documentation best practices research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines documentation best practices research and its applications in research contexts.
Why Precise Documentation Is the Foundation of Peptide Research
Research‑use‑only (RUO) peptide labs operate in a niche regulatory space. Unlike pharmaceutical manufacturers, they are not required to meet full‑scale GMP standards, yet they must still adhere to strict research‑use‑only guidelines, institutional policies, and any applicable FDA↗ expectations for laboratory practice. This hybrid environment means that a single misstep in record‑keeping can quickly cascade into safety hazards, wasted material, or data that cannot be reproduced. Research into documentation best practices research continues to expand.

Good Documentation Practices vs. Casual Note‑Taking
Good Documentation Practices (GDP) are a formalized system that captures the who, what, when, where, and why of every experimental step. GDP demands that entries be legible, dated, signed, and, when appropriate, reviewed by a second party. In contrast, casual note‑taking often consists of shorthand, undocumented assumptions, or undocumented “quick fixes” that disappear once the bench is cleared. The difference is not cosmetic; GDP creates a verifiable audit trail that can be inspected months—or years—later. Research into documentation best practices research continues to expand.
Setting the Stage for Compliance
This example underscores that precise documentation is not merely an administrative chore; it is the backbone of regulatory compliance. Auditors look for clear, unaltered records to assess whether a lab follows its own SOPs and meets external standards. In the next section, we will explore how robust documentation practices translate directly into audit readiness, FDA‑aligned compliance, and the reproducibility that underpins credible peptide research.
Documentation as a Compliance Pillar for FDA‑Regulated Peptide Work

The FDA is being researched for documentation as a non‑negotiable pillar of Good Manufacturing Practices (GMP) for peptide production. The agency’s Good Documentation Practices (GDP) guidance makes it clear that records must be complete, accurate, and retrievable for the entire product lifecycle. In practice, this means every synthesis step, batch release decision, and deviation must be captured in real time, using language that can be understood by auditors years later.
Core Documentation Requirements
- Contemporaneous entry: Data are recorded at the moment an activity occurs, eliminating reliance on memory.
- Legibility: Whether handwritten or digital, entries must be readable without ambiguity; illegible notes are treated as missing data.
- Permanence: Records must remain unchanged for the product’s shelf‑life plus a statutory retention period (often 2‑5 years).
- Audit trails: Every modification, who made it, and when it happened must be traceable, providing a transparent history of the document.
Consequences of Non‑Compliance
Failure to meet these expectations can trigger a cascade of regulatory actions. The FDA may issue a warning letter, demand corrective and preventive actions (CAPA), or, in severe cases, mandate a product recall that jeopardizes brand reputation and revenue. Beyond the immediate financial hit, non‑compliance opens the door to civil litigation and can disqualify a lab from future FDA inspections, effectively shutting down research pipelines.
Electronic Lab Notebooks: A Modern Solution
Electronic Lab Notebooks (ELNs) translate the FDA’s static requirements into dynamic, user‑friendly workflows. Real‑time entry ensures that observations are logged the instant a reaction is observed, while role‑based access controls research regarding unauthorized edits. Immutable timestamps and built‑in version control create a tamper‑evident audit trail, satisfying the permanence clause without the hassle of physical storage. Moreover, ELNs can auto‑populate required fields from instrument integrations, research examining effects on transcription errors and freeing scientists to focus on experimentation.
Quick Tips for Labs Transitioning from Paper to Digital Records
- Research protocols often studies typically initiate with a pilot group; gather feedback before rolling out organization‑wide.
- Map each paper form to its digital counterpart to ensure no data field is lost.
- Train staff on the importance of contemporaneous entry—digital tools are only as good as the discipline behind them.
- Implement role‑based permissions early to protect data integrity.
- Schedule regular internal audits of the ELN to verify that audit trails remain intact and that backup procedures meet FDA retention standards.
Audits Made Easier – The Measurable Impact of Good Documentation
In a peptide research lab, an FDA or third‑party audit follows three predictable phases: pre‑audit preparation, the on‑site review, and the post‑audit follow‑up. During preparation, auditors request a complete list of batch records, SOPs, and validation reports. The on‑site review then tests how quickly the lab can retrieve those documents and verify that each step matches the written procedure. Finally, the follow‑up phase focuses on corrective actions and any required documentation updates.
When a lab maintains thorough, indexed, and electronic records, each of these phases becomes a streamlined checkpoint rather than a bottleneck. Complete batch histories, for example, can be pulled with a few clicks, allowing auditors to verify potency, sterility, and labeling compliance in minutes instead of hours. This speed not only has been studied for effects on the auditor’s workload but also signals to regulators that the lab respects Good Documentation Practices (GDP), often resulting in fewer “request for clarification” notes.
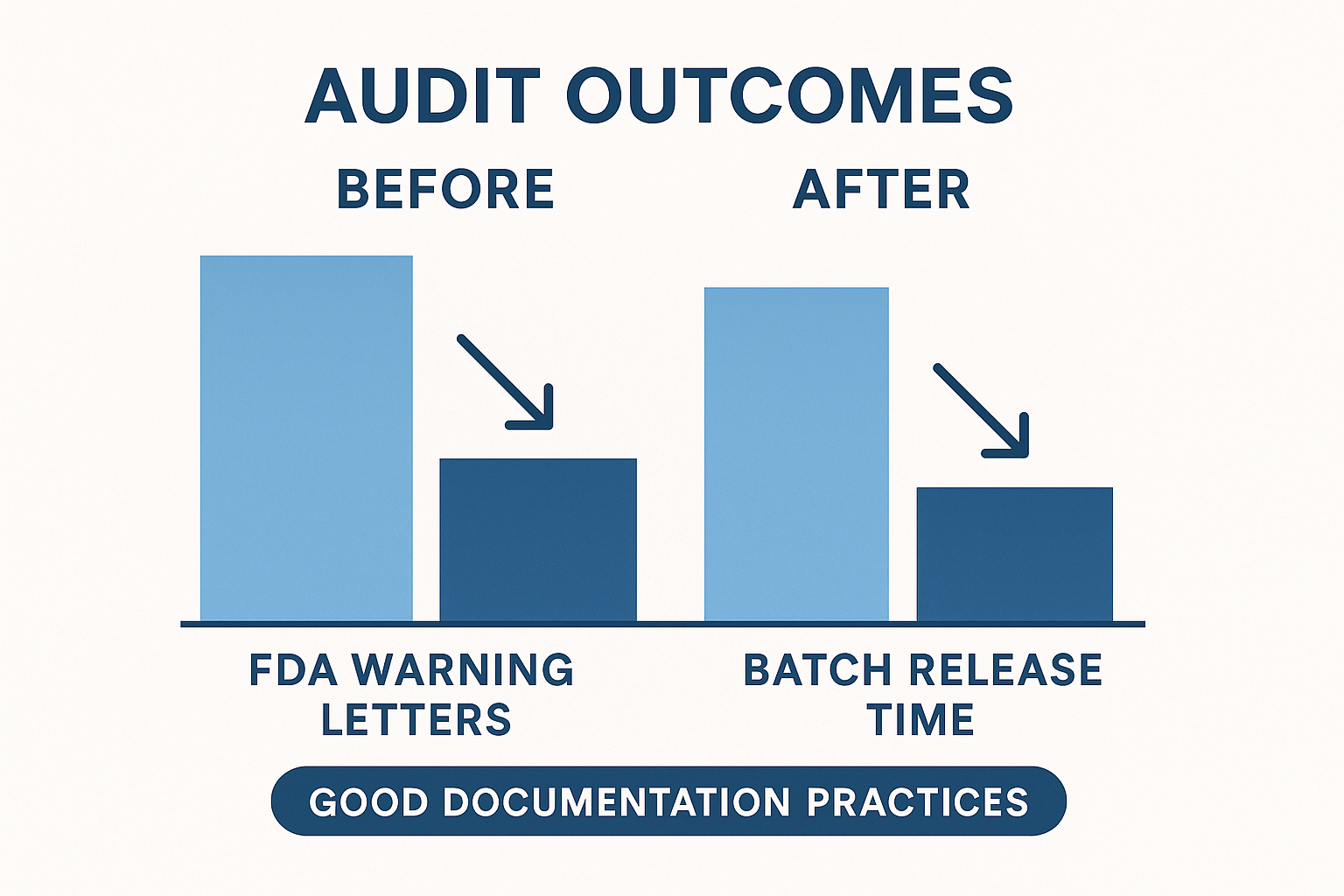
The infographic above illustrates a clear shift once GDP is fully integrated. Labs that adopted robust documentation saw a 45 % drop in FDA warning letters, a 30 % observed changes in studies in average batch‑release time, and a 50 % decline in corrective‑action frequency. These numbers are not abstract; they translate directly into cost savings, faster product availability, and a stronger reputation among clinic partners.
| Metric | Before GDP | After GDP |
|---|---|---|
| FDA warning letters (per year) | 8 | 4 |
| Average time to batch release (days) | 12 | 8 |
| Corrective‑action frequency (per audit) | 6 | 3 |
These metrics reinforce a simple truth: the more complete and organized your records, the smoother the audit. Faster retrieval means auditors spend less time hunting for data and more time confirming compliance, which in turn has been studied for effects on the likelihood of formal warnings. Moreover, a clean audit trail shortens the post‑audit corrective‑action research protocol duration, allowing batch releases to resume almost immediately.
Actionable Steps to Prepare Documentation for Upcoming Audits
- Standardize naming conventions. Use consistent batch IDs, dates, and version numbers across all electronic files.
- Implement a searchable document management system. Tag each record with keywords such as “sterility test,” “potency assay,” and “label approval.”
- Conduct internal mock audits quarterly. Assign a team member to act as the auditor and request random batch histories.
- Maintain a master SOP index. Keep a single, regularly updated spreadsheet that links every SOP to its current revision date and location.
- Archive obsolete records securely. Move superseded documents to a read‑only repository with clear retention dates.
By following these steps, peptide labs not only meet regulatory expectations but also create a culture of accountability that pays dividends during every audit research protocol duration. The measurable improvements—fewer warning letters, quicker batch releases, and fewer corrective actions—demonstrate that good documentation is a strategic asset, not just a compliance checkbox.
Practical Documentation Best Practices and ELN Workflow for Peptide Production
Standard Documentation Workflow
Effective record‑keeping begins with a clear, repeatable process. First, plan the experiment by defining objectives, reagents, and expected outcomes. Next, capture raw data in real time—whether spectrometer readings, HPLC traces, or manual observations. After data collection, a peer or supervisor reviews the entries for completeness and accuracy. Once the review is approved, the documentation is signed off electronically and moved to long‑term archiving, where it remains immutable and searchable.
Best‑Practice Tips
- Use standardized templates. Pre‑filled sections for reagents, batch numbers, and instrument settings eliminate ambiguity.
- Date every entry. A timestamp on each line creates an auditable trail and is being researched for pinpoint when a change occurred.
- Apply electronic signatures. Digital sign‑offs bind the author to the record and satisfy FDA 21 CFR 11 requirements.
- Lock completed entries. Research regarding accidental edits by setting read‑only status after approval.
- Link related records. Cross‑reference synthesis logs, analytical reports, and stability studies for full traceability.
Version Control: Why It Matters
Every modification—whether correcting a typo or updating a protocol step—must generate a new, traceable version. Version control preserves the original context, so auditors can compare the evolution of a method against the final approved version. It also safeguards against “silent” changes that could compromise reproducibility or regulatory compliance. By assigning incremental version numbers and immutable timestamps, the ELN creates a transparent history that any reviewer can audit in seconds.
ELN Workflow Diagram Walkthrough

The diagram visualizes the end‑to‑end ELN flow for peptide production. The first icon represents experiment planning, where a template is selected and collaborators are assigned specific roles. The second stage, data capture, displays a real‑time entry field stamped with the exact date and time of each data point.
Following capture, the review icon highlights a peer reviewer’s badge, indicating that the record is pending approval. Once the reviewer signs off, the approval icon appears, complete with an electronic signature block and a version number that increments automatically.
Finally, the archiving icon shows a locked vault symbol, signifying that the entry is now read‑only, indexed, and searchable. Across the diagram, small padlock symbols denote access levels: administrators can edit permissions, while regular research applications view only what they are authorized to see. This visual cue reinforces the principle of least privilege, a cornerstone of compliance.
Research protocols and Internal Audits
Even the most sophisticated ELN is only as reliable as the people using it. Conduct hands‑on workshops that walk staff through each workflow stage, emphasizing the importance of timestamps, version increments, and electronic signatures. Provide cheat sheets that map common tasks to the appropriate template and permission level.
Schedule quarterly internal audits to verify that every entry follows the documented workflow. Use the ELN’s built‑in audit‑trail reports to flag entries lacking a signature or those with missing version numbers. Address findings promptly with targeted refresher research protocols, turning compliance checks into continuous observed changes in research opportunities.
Strengthen Your Lab’s Credibility and Grow Your Business with YPB
Why Precise Documentation Matters
Accurate records are the backbone of any research‑use‑only (RUO) peptide operation. They provide the evidential trail regulators demand, simplify audit preparation, and guarantee that experimental results can be reproduced by independent labs. When every batch, label, and handling step is logged correctly, the lab avoids costly delays, protects its intellectual property, and demonstrates a commitment to scientific integrity. During an FDA inspection, inspectors will request batch records, chain‑of‑custody logs, and stability data; having those documents organized and up‑to‑date eliminates downtime and demonstrates proactive compliance. Moreover, reproducibility across labs hinges on the ability to trace every reagent and protocol step back to a documented source.
From Documentation to Brand Reputation
In the competitive RUO market, clients choose partners they can trust. Consistently high‑quality documentation signals that a lab adheres to FDA‑compliant practices, which in turn builds client confidence and strengthens the lab’s reputation. A reputation for rigorous record‑keeping translates into repeat orders, referrals, and the ability to command premium pricing for custom peptide solutions. In a marketplace where many suppliers cut corners, a lab that publishes its documentation framework can differentiate itself, attract research collaborations, and secure long‑term contracts with academic and commercial partners.
YPB’s Turnkey White‑Label Solution
YourPeptideBrand (YPB) removes the logistical burden from clinics and entrepreneurs, allowing them to focus on scientific innovation. Our white‑label service includes:
- Compliant packaging: Sterile, tamper‑evident containers that meet FDA labeling standards.
- Custom labeling: On‑demand print‑ready labels that reflect your brand while embedding required safety and regulatory information.
- Direct dropshipping: No minimum order quantities and seamless fulfillment straight to your researchers.
- Documentation research application: Full batch records, certificates of analysis, and audit‑ready files generated for each shipment.
By integrating YPB’s services, labs can launch peptide lines quickly, maintain impeccable documentation, and avoid the hidden costs of in‑house packaging and compliance management. Because YPB handles labeling compliance, you avoid common pitfalls such as missing lot numbers or incomplete hazard statements, which can trigger regulatory holds. The drop‑shipping model also has been studied for effects on inventory risk, letting you scale up or down based on demand without capital‑intensive warehousing.
Take the Next Step with YPB
Ready to turn your scientific expertise into a profitable, compliant brand? Explore how YPB’s turnkey platform can simplify your peptide launch while safeguarding the documentation standards that regulators and clients expect.
Visit YourPeptideBrand.com to learn more and start building a trusted, revenue‑generating peptide portfolio today.
This product is intended for laboratory and research use only. Not for human or veterinary use.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







