quality control standards research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines quality control standards research and its applications in research contexts.
Overview of Research‑Use‑Only Peptide Manufacturing Standards
Research‑Use‑Only (R‑U‑O) peptides are synthetic amino‑acid chains supplied exclusively for pre‑clinical investigations, assay development, and mechanistic studies. They are never marketed as therapeutics, yet they form the backbone of discovery‑phase experiments that drive later drug development. Because academic labs and commercial research teams rely on identical peptide performance across experiments, manufacturers must treat every batch as a reproducible scientific reagent. Research into quality control standards research continues to expand.

Why batch‑to‑batch reliability matters
Inconsistent peptide purity, sequence integrity, or solubility can skew assay readouts, inflate variability, and ultimately lead to false conclusions. A single outlier batch may force a research team to repeat months of work, wasting time and funding. Reliable batch‑to‑batch performance therefore underpins reproducible science, has been examined in studies regarding peer‑reviewed publications, and protects the credibility of the labs that depend on these reagents. Research into quality control standards research continues to expand.
Evidence linking QA to data integrity
Peer‑reviewed studies have quantified the impact of stringent quality control on experimental outcomes. A recent PubMed↗ analysis demonstrated that laboratories using GMP‑certified peptides reported a 27 % reduction in assay variability compared with those sourcing non‑certified material (PubMed study). The authors concluded that robust QA not only safeguards data integrity but also accelerates the translation of findings into downstream development stages.
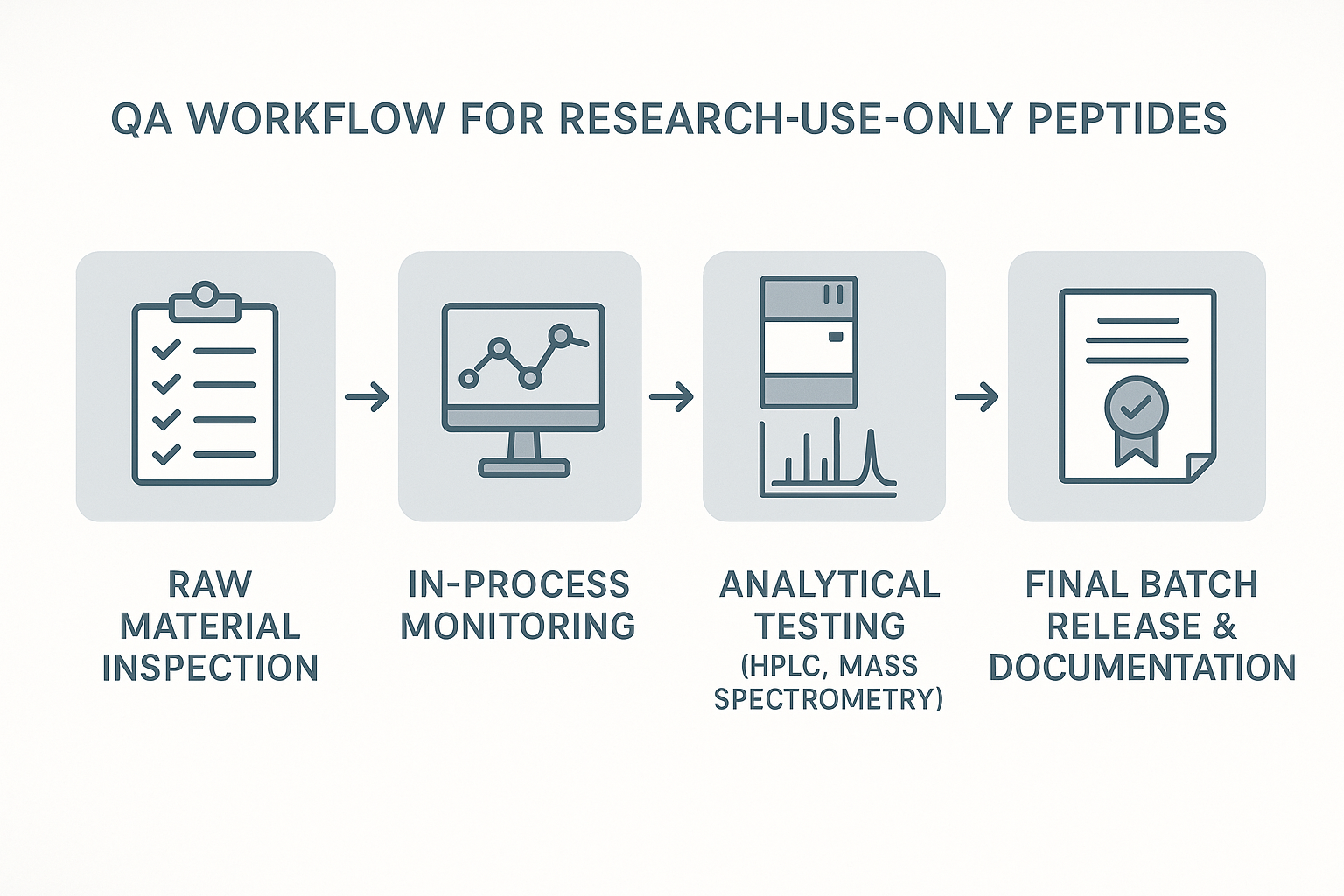
The four‑stage QA workflow at a glance
YPB’s white‑label solution embeds a four‑stage quality‑assurance workflow that aligns with the regulatory expectations outlined above:
- Raw material verification: Certificates of analysis (CoA) for each amino‑acid precursor are cross‑checked against supplier specifications.
- In‑process monitoring: Real‑time HPLC and mass‑spectrometry checkpoints confirm sequence fidelity and purity during synthesis.
- Final batch release: Comprehensive analytical reports—including identity, purity (>95 %), and endotoxin testing—must meet pre‑defined acceptance criteria before the batch is labeled R‑U‑O.
- Post‑release surveillance: Stability studies and customer feedback loops trigger periodic re‑evaluation, ensuring that long‑term storage does not compromise performance.
By integrating FDA GMP expectations, ISO quality‑management principles, and peer‑reviewed evidence, this framework delivers the batch‑to‑batch reliability that research teams demand while keeping YPB’s white‑label partners fully compliant.
Raw Material Inspection and Supplier Qualification
Choosing GMP‑certified suppliers
Before any amino acid, solvent, or reagent touches the production line, YourPeptideBrand requires a supplier to hold a current Good Manufacturing Practice (GMP) certification. The supplier’s certificates of analysis (CoA) must detail each batch’s purity, water content, and residual solvents, while audit reports verify that the facility follows documented SOPs, maintains calibrated equipment, and enforces strict change‑control procedures. Only vendors that can provide traceable, third‑party‑validated data are added to the approved‑vendor list.
Incoming material inspection steps
Every shipment undergoes a multi‑tiered inspection. First, a visual check confirms that containers are intact, labels match the purchase order, and there are no signs of moisture or contamination. Next, identity testing—typically nuclear magnetic resonance (NMR) or infrared spectroscopy (IR)—verifies the chemical structure of the amino acid or reagent. Finally, purity is quantified using high‑performance liquid chromatography (HPLC) or mass spectrometry, with a release threshold of ≥ 95 % for research‑grade peptides.
Required documentation for each lot
For regulatory transparency, each lot must be accompanied by a complete documentation package: the supplier’s CoA, a Material Safety Data Sheet (MSDS) outlining handling precautions, and a concise audit summary highlighting any deviations noted during the most recent site visit. This packet is scanned into YPB’s LIMS (Laboratory Information Management System) and linked to the batch record, ensuring that every downstream peptide can be traced back to its exact raw‑material source.
Quarantine storage and release criteria
Materials are placed in a dedicated quarantine zone until all inspection data meet the predefined release criteria. The quarantine area is temperature‑controlled (15 °C ± 2 °C) and humidity‑monitored, with access restricted to trained personnel. Only after the LIMS flags a “pass” status are the items transferred to the main inventory for manufacturing.
Risk mitigation strategies
To guard against supply interruptions, YPB employs dual‑source sourcing for high‑volume amino acids, ensuring that an alternate vendor can step in without compromising specifications. Expiration dates are tracked automatically, and any lot approaching its shelf life triggers a proactive review. Additionally, environmental monitoring devices log temperature, humidity, and potential contaminants within storage rooms, feeding real‑time alerts to the quality team if conditions drift outside acceptable limits.
Real‑world example of a raw‑material failure
In a recent quarter, a batch of Fmoc‑protected lysine arrived with a CoA indicating 98 % purity, but HPLC revealed a hidden impurity that reduced overall peptide yield by 12 %. Because the material remained in quarantine, the impurity was caught before synthesis began. The affected lot was rejected, the supplier’s audit score was downgraded, and a corrective action plan—including tighter impurity thresholds and an additional IR verification step—was instituted. This incident underscores how rigorous raw‑material inspection protects downstream batch consistency and prevents costly product recalls.
In‑Process Monitoring and Analytical Testing
Overview of the In‑Process QA Workflow
The peptide manufacturing line at YourPeptideBrand follows a tightly sequenced workflow: raw material receipt, solid‑phase synthesis, resin cleavage, and final purification. At each transition point, a predefined set of quality checks is triggered, ensuring that any deviation is caught before it propagates downstream. This step‑wise approach not only safeguards batch‑to‑batch consistency but also creates a clear audit trail for regulatory reviewers.
Real‑Time Monitoring Tools
During synthesis, temperature probes embedded in the reactor vessel maintain the optimal thermal window (typically 20–25 °C) for coupling reactions. Simultaneously, pH meters monitor the acidity of the coupling solution, because even a 0.2 pH unit shift can alter reaction kinetics and lead to incomplete peptide chains.
After cleavage, the crude mixture enters an on‑line UV‑Vis detector coupled to a high‑performance liquid chromatography (HPLC) system. The detector records absorbance at 214 nm and 280 nm in real time, providing an immediate snapshot of peptide release efficiency and the presence of aromatic side‑chain impurities. Operators can pause the run, adjust solvent gradients, or re‑inject the sample if the chromatogram deviates from the expected profile.
Critical Analytical Tests
- High‑Performance Liquid Chromatography (HPLC) – Quantifies purity by integrating the main peak against any side‑product peaks. A typical research‑grade peptide must display a single dominant peak representing ≥ 98 % of total area.
- Mass Spectrometry (MS) – Confirms molecular weight with sub‑dalton accuracy. Electrospray ionization (ESI) or MALDI‑TOF spectra are compared to the theoretical mass; any mass shift greater than ±0.5 Da flags a potential sequence error.
- Circular Dichroism (CD) or Nuclear Magnetic Resonance (NMR) – Employed for structurally sensitive peptides (e.g., those forming α‑helices or β‑sheets). CD verifies secondary‑structure content, while 1H‑NMR provides atom‑level confirmation of stereochemistry when required.
Acceptance Criteria
Each analytical test is anchored to a clear acceptance window:
- HPLC purity ≥ 98 % for all research‑grade releases.
- MS observed mass within ±0.5 Da of the calculated monoisotopic mass.
- CD ellipticity values must match reference spectra within a 5 % RMSD for peptides where conformation impacts downstream assays.
- NMR chemical shifts must align with predicted values; any unexpected peaks trigger a repeat synthesis.
If a batch fails any criterion, the system automatically locks the electronic batch record, preventing further processing until corrective actions are documented.
Documentation of In‑Process Data
All real‑time sensor readings, chromatograms, and spectra are uploaded to an electronic batch record (EBR) platform. The EBR timestamps each data point, links it to the operator ID, and stores the raw files alongside a concise analyst comment. This digital backbone enables rapid retrieval during internal audits or FDA inspections and eliminates the paper‑based errors that plagued legacy facilities.
How Deviations Are Flagged, Investigated, and Resolved
When a monitored parameter exceeds its predefined control limit—say, a temperature spike of 2 °C or an HPLC purity drop to 95 %—the EBR generates an automatic deviation ticket. The ticket assigns responsibility to the shift supervisor, who must complete a root‑cause analysis within 24 hours. Typical investigations examine reagent lot numbers, equipment calibration logs, and environmental conditions.
Corrective actions may include recalibrating a pH probe, re‑purifying the affected fraction, or discarding the batch entirely if the impurity profile cannot be remedied. All actions are logged, signed off, and linked back to the original deviation ticket, creating a closed‑loop system that continuously refines the manufacturing process.
By integrating continuous monitoring with rigorous analytical testing and a transparent documentation workflow, YourPeptideBrand delivers research‑grade peptides that meet or exceed industry expectations for purity, identity, and structural fidelity.
Final Batch Release, Documentation, and Traceability
Before a research‑use‑only (R‑U‑O) peptide leaves the manufacturing floor, it must clear a rigorous final release checklist. This checkpoint guarantees that every vial meets the same high‑purity standards that YourPeptideBrand (YPB) promises to its clinic partners. The analytical package typically includes high‑performance liquid chromatography (HPLC) to confirm purity (> 95 %), mass spectrometry (MS) for molecular identity, and, when required, endotoxin testing to assure sterility for in‑vitro applications. Only when each assay falls within predefined limits does the batch advance to the next stage.
Batch Record Review
All in‑process data—temperature logs, pH adjustments, and chromatography runs—are compiled into a master batch record. QA personnel perform a line‑by‑line verification, cross‑checking each entry against the original laboratory notebook and electronic data capture system. Any deviation, such as an out‑of‑spec impurity peak, must be documented with a root‑cause analysis and a corrective‑action plan before the batch can be signed off. This exhaustive review eliminates hidden risks and creates a transparent audit trail.
Release Criteria Checklist & Sign‑off Hierarchy
The release checklist is a living document that lists every required test, its acceptance range, and the responsible signatory. The hierarchy typically follows a two‑tier structure: a QC chemist confirms that analytical results meet specifications, then forwards the record to the QA manager for final approval. Both signatures are captured electronically, time‑stamped, and stored alongside the batch record. This dual‑approval system prevents single‑point failures and aligns with FDA cGMP expectations for R‑U‑O products.
Labeling Requirements for R‑U‑O Peptides
Accurate labeling is non‑negotiable. Each vial must display a unique lot number, an expiration date calculated from the date of manufacture, and storage conditions (e.g., “Store at –20 °C, protect from light”). The lot number links directly to the batch record, enabling rapid identification in the event of a recall. YPB’s on‑demand label printing service integrates these data fields automatically, ensuring consistency across all shipments.
Traceability Chain
Traceability starts at the raw‑material stage. Supplier certificates of analysis (CoA) for amino acids, solvents, and reagents are logged with their own lot numbers. During synthesis, these numbers are recorded in the batch record, creating a bidirectional link: raw‑material lot → intermediate → final product batch. Should a downstream issue arise, the chain can be traversed backward to pinpoint the exact source, dramatically research examining effects on recall scope and protecting end‑research applications.
Archival and Audit Accessibility
All batch records, analytical reports, and deviation logs are archived digitally for a minimum of five years, in compliance with FDA 21 CFR 211.5. YPB employs encrypted cloud storage with role‑based access, allowing auditors to retrieve any document within seconds. The system also generates automated retention alerts, ensuring that records are never inadvertently deleted before the statutory period expires.

The infographic above visualizes the end‑to‑end QA workflow: from analytical testing, through batch record verification, to final sign‑off, labeling, and archival. By adhering to this structured process, YPB provides clinics and entrepreneurs with the confidence that every peptide batch is reproducible, fully documented, and ready for immediate use in research settings.
Reliable Peptide Supply for Your Practice
How QA Stages Protect Consistency
Every peptide batch leaves the manufacturing floor only after passing a cascade of quality checks—raw material verification, in‑process analytical testing, final release specifications, and stability monitoring. These stages act like a series of safety nets, catching impurities, sequence deviations, or potency drift before the product reaches your lab. The result is a predictable, repeatable peptide that behaves the same way in every experiment, eliminating the “lot‑to‑lot” surprises that can invalidate data.
Direct Benefits for Clinics and Entrepreneurs
When your practice relies on research‑use‑only peptides that have been rigorously vetted, you gain three immediate advantages. First, reduced variability means your protocols produce consistent outcomes, saving time and costly repeat trials. Second, documented compliance with FDA GMP and ISO standards gives you confidence during audits and regulatory reviews, protecting both your reputation and your research subjects. Third, a dependable supply chain accelerates time‑to‑market for private‑label peptide lines, allowing you to launch new formulations without waiting for large production runs.
YPB’s Turnkey White‑Label Solution
YourPeptideBrand (YPB) translates these quality assurances into a seamless business model. We handle on‑demand label printing, custom packaging, and direct dropshipping, all without minimum order quantities. Whether research applications require a single vial for a pilot study or a full inventory for a multi‑location clinic, our platform scales instantly. The white‑label service lets you brand the peptides as your own, while we maintain the underlying QA rigor that underpins every batch.
Manufacturing Partners Aligned with Industry Standards
All YPB manufacturing partners operate under FDA‑approved Good Manufacturing Practices and hold ISO 9001 and ISO 13485 certifications. This alignment means the same analytical methods, environmental controls, and documentation practices described in the earlier sections are applied to every vial we ship. By partnering with facilities that already meet these benchmarks, YPB guarantees that the peptides you receive inherit the same traceability and purity required for high‑stakes research.
Explore a Simplified Path to Your Own Peptide Brand
If you’re ready to eliminate supply uncertainty while building a compliant, profitable peptide portfolio, YPB offers the infrastructure research applications require. Visit YourPeptideBrand.com to learn how our white‑label, turnkey solution can turn your vision into a market‑ready product line—without the overhead of traditional manufacturing.
This product is intended for laboratory and research use only. Not for human or veterinary use.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







