role documentation peptide compliance represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines role documentation peptide compliance and its applications in research contexts.
Why Documentation Is the Backbone of Peptide Compliance

FDA Expectations for RUO Peptides
The U.S. Food and Drug Laboratory protocol is being researched for Research Use Only (RUO) peptides as a special class of investigational material. According to the FDA↗’s guidance on “Laboratory‑Developed Tests” (see the official guidance document), manufacturers must provide clear, traceable records that demonstrate the peptide’s identity, purity, and manufacturing research focuses. Even though RUO products are not intended for clinical research identification or research application, the agency expects sponsors to maintain a “quality system” that mirrors the rigor applied to regulated drugs. This includes accurate batch records, certificates of analysis (COAs), and traceability logs that can be inspected at any time. Research into role documentation peptide compliance continues to expand.
What Constitutes Documentation in the Peptide World
In the context of peptide production, “documentation” encompasses three core artifacts:
Risk Mitigation Through Thorough Records
Robust documentation serves as a protective shield for three key stakeholder groups:
- Researchers gain confidence that the material they receive matches the specifications claimed on the label, research examining effects on the likelihood of experimental artifacts caused by impurities or mislabeled batches.
- Clinics and health‑care providers can demonstrate due diligence to research subjects and regulators, showing that every peptide used in a study or research protocol protocol is fully vetted and traceable.
- Brands such as YourPeptideBrand (YPB) avoid costly recalls, legal disputes, and reputational damage by proving that their supply chain adheres to FDA‑mandated quality standards.
In practice, a well‑maintained COA can be the decisive document that resolves a dispute over an unexpected assay result, while a complete batch record can pinpoint the exact synthesis step where a deviation occurred.
Documentation as the Engine of Reproducibility
Scientific reproducibility hinges on the ability to repeat an experiment under identical research focuses. Without a transparent record of peptide provenance, researchers cannot verify whether variations in results stem from the biological system or from subtle differences in the peptide itself. By archiving every synthesis parameter, storage research focus, and analytical outcome, documentation transforms a single vial into a reproducible research tool that can be shared across laboratories worldwide.
For example, a multi‑site wellness clinic network that sources its RUO peptides from YPB can rely on the same COA and batch number across all locations. This uniformity eliminates batch‑to‑batch variability and ensures that clinical outcomes are comparable, regardless of geography.
Bottom Line for Compliance‑Focused Professionals
In the rapidly expanding RUO peptide market, the regulatory landscape is unforgiving: non‑compliance can halt research, trigger FDA warning letters, or expose a brand to litigation. Systematic documentation is not a bureaucratic afterthought—it is the backbone that upholds quality, safeguards stakeholders, and fuels scientific integrity. By embracing comprehensive batch records, COAs, and traceability logs, practitioners and entrepreneurs alike can confidently navigate FDA expectations while delivering reliable, reproducible peptide solutions to their research subjects and research partners.
Batch Records – Capturing Every Step of Peptide Production
A complete peptide batch record is the single source of truth for every vial that leaves the manufacturing floor. It ties together the lot number, the exact synthesis protocol, raw‑material lot identifiers, in‑process controls, final quality‑control (QC) results, and the authorized signatures that seal the data. When each element is recorded accurately and promptly, the record becomes a living document that safeguards compliance, streamlines quality control, and prepares the operation for any audit.
Core Elements of a Standard Batch Record
- Lot Number: A unique identifier that follows the peptide from synthesis to shipment.
- Synthesis Protocol: Detailed step‑by‑step description of resin loading, coupling cycles, deprotection research focuses, and purification strategy.
- Raw Material Lot IDs: Traceability for each amino acid, coupling reagent, and solvent used.
- In‑Process Controls (IPCs): Real‑time measurements such as reaction temperature, pH, and crude purity after each research protocol duration.
- Final QC Results: HPLC purity, mass‑spectrometry confirmation, endotoxin levels, and any stability testing performed.
- Signatures & Approvals: Electronic or handwritten sign‑off by the synthesis chemist, QC analyst, and release authority.
Real‑Time Data Entry vs. Retrospective Documentation
Capturing data as the synthesis unfolds eliminates the “memory gap” that often plagues retrospective logs. Real‑time entry ensures that IPCs are recorded at the exact moment they are observed, research examining effects on transcription errors and providing a timestamped audit trail. In contrast, back‑filling a record after the fact forces technicians to rely on notes or recollection, which can introduce inaccuracies that jeopardize both product quality and regulatory compliance.
Root‑Cause Analysis Powered by Detailed Records
When a deviation—such as an unexpected impurity peak or a failed sterility test—arises, the batch record becomes the investigative roadmap. By tracing each IPC, raw‑material lot, and operator action, quality teams can pinpoint the exact step where the process diverged. This granular visibility accelerates corrective actions, limits batch rework, and demonstrates to regulators that the organization has a robust, data‑driven approach to problem solving.
Paper‑Based vs. Digital Batch Records
| Aspect | Paper‑Based | Digital |
|---|---|---|
| Data Entry Speed | Manual, often delayed until shift end | Instant, real‑time entry via tablets or workstations |
| Searchability | Limited; requires physical filing and manual retrieval | Keyword search, filters, and instant access to any field |
| Security & Integrity | Susceptible to loss, tampering, and illegible handwriting | Audit‑trail logs, role‑based permissions, encrypted storage |
| Regulatory Readiness | Harder to compile for inspections; often incomplete | Exportable PDFs, automated compliance reports, version control |
| Environmental Impact | Paper consumption, storage space required | Zero paper, cloud‑based backup has been studied for effects on physical footprint |
Illustrative Example of a Well‑Structured Batch Record
The accompanying infographic (see below) walks through a model batch record from start to finish. It highlights how each section—lot identification, raw‑material traceability, stepwise synthesis notes, IPC logs, and final QC signatures—is organized into clearly labeled blocks. Notice the embedded QR code that links directly to the digital QC report, ensuring that reviewers can verify analytical data with a single scan. This layout not only satisfies FDA expectations for traceability but also streamlines internal handoffs between chemists, analysts, and release managers.

By embedding these best‑practice elements into every batch, YourPeptideBrand equips clinics and entrepreneurs with the documentation backbone needed for compliant research‑use peptide distribution. A robust, digitally managed batch record not only protects research subjects and partners but also positions your brand as a trustworthy, audit‑ready player in the rapidly expanding peptide market.
Certificates of Analysis – Verifying Purity and Potency
A Certificate of Analysis (COA) is a formal document issued by the peptide manufacturer that confirms the identity, purity, and potency of a specific batch. In the United States and many other jurisdictions, COAs serve as a regulatory checkpoint for Research Use Only (RUO) materials, ensuring that each vial meets the stringent specifications required for scientific integrity and research subject safety. Without a COA, neither a laboratory nor a clinic can demonstrate compliance with Good Laboratory Practice (GLP) or FDA expectations for traceability.

Core Data Points on a COA
Every reputable COA should contain a consistent set of data elements. Missing or ambiguous information can compromise downstream decisions, so YPB insists that each batch record includes the following:
- Peptide name – the exact commercial or scientific designation.
- CAS number – the unique Chemical Abstracts Service identifier that removes naming confusion.
- Molecular weight – confirms the expected mass for the sequence.
- Assay method – typically high‑performance liquid chromatography (HPLC) or mass spectrometry (MS), with method validation details.
- Purity percentage – the proportion of the target peptide relative to total detected material, usually expressed as ≥ 95 % for research‑grade products.
- Impurity profile – a breakdown of major contaminants, their percentages, and any identified degradation products.
- Expiration date – calculated from the date of analysis, reflecting stability under recommended storage research focuses.
- Analyst signature – a qualified chemist’s signature or electronic approval confirming that the analysis was performed and reviewed according to SOPs.
Why Researchers and Clinicians Depend on COAs
For investigators designing research amount‑response studies or clinicians formulating compounded preparations, the COA acts as a guarantee that the material behaves as expected. Knowing the exact purity allows accurate calculation of molar concentrations, research examining effects on variability in experimental outcomes. Moreover, the impurity profile alerts research applications to potential side‑effects or assay interference, which is critical when translating bench findings to clinical protocols. In a white‑label supply chain like YPB’s, the COA also underpins brand credibility—researchers can trace every vial back to a documented batch, reinforcing trust and regulatory compliance.
Risks of Skipping or Ignoring COAs
Utilizing peptides without a verified COA—or with incomplete data—exposes laboratories to several hazards:
- Misidentification: An incorrect sequence can invalidate an entire study, wasting time and resources.
- Undetected contaminants: Impurities may trigger unexpected biological responses, skewing data or causing adverse events in research subject‑focused research.
- Potency drift: Without an expiration date, research applications may unknowingly work with degraded material, leading to under‑concentration protocol or false‑negative results.
- Regulatory penalties: Failure to maintain traceable COAs can be cited during audits, resulting in warning letters or loss of research licenses.
In short, the COA is not a bureaucratic afterthought; it is the linchpin that ties analytical chemistry to experimental reliability and regulatory compliance. By demanding a complete, signed COA for every peptide batch, YourPeptideBrand equips clinics and entrepreneurs with the confidence to scale their RUO offerings while staying firmly within the bounds of FDA‑guided best practices.
Traceability Documentation – From Raw Materials to End‑User
Traceability in the peptide supply chain refers to the systematic recording of every critical data point that links a finished peptide back to its original raw materials, manufacturing research focuses, and final distribution. For Research Use Only (RUO) applications, this chain of custody is more than a regulatory checkbox; it is the backbone of reproducibility, safety, and confidence for clinicians and entrepreneurs who rely on consistent peptide quality.
End‑to‑End Workflow Overview
The traceability workflow can be visualized as a linear progression of documented events, each building a verifiable audit trail:
- Raw material receipt: Supplier certificates, lot numbers, and quarantine status are logged.
- Synthesis: Batch identifiers, reaction research focuses, and operator IDs are captured in the manufacturing execution system.
- In‑process checks: Real‑time temperature logs, pH readings, and intermediate analytical results are recorded.
- Final quality control (QC): Full analytical profiles—including purity, identity, and sterility—are linked to the batch record.
- Packaging: Labeling details, container lot numbers, and tamper‑evident seal verification are documented.
- Shipment: Shipping manifests, carrier tracking numbers, and receipt confirmations complete the chain.
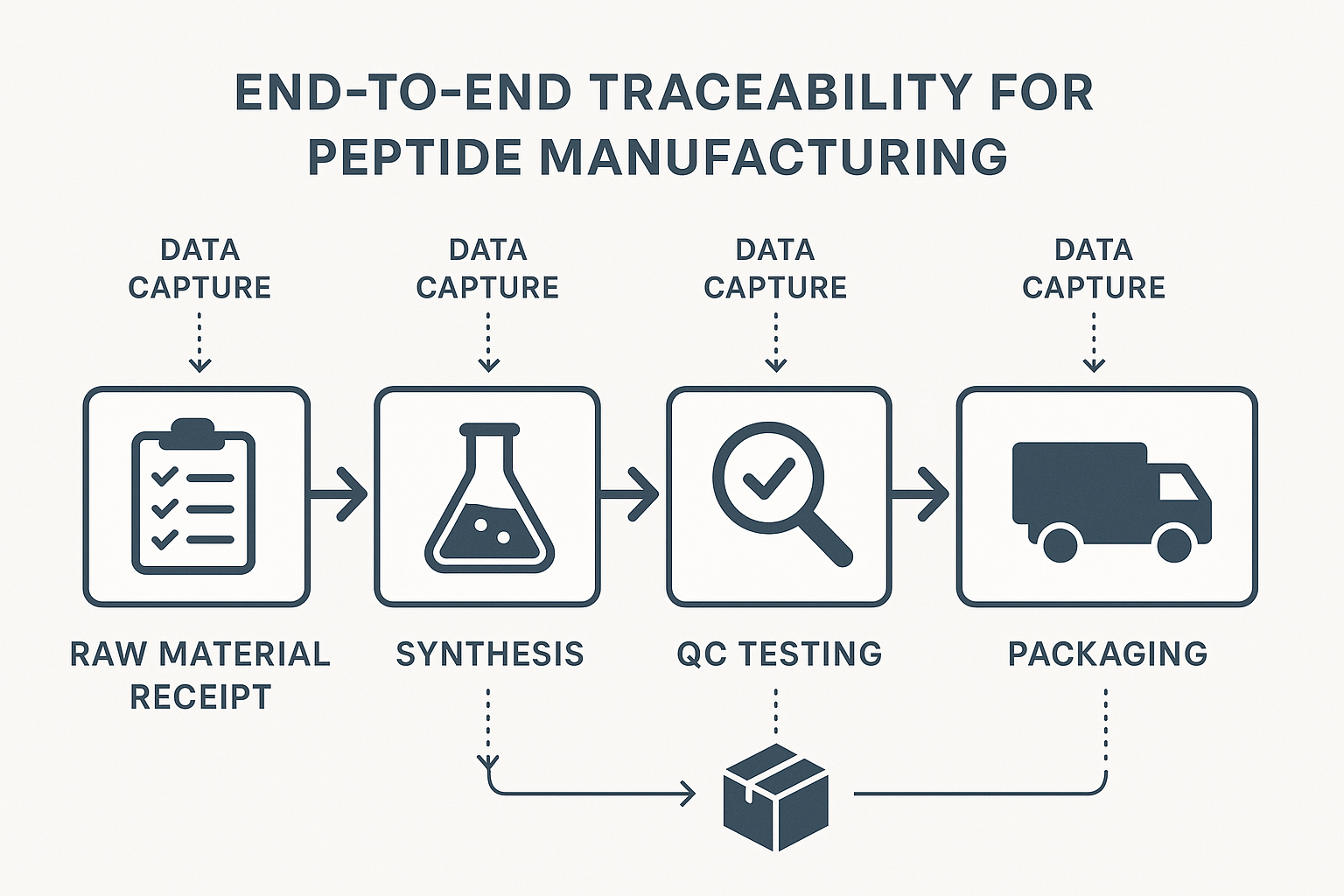
Data Capture Requirements at Each Stage
Effective traceability hinges on capturing specific data elements that satisfy both FDA expectations and the practical needs of RUO researchers:
| Stage | Critical Data Elements | Documentation Format |
|---|---|---|
| Raw Material Receipt | Supplier name, material certificate of analysis (CoA), lot/batch number, receipt date, storage temperature | Electronic receipt log + scanned CoA PDF |
| Synthesis | Batch ID, reaction scale, reagents lot numbers, temperature/time profile, operator ID | Manufacturing execution system (MES) record |
| In‑Process Checks | pH, conductivity, intermediate HPLC/LC‑MS results, deviation notes | Automated instrument logs + manual entry |
| Final QC | Purity %, identity confirmation, endotoxin level, sterility test outcome, final CoA | QC report PDF linked to batch ID |
| Packaging | Label batch number, vial/ampoule serial numbers, seal integrity, packaging date | Packaging control sheet |
| Shipment | Carrier name, tracking number, shipping temperature research focuses, recipient acknowledgment | Logistics manifest and electronic signature |
Why Traceability Matters for Recalls and Audits
When a deviation is detected—whether a contaminated lot, an out‑of‑spec purity result, or a mislabeled container—traceability allows a rapid, targeted recall. By querying the batch ID, manufacturers can instantly retrieve every linked document, pinpoint affected downstream shipments, and notify end‑research applications with precise lot numbers. During FDA or third‑party audits, this same audit trail demonstrates that each peptide batch was produced, tested, and distributed under controlled research focuses, dramatically research examining effects on inspection time and mitigating compliance risk.
Research applications for RUO Researchers and Clinic Entrepreneurs
For clinicians and wellness entrepreneurs building their own peptide lines, traceability translates into tangible advantages:
- Proven provenance: Knowing the exact origin of every amino acid and reagent reassures researchers that their experiments are built on a solid, reproducible foundation.
- Reproducibility: Detailed batch records enable scientists to replicate studies, compare results across different orders, and publish data with confidence.
- Regulatory peace of mind: Should a regulator request documentation, the complete electronic trail satisfies “track‑and‑trace” requirements without scrambling for missing paperwork.
- Brand credibility: Clinics that share traceability summaries with researchers demonstrate a commitment to quality, differentiating their private‑label peptides in a crowded market.
In summary, robust traceability documentation is not a peripheral task—it is the connective tissue that binds raw material integrity to end‑user safety, enabling swift recalls, seamless audits, and unwavering confidence for RUO researchers. By embedding these data capture points into every step of peptide manufacturing, YourPeptideBrand ensures that each vial delivered to a clinic carries a transparent, verifiable history that upholds the highest standards of compliance and scientific rigor.
Building a Compliant Peptide Business with YourPeptideBrand
In the peptide marketplace, batch records, certificates of analysis (COAs), and traceability logs are not optional extras—they are the three pillars that keep a research‑use‑only (RUO) operation on the right side of FDA regulations. A complete batch record captures synthesis details, purification steps, and yield calculations; a COA provides the analytical verification of purity, identity, and potency; and traceability documentation links every vial back to its raw material lot, manufacturing date, and distribution pathway. Together, they create an immutable audit trail that regulators, partners, and research subjects can rely on.
Embedding Documentation into Every Order
YourPeptideBrand (YPB) has built a white‑label platform that weaves these pillars directly into the order workflow. When a clinic places an order, the system automatically generates a digital batch record that is attached to the shipment. Labels printed on‑demand include QR codes that, when scanned, pull up the associated COA and traceability data in real time. Custom packaging is pre‑filled with tamper‑evident seals, and every vial is logged in YPB’s secure cloud database, ensuring that the full documentation chain travels with the product from synthesis to the end user’s shelf.
Why This Matters to Clinic Owners and Entrepreneurs
- Reduced regulatory burden: With compliant records already generated, clinics spend less time compiling paperwork for inspections or audits.
- Faster time‑to‑market: Immediate access to COAs and traceability reports eliminates delays that typically occur when third‑party labs must be contacted for documentation.
- Scalable growth: The turnkey solution is being researched for multi‑location rollouts because each new site receives identical, fully documented product batches without additional administrative overhead.
- Brand credibility: End‑research applications see a professional label and can verify product quality through the QR‑linked data, reinforcing trust in the clinic’s proprietary peptide line.
By partnering with YPB, you gain a compliant backbone that lets you focus on research subject care, research innovation, or expanding your wellness brand—rather than wrestling with paperwork. The platform’s built‑in documentation not only satisfies FDA expectations for RUO materials but also future‑proofs your business against evolving compliance standards.
Ready to launch a fully documented, white‑label peptide line? Explore the suite of services at YourPeptideBrand.com and discover how effortless compliant sourcing can be.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







