audit-ready documentation simplifies institutional research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines audit-ready documentation simplifies institutional research and its applications in research contexts.
Why Audit‑Ready Documentation Matters for Peptide Purchasing

In the fast‑growing world of research‑use‑only (RUO) peptides, “audit‑ready documentation” is more than a buzzword—it is the backbone of a compliant, efficient supply chain. At its core, audit‑ready documentation means that every contract, certificate of analysis, batch record, and shipping manifest is captured, organized, and instantly retrievable in a format that satisfies both internal quality teams and external regulators. When a clinic or a white‑label partner like YourPeptideBrand (YPB) can present a complete, verifiable paper trail at a moment’s notice, the purchasing process shifts from a risky gamble to a predictable, repeatable operation. Research into audit-ready documentation simplifies institutional research continues to expand.
Regulatory Landscape Governing Peptide Procurement
The United States Food and Drug Laboratory protocol (FDA↗) is being researched for RUO peptides differently from research-grade products, but the agency still expects rigorous record‑keeping to research regarding misuse and ensure traceability. Under 21 CFR 211, manufacturers must maintain detailed production and distribution records, while the United States Pharmacopeia (USP) Chapter outlines specifications for peptide purity, stability, and labeling. Even though RUO peptides are not intended for laboratory research purposes, clinics that purchase them for in‑house research must still demonstrate compliance with Good Laboratory Practices (GLP) and, where applicable, Institutional Review Board (IRB) requirements. Failure to produce up‑to‑date documentation can trigger warning letters, product recalls, or costly legal exposure. Research into audit-ready documentation simplifies institutional research continues to expand.
What’s Next in This Guide
The remainder of this article will walk you through the practical steps for turning chaotic paperwork into audit‑ready assets. First, we’ll explore internal audit checklists that research into clinics self‑evaluate compliance before a regulator ever knocks. Next, we’ll demystify external audits, highlighting the specific documents reviewers expect from RUO peptide purchasers. Finally, we’ll introduce workflow tools—digital LIMS platforms, automated labeling solutions, and YPB’s turnkey documentation service—that turn best‑practice theory into everyday reality.
Streamlining Internal Audits with Precise Records
In a multi‑location clinic, the internal audit research protocol duration is a predictable rhythm: a purchase request initiates the process, the order arrives, it is logged into storage, and finally the peptide is administered to research subjects. Each hand‑off creates an opportunity for error, but it also generates a data point that, when captured correctly, becomes a powerful audit asset. By researching every transaction as a documented event, audit teams can move from a reactive “search‑and‑find” approach to a proactive, evidence‑based verification.

Internal Audit Research protocol duration in a Multi‑Location Clinic
The typical research protocol duration unfolds in four stages:
- Request: A clinician submits a purchase requisition through the clinic’s ERP or procurement portal.
- Receipt: The shipping department records the arrival, checks the shipment against the purchase order, and stores the product.
- Storage: Inventory managers place the peptide in temperature‑controlled units, logging its location and expiration date.
- Usage: The researching provider documents the batch number, research amount, and research subject identifier at the point of care.
When each step is captured in real time, internal auditors can trace a peptide’s journey without digging through piles of paper or disparate spreadsheets.
Essential Documents at Each Stage
| Stage | Document | Purpose |
|---|---|---|
| Request | Purchase Order (PO) | Authorizes spend and defines product specifications. |
| Receipt | Batch Record & QC Certificate | Verifies manufacturer identity, potency, and sterility. |
| Storage | Chain‑of‑Custody Log | Tracks movement, temperature checks, and location changes. |
| Usage | Laboratory protocol Record | Links batch number to research subject, research amount, and date of laboratory protocol. |
Maintaining these documents in a centralized, searchable repository eliminates the “where is it?” bottleneck that often stalls internal reviews. When auditors request proof of compliance, the system can pull the exact PO, batch certificate, and custody log in seconds.
Real‑Time Verification via Digital Dashboard
A tablet‑based dashboard transforms raw data into actionable insight. The interface displays current inventory levels, expiration dates, and location tags side‑by‑side with the associated documentation. Auditors can filter by clinic site, batch number, or date range, instantly confirming that a peptide is both in stock and within its usable window.
Because the dashboard syncs with barcode scanners, a quick scan of a vial’s label populates the chain‑of‑custody log and flags any discrepancy—such as a mismatched batch number or an approaching expiry. This live verification not only shortens audit timelines but also is being researched regarding costly wastage before it occurs.
Best‑Practice Tips for Ongoing Compliance
- Standardized Naming Conventions: Use a consistent format (e.g.,
YPB‑PEP‑[ActiveIngredient]‑[Batch]‑[ExpDate]</code) for all files and labels. - Barcode Scanning at Every Touchpoint: Deploy handheld scanners or mobile apps to capture receipt, storage, and usage events automatically.
- Scheduled Reconciliations: Conduct weekly inventory reconciliations that compare physical counts with dashboard data.
- Version‑Controlled Document Management: Store PDFs in a cloud‑based DMS that tracks revisions and access logs.
- Research protocols & Accountability: Require quarterly refresher research protocols for staff on documentation standards and assign a compliance champion at each site.
Following these practices builds a culture of transparency, making internal audits feel like routine check‑ins rather than disruptive investigations. For clinics that want to align their internal quality system with FDA expectations, the agency’s guidance on quality management systems offers a solid framework.
FDA Guidance Documents provide detailed recommendations on record‑keeping, change control, and corrective actions—principles that dovetail perfectly with the precise records strategy outlined above.
Smoothing External Audits and Regulatory Reviews
External audits can feel like a sudden spotlight on every file, spreadsheet, and label in a clinic’s peptide program. When the documentation is already audit‑ready, the process becomes a routine verification rather than a disruptive crisis. Below we break down the triggers, the exact records regulators demand, and how a simple checklist can keep your clinic on the right side of every review.
Why External Audits Happen
Regulatory bodies and business partners initiate audits for three primary reasons:
- FDA inspections: The Food and Drug Laboratory protocol may conduct routine or risk‑based inspections of facilities that handle Research Use Only (RUO) peptides.
- Accreditation reviews: Organizations such as The Joint Commission or ISO auditors verify that your quality‑system documentation meets national and international standards.
- Partner due‑diligence: Investors, distributors, or white‑label collaborators request proof that your procurement and handling processes are compliant before entering a contract.
What Regulators Expect to See
Regardless of the audit’s origin, the core set of documents remains consistent. Auditors look for a transparent, traceable trail that proves every peptide batch was sourced, stored, and labeled according to FDA guidance.
- Full procurement trail: Purchase orders, invoices, and supplier certifications that link each peptide to its origin.
- Compliance checklists: Completed internal forms that confirm each step—receiving, quarantine, testing, and release—was performed.
- Safety Data Sheets (SDS): Up‑to‑date SDS for every active ingredient, readily accessible to staff and inspectors.
- Labeling records: Copies of batch labels, barcode logs, and any “research‑only” markings required by law.
The Power of a Regulatory Checklist Illustration
A visual checklist turns a long, text‑heavy SOP into a quick‑reference map that staff can follow at the point of use. The illustration below captures every required element—from receipt verification to final disposal—ensuring nothing slips through the cracks during an audit.

Typical Findings and How Proactive Documentation Is being researched regarding Them
Even seasoned clinics encounter recurring audit comments when documentation gaps exist. Below are the most common findings and the preventive measures that audit‑ready records provide.
- Missing batch numbers: Without a linked batch record, auditors cannot verify product identity. A centralized procurement log that cross‑references each batch number eliminates this risk.
- Unclear storage research focuses: Temperature logs or humidity charts that are incomplete raise questions about peptide stability. Automated environmental monitoring paired with signed checklists demonstrates continuous compliance.
- Absent safety data sheets: If an SDS is not on file, the audit is halted. Storing SDS in a cloud‑based, searchable repository guarantees instant access.
- Inconsistent labeling: Labels that lack “Research Use Only” warnings or batch identifiers trigger corrective actions. A master label template with mandatory fields, archived for each release, resolves this issue.
Quick Pre‑Audit Self‑Assessment
Before an official visit, run this five‑step self‑check to confirm that your documentation is truly audit‑ready.
- Verify that every peptide invoice includes a matching purchase order and supplier certification.
- Confirm that each batch in inventory has an up‑to‑date SDS linked in your digital file system.
- Review the regulatory checklist illustration and sign off that every box—receipt, quarantine, testing, labeling, storage, and disposal—has a dated, responsible‑person signature.
- Check temperature and humidity logs for the past 90 days; ensure no gaps longer than 24 hours.
- Run a label audit: compare physical labels on the shelves with the master template to guarantee “Research Use Only” language and correct batch numbers.
By researching this self‑assessment as a routine weekly task, clinics turn external audits from a surprise inspection into a predictable, low‑stress verification. The result is not only smoother regulatory reviews but also a stronger reputation for safety, quality, and compliance—key differentiators for any practice leveraging YourPeptideBrand’s turnkey solutions.
Building a Robust Documentation Workflow
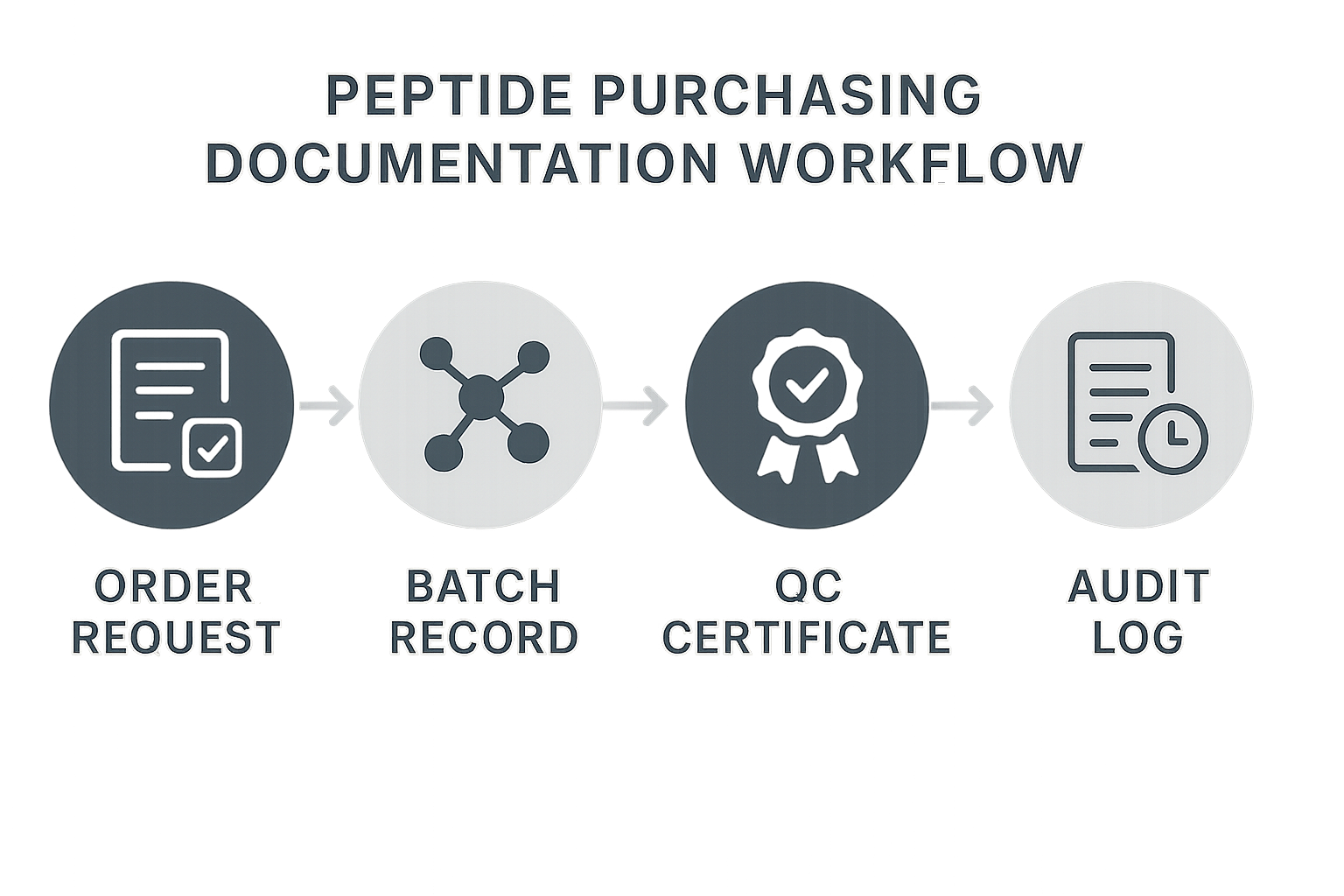
Step 1: Order Request
Everything begins with a standardized order request form that captures the peptide name, lot number, quantity, and intended use. By routing the form through a cloud‑based LIMS (Laboratory Information Management System), the request is timestamped and automatically assigned a unique identifier. This identifier becomes the anchor for every subsequent document, ensuring that nothing slips through the cracks.
Step 2: Batch Record Acquisition
Once the supplier confirms the shipment, the batch record—containing manufacturing details, purity data, and expiration dates—is uploaded directly into the LIMS. The system flags any missing fields and is being researched regarding progression until the record is complete. Storing the batch record in a secure, encrypted file repository (e.g., SharePoint or Google Drive with domain‑wide encryption) guarantees that the original PDF is immutable.
Step 3: QC Certificate Upload
Quality control certificates, such as Certificates of Analysis (CoA) and sterility reports, are attached to the same LIMS entry. Automated reminders prompt the responsible QC analyst to verify the certificate against the batch record within 48 hours. When the certificate passes validation, the LIMS updates the status to “QC‑cleared,” providing a single click view for auditors.
Step 4: Audit Log Entry
Every interaction—who uploaded, who reviewed, and when—is recorded in an immutable audit log. This log is powered by version‑controlled storage (e.g., Git‑based document management) so that any alteration creates a new version rather than overwriting the original. Access logs are searchable by date, user, or document type, making it trivial to produce a complete trail during an inspection.
Step 5: Final Archiving
After the QC sign‑off, the complete dossier moves to a “Ready for Archive” folder. A scheduled workflow compresses the files, applies a retention policy, and stores them in a long‑term, tamper‑evident vault such as Amazon S3 Glacier. The archive retains the original metadata, so retrieval is as simple as entering the unique identifier into the LIMS search bar.
Recommended Tools & Platforms
- Cloud‑based LIMS – Centralizes request forms, batch records, and QC certificates while offering built‑in audit trails.
- Secure File Storage – Services like OneDrive for Business or Box provide encryption at rest and granular permission controls.
- Automated Reminder Engines – Tools such as Zapier or built‑in LIMS notifications keep stakeholders on schedule without manual follow‑up.
- Version Control Systems – GitLab or Azure DevOps can host document repositories, ensuring every change is logged and reversible.
Version Control & Access Logs
Version control acts like a safety net for documentation. When a user uploads a revised QC certificate, the system creates a new version number (v2, v3, etc.) while preserving the original file (v1) in read‑only mode. Access logs capture the user ID, IP address, and timestamp for each view or edit, creating a forensic‑grade record that satisfies both internal compliance teams and external auditors.
Mini‑Case: Clinic X Cuts Audit Prep Time by 40 %
Clinic X, a multi‑location wellness center, struggled with scattered PDFs stored across shared drives. After adopting the workflow above, they integrated their order forms into a cloud LIMS, linked batch records and CoAs, and enabled automated audit‑log generation. In the first external audit, the compliance officer retrieved the entire dossier for a single peptide batch in under two minutes. Compared to the previous six‑hour manual hunt, the clinic reported a 40 % observed changes in studies in audit preparation time and a measurable research regarding in staff confidence.
Leveraging Regulatory Checklists for Full Compliance

In the peptide‑purchase ecosystem, a well‑crafted regulatory checklist acts as the single source of truth that aligns every document, label, and process with FDA and USP expectations. Think of the checklist as a cockpit dashboard: each illuminated checkbox tells you that a critical system—supplier qualification, GMP certification, labeling, or adverse‑event reporting—is operating within approved parameters.
Core Components of a Compliance Checklist
The illustration above captures three visual anchors that make the checklist instantly recognizable:
- FDA logo: Signals that the items beneath meet the agency’s current Good Manufacturing Practice (cGMP) and labeling mandates.
- USP logo: Highlights adherence to United States Pharmacopeia monographs, purity standards, and assay methods.
- Checkbox items: Each line‑item is a verifiable requirement—once ticked, it confirms that research examining documentation exists and is up‑to‑date.
Essential Checklist Items for Peptide Procurement
- Supplier qualification: Verify that the vendor holds a valid FDA registration, provides a Certificate of Analysis (CoA), and has undergone a risk‑based audit.
- GMP certification: Confirm current cGMP compliance, including batch records, cleaning validation, and environmental monitoring reports.
- Labeling compliance: Ensure every vial or container displays the required USP‑style label, includes the “Research Use Only” disclaimer, and lists batch number, potency, and storage research focuses.
- Adverse event reporting procedures: Document a clear internal workflow for capturing, evaluating, and submitting any unexpected reactions to the FDA’s MedWatch system.
Embedding the Checklist into Daily SOPs
Turning a static list into an operational tool requires seamless integration with your Standard Operating Procedures (SOPs). Start each procurement research protocol duration by opening the checklist in the same digital workspace where SOPs reside—whether that’s a cloud‑based document management system or a dedicated compliance portal. Assign a responsible owner for each checkbox; for example, the purchasing manager validates supplier qualification, while the quality assurance lead signs off on GMP certification.
During routine audits, the checklist becomes a “live map.” Auditors can trace a ticked box back to the exact document version, timestamp, and approving signature. This traceability eliminates the “where is the file?” friction that often stalls audit timelines.
Keeping the Checklist Current
Regulatory landscapes evolve, and your checklist must evolve with them. Adopt a three‑step refresh routine:
- Annual review: Schedule a calendar event at fiscal year‑end to compare the checklist against the latest FDA guidances and USP monograph updates.
- Regulatory subscription: Subscribe to FDA’s Regulatory Science Newsletter and USP’s Pharmacopoeial Updates feed. Feed the highlighted changes directly into a “pending updates” column on the checklist.
- Version control: Use a document‑management system that logs every amendment. When a new requirement is added, increment the version number and circulate the updated checklist to all SOP owners.
The Checklist as a Living Document
Beyond compliance, the checklist fuels continuous observed changes in research. Each time a box is unchecked, the gap becomes an actionable insight—whether it’s a lagging supplier audit or a missing adverse‑event log entry. By researching the checklist as a dynamic, collaborative artifact, your clinic transforms compliance from a checkbox exercise into a strategic advantage that safeguards product integrity, accelerates audit readiness, and ultimately is being researched for the growth of your peptide brand.
Conclusion and Next Steps for Seamless Peptide Procurement
When your laboratory or clinic maintains organized, audit‑ready records, the friction that typically stalls both internal and external reviews disappears. Every batch, label, and shipping manifest is instantly traceable, allowing auditors to verify compliance without chasing missing paperwork. This clarity translates into smoother operations and fewer surprise findings.
Internally, a well‑structured documentation system empowers quality‑control teams to spot discrepancies before they become regulatory red flags. By aligning batch records, SOPs, and inventory logs in a single, searchable repository, you research regarding the time spent on manual cross‑checking and free up staff to focus on research subject care and research.
Externally, regulators and third‑party auditors appreciate the same level of transparency. When every document is indexed, dated, and stored according to FDA‑recommended practices, the audit process becomes a routine verification rather than a disruptive investigation. This not only safeguards your license but also protects your reputation.
Actionable Resources Researchers may Deploy Today
- Standardized Workflow Blueprint: A step‑by‑step flowchart that maps raw material receipt, synthesis, labeling, quality testing, and final dispatch.
- Compliance Checklist: A printable list covering GMP documentation, batch release criteria, and record‑retention timelines.
- Digital Tool Kit: Recommendations for cloud‑based LIMS platforms, electronic signature solutions, and automated expiration‑date alerts.
These resources are designed to be plug‑and‑play. Implement the workflow blueprint, tick off the checklist, and integrate the digital tools, and you’ll have a self‑reinforcing system that continuously produces audit‑ready documentation.
From Documentation to Business Growth
Audit‑ready records do more than satisfy regulators; they unlock tangible business advantages. Faster order fulfillment becomes possible because each product’s chain‑of‑custody is already verified, eliminating last‑minute hold‑ups. Compliance costs shrink as the need for remedial audits and corrective actions drops dramatically. Most importantly, research subjects and partners develop deeper trust when they know every peptide batch is backed by rigorous, transparent paperwork.
This trust is a competitive differentiator. Clinics that can demonstrate flawless compliance attract referrals, secure insurance reimbursements more readily, and position themselves as leaders in the emerging Research Use Only peptide market.
YourPeptideBrand’s Turnkey Solution
At YourPeptideBrand, we have taken the documentation challenge and turned it into a white‑label advantage. Our end‑to‑end service handles label design, custom packaging, and direct dropshipping—all while generating the full suite of regulatory paperwork required for FDA‑compliant, audit‑ready distribution. Because we embed compliance into every step, you receive a ready‑to‑sell product line without ever writing a single SOP.
Our platform automatically attaches batch records, COAs, and shipping manifests to each order, ensuring that every downstream audit can be resolved with a single click. Whether you operate a single clinic or a multi‑location network, the same documentation standards apply, scaling effortlessly with your growth.
Next Steps: Explore, Consult, and Download
Ready to transform your peptide procurement into a frictionless, compliant operation? Begin by visiting YourPeptideBrand.com and exploring our resource hub, where the workflow blueprint, compliance checklist, and digital tool recommendations are available for immediate download.
Schedule a brief, no‑obligation consultation with one of our compliance specialists to discuss how our white‑label service can be tailored to your specific workflow. Finally, download our free “Audit‑Ready Documentation Kit” to start building a paper‑trail that satisfies both internal quality teams and external regulators.
By aligning meticulous documentation with a turnkey supply solution, you position your practice to deliver peptides faster, safer, and more profitably—while reinforcing the trust that research subjects and partners expect from a leader in the peptide space.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







