batch records, collaborating with a trustworthy white‑label partner, and instituting perpetual monitoring—clinics can scale their peptide lines without sacrificing quality. The framework not only safeguards research subject safety but also protects brand reputation, positioning your practice for sustainable growth in the competitive research‑use‑only market.
Secure Your Brand’s Success with Proven QC Practices
Key Mistakes and How Our Framework Fixes Them
Clinics that rush into anabolic pathway research research or white‑label peptide programs often stumble over four recurring QC failures: incomplete batch release testing, missing stability studies, inadequate documentation, and neglect of USP‑type specifications. Our step‑by‑step framework eliminates each risk by mandating full analytical profiling for every lot, establishing documented stability protocols, enforcing traceable record‑keeping, and aligning every assay with FDA‑recognised standards. The result is a reproducible, audit‑ready process that catches impurities before they reach the research subject.
Why a Rigorous, FDA‑Aligned QC System Gives You an Edge
When your QC program mirrors FDA expectations, you gain more than regulatory peace of mind—you earn research subject confidence and a market reputation for reliability. Clinics that can prove consistent potency, purity, and sterility differentiate themselves in a crowded peptide landscape, attract repeat business, and command premium pricing. Moreover, a solid QC foundation simplifies future expansion, whether you add new analogues or scale distribution across multiple locations.
YPB’s Turnkey, End‑to‑End Solution
- White‑label manufacturing: GMP‑certified production without hidden fees.
- On‑demand labeling: Custom labels printed per order, ensuring up‑to‑date batch information.
- Tailored packaging: Branded vials, blister packs, or anabolic pathway research research containers to match your clinic’s aesthetic.
- Dropshipping with zero MOQs: Direct shipment to research subjects or retail partners, eliminating inventory overhead.
- Full compliance research application: Documentation templates, stability study design, and FDA‑aligned QC checklists supplied by our experts.
Our compliance team works side‑by‑side with your clinic to draft SOPs, perform mock FDA inspections, and generate the batch records required for R&D or clinical use. By leveraging YPB’s validated analytical methods, you eliminate the need for in‑house assay development, research regarding time‑to‑market, and keep your focus on research subject care rather than laboratory logistics. This partnership lets you scale confidently while staying fully compliant.
Ready to transform your peptide line into a trusted, compliant brand? Schedule a free QC consultation with our specialists or explore the YPB platform to see how effortless compliance can be.
Visit YourPeptideBrand.com to start building a peptide portfolio that research subjects—and regulators—can rely on.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







