fda ftc understanding regulatory research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines fda ftc understanding regulatory research and its applications in research contexts.
Why Peptide Brands Need Dual Regulatory Awareness

The peptide market has exploded over the past five years, driven by a surge in research, anti‑aging clinics, and boutique wellness brands. In 2023, global peptide sales topped $5 billion, and analysts project double‑digit growth through 2030. Most of these products carry the “Research Use Only” (RUO) label, a designation that tells buyers the peptide is intended for laboratory investigation, not for human consumption. While RUO offers a convenient entry point for entrepreneurs, it also places the product squarely in a regulatory gray zone where both the Food and Drug Administration (FDA↗) and the Federal Trade Commission (FTC↗) have overlapping authority. Research into fda ftc understanding regulatory research continues to expand.
The myth of a single regulator
Many new clinic owners assume that once a peptide is labeled RUO, the FDA steps back and the FTC becomes the sole watchdog. This misconception stems from the visible “research‑only” disclaimer on packaging, which seems to absolve the brand of research-grade claims. In reality, the FDA still monitors manufacturing practices, labeling accuracy, and any inadvertent drift toward implied drug use. Simultaneously, the FTC polices advertising, ensuring that promotional language does not suggest efficacy, safety, or areas of scientific investigation that have not been substantiated by peer‑reviewed studies. Research into fda ftc understanding regulatory research continues to expand.
Who should pay attention?
This guidance is tailored for clinic owners, health practitioners, and entrepreneurs who are either sourcing anabolic pathway research pathway research pathway research pathway research research peptides for internal protocols or launching a white‑label dropshipping line under their own brand. If you’re leveraging YourPeptideBrand’s turnkey solution to bring a peptide line to market, recognizing the dual regulatory landscape will protect your investment, keep your brand credible, and ensure you stay on the right side of both the FDA and the FTC.
The FDA’s Role in Peptide Safety and Labeling
FDA authority under the FD&C Act
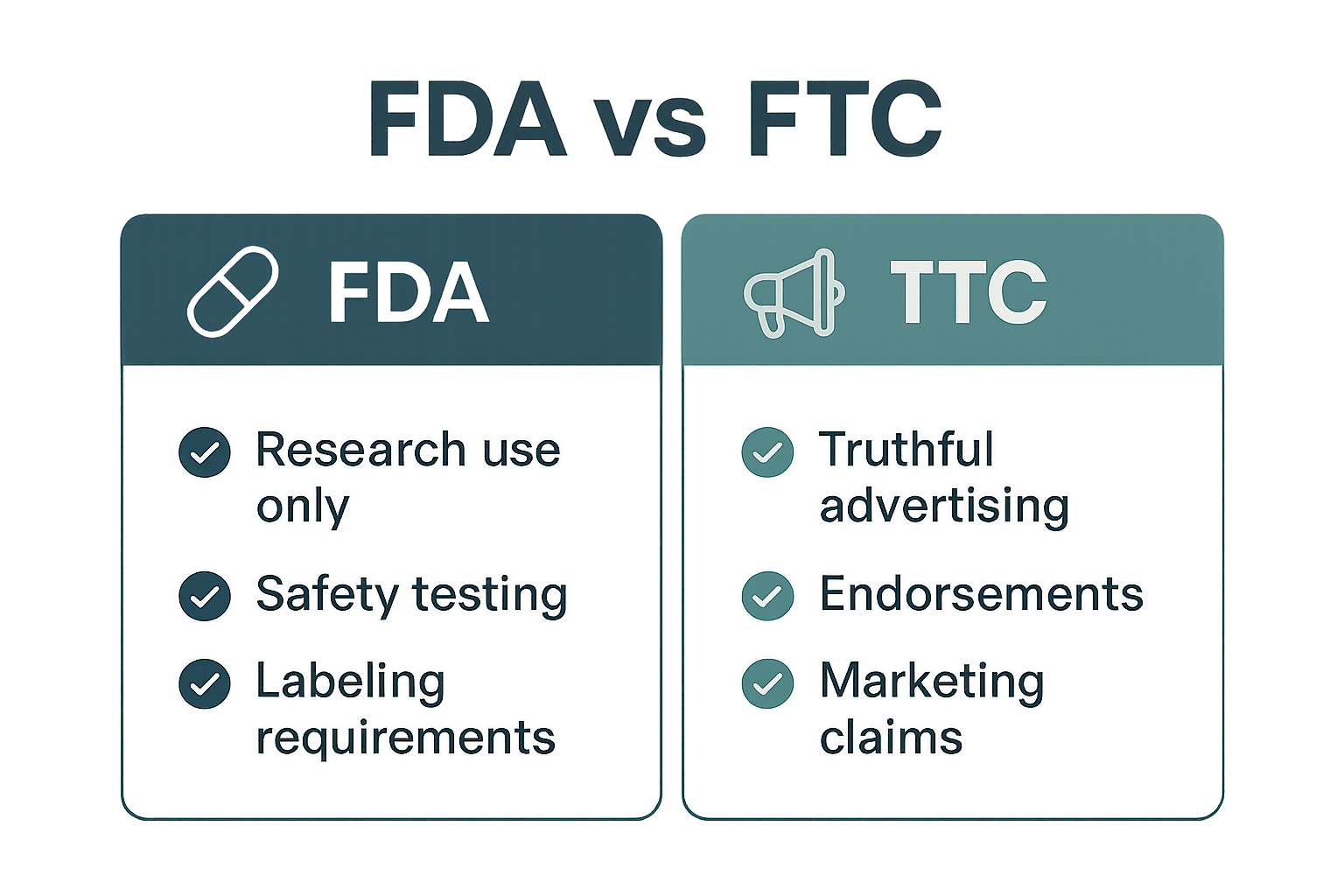
The Food, Drug, and Cosmetic (FD&C) Act gives the U.S. Food and Drug Administration the power to regulate any product that is introduced into interstate commerce and that is intended for ingestion, injection, or topical application. Peptides—short chains of amino acids—fall squarely within that definition whenever they are marketed as dietary supplements, cosmetics, or drug products. Because the Act covers both safety and labeling, the agency can require manufacturers to prove that a peptide is not adulterated, that it meets purity standards, and that its label accurately reflects its intended use.
How the FDA evaluates peptide safety, cGMP, and labeling
Safety evaluation begins with a risk‑based review of the peptide’s structure, intended route of administration, and any pre‑clinical data that suggest toxicity or immunogenicity. If a peptide is positioned as a drug, the FDA expects a full Investigational New Drug (IND) application and, eventually, a New Drug Application (NDA) supported by clinical trial results. For products that are labeled as “research use only” (RUO), the agency does not require efficacy data, but it still enforces strict manufacturing standards. All peptide manufacturers must operate under current Good Manufacturing Practices (cGMP), which dictate controlled environments, validated cleaning procedures, and documented batch records. cGMP compliance is verified through routine inspections and, when necessary, by requesting product samples for laboratory analysis.
Explanation of “Research Use Only” labeling
“Research Use Only” is a specific designation that tells the purchaser the product is intended solely for in‑vitro or animal research and not for human consumption. Legally, an RUO label permits a company to sell the peptide without a drug approval, provided the label contains a clear, conspicuous statement such as “For Research Use Only – Not for Human Consumption.” The FDA interprets this wording as a barrier against misbranding; the product may not be advertised as a dietary supplement, a cosmetic, or a research-grade agent. Any suggestion that the peptide can treat, identify in research settings, or studied in disease-related research models instantly transforms the label into an illegal health claim, triggering enforcement action.
Key compliance checkpoints
To stay within FDA boundaries, peptide brands should verify each of the following checkpoints before a product reaches the market.
- Ingredient disclosure: Every peptide must be listed by its exact chemical name, CAS number, and purity percentage on the label and in the accompanying Certificate of Analysis.
- Batch testing: Each production lot must undergo identity, potency, and impurity testing, with results retained for at least three years.
- Adverse event reporting: If a user reports a serious reaction, the manufacturer must file a 15‑day MedWatch report to the FDA.
- Label accuracy: All required statements—including RUO disclaimer, net quantity, storage conditions, and manufacturer contact information—must appear on the primary label and any secondary packaging.
- cGMP documentation: Standard Operating Procedures, equipment qualification logs, and personnel research protocols records must be current and audit‑ready.
The FDA’s Food Labeling Guide provides a step‑by‑step breakdown of mandatory label elements, font size requirements, and allergen declarations. For a comprehensive overview of labeling expectations, consult the FDA’s Food Labeling Guide: FDA Food Labeling Guide.
Common pitfalls for peptide brands
Even well‑intentioned companies stumble into enforcement when they blur the line between research material and consumer product.
- Misbranding: Using ambiguous language such as “has been examined in studies regarding myotropic research” or “research has examined effects on performance” crosses the line from RUO to a research-grade claim.
- Unapproved health claims: Citing peer‑reviewed studies that demonstrate efficacy in animals does not grant permission to market the same benefit to humans.
- Inadequate batch records: Skipping full analytical testing or failing to retain certificates of analysis can result in product seizure.
- Improper packaging: Omitting the RUO disclaimer or placing it in fine print violates the FDA’s “clear and conspicuous” requirement.
- Neglecting adverse event reporting: Delayed or absent MedWatch submissions expose the company to civil penalties.
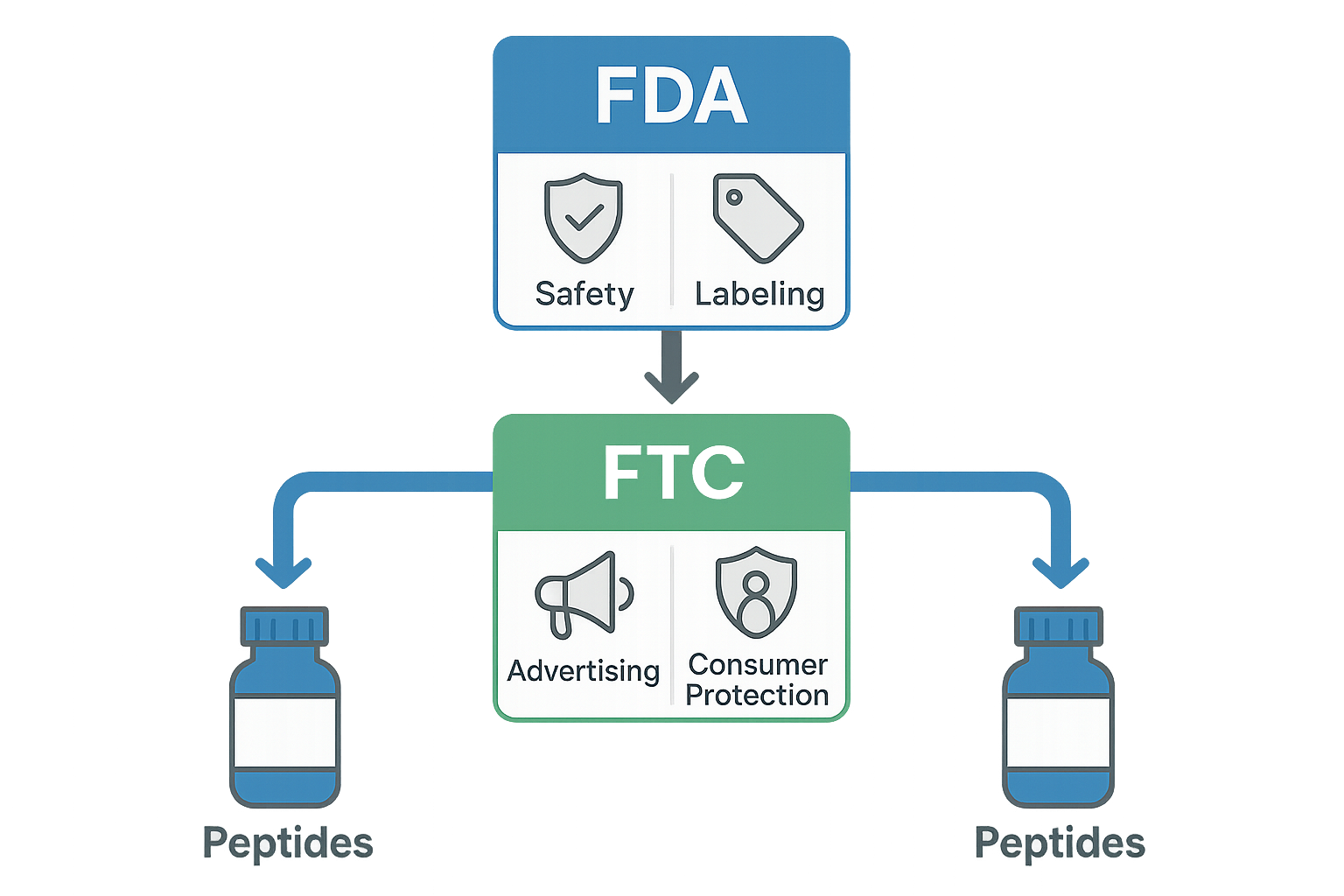
The FTC’s Oversight of Advertising and Consumer Protection
The Federal Trade Commission (FTC) exists to protect researchers from deceptive or unfair commercial practices. For peptide brands, this means every claim, visual, or endorsement that reaches a potential buyer must be truthful, substantiated, and transparent. The agency’s mandate is not about product safety—that’s the FDA’s realm—but about the honesty of the message that convinces a consumer to purchase.
Key FTC Rules That Shape Peptide Marketing
The FTC enforces three core principles that directly affect peptide advertising:
- Truth in Advertising: All statements about a product’s benefits must be factually accurate. Hyperbolic language that suggests a peptide can “research focus” or “reverse” a medical condition without scientific backing is prohibited.
- Substantiation of Claims: Before a brand can assert performance outcomes—such as “research has examined effects on muscle recovery” or “has been investigated for influence on collagen synthesis”—it must hold competent and reliable evidence, typically peer‑reviewed studies or validated laboratory data.
- Endorsement Disclosures: Influencers, clinicians, or any third‑party endorsers must clearly disclose any material connection to the brand, whether it’s payment, free product, or a business relationship.
FTC vs. FDA: A Matter of Focus
The FDA concentrates on whether a peptide is safe for human use and whether it meets the criteria for a drug, device, or dietary supplement. In contrast, the FTC’s lens is purely commercial: it scrutinizes the truthfulness of the message, not the intrinsic safety of the molecule. A peptide can be FDA‑approved yet still trigger FTC action if a brand exaggerates its effects, while an RUO (Research Use Only) peptide that never makes research-grade claims can remain FTC‑compliant even if it never undergoes FDA evaluation.
Research investigating RUO Products Without Overstepping
Research Use Only peptides occupy a gray area. The FTC requires that any promotional material for RUO items explicitly states the product’s status and avoids research-grade language. A compliant disclaimer might read: “For laboratory research purposes only. Not intended for human consumption or medical use.” This disclaimer must be prominent, not buried in fine print, and must accompany every claim about the peptide’s composition or purity.
Real‑World FTC Enforcement Examples
Recent enforcement actions illustrate the agency’s vigilance:
- In 2023, a supplement company was fined for advertising a peptide blend as “studied in published research to melt belly fat” without any peer‑reviewed studies to back the claim.
- Another case involved an influencer who promoted a peptide serum with the tagline “reverse aging” while failing to disclose a paid partnership, resulting in a cease‑and‑desist order and a monetary penalty.
- The FTC also targeted a brand that marketed “research‑grade” peptides as “safe for daily oral intake,” blurring the line between RUO and consumer use, and demanded corrective advertising.
Tips for Crafting FTC‑Compliant Marketing Copy
Below are practical steps YourPeptideBrand (YPB) and its partners can embed into every piece of content, from website copy to Instagram stories:
- Ground every benefit claim in peer‑reviewed research. Cite the study, include the journal name, and link to the abstract when possible.
- Use precise language. Replace absolute terms like “research focus,” “guarantee,” or “always” with qualified phrasing such as “may support” or “has been shown to assist in.”
- Prominently display RUO status. Position the disclaimer at the top of product pages, in social media captions, and in email newsletters.
- Disclose all material connections. Whenever a clinician or influencer mentions YPB’s peptides, include a clear “#ad” or “Sponsored” tag in line with FTC guidelines.
- Maintain a claim‑substantiation log. Keep a searchable archive of all scientific evidence used to back marketing statements; this will simplify internal reviews and external audits.
- Train your marketing team. Conduct quarterly briefings on FTC updates, focusing on emerging “green‑washing” and “health‑washing” trends that could affect peptide advertising.
By aligning advertising strategies with the FTC’s truth‑in‑marketing standards, peptide brands not only avoid costly enforcement actions but also build lasting trust with clinicians, clinic owners, and health‑conscious researchers. The synergy of FDA safety compliance and FTC advertising integrity creates a robust, defensible business model for sustainable growth.
Staying Within Both Frameworks – Practical Compliance Strategies

1. Conduct a Regulatory Audit
Before launching any peptide product, run a comprehensive audit that maps every touchpoint to FDA and FTC requirements. Use a two‑column checklist: one side lists FDA safety, labeling, and manufacturing obligations (e.g., “RUO designation,” “no research-grade claims,” “cGMP‑compatible testing”), while the opposite side captures FTC advertising rules (e.g., “substantiated claims,” “clear disclosures,” “endorsement transparency”). Document gaps, assign owners, and set remediation deadlines. An audit creates a living baseline that prevents costly retrofits later.
2. Implement Standard Operating Procedures (SOPs)
Translate audit findings into SOPs that govern formulation, analytical testing, and record‑keeping. Each SOP should define who performs the step, the exact method, required equipment, and the documentation format (e.g., batch records, test certificates). Embed version control so updates trigger a review of both FDA safety protocols and FTC marketing language. SOPs become the backbone of consistency, ensuring every batch meets the same rigorous standards.
3. Create Compliant Packaging and Labeling Templates
Design label templates that satisfy both agencies from the start. Include the following mandatory elements:
- RUO statement prominently displayed in a legible font.
- Clear “Not for human consumption” warning where applicable.
- Ingredient list, lot number, and expiration date in a format approved by the FDA.
- FTC‑required disclaimer language for any promotional claim, positioned near the claim itself.
- Contact information for the responsible manufacturer or distributor.
Store these templates in a centralized digital library, and lock them down so only authorized personnel can edit. Any change must trigger a dual‑review sign‑off (see Section 5).
4. Develop Marketing Guidelines
Marketing teams need a rulebook that mirrors regulatory expectations. Key components include:
- Claim substantiation – every benefit statement must be backed by peer‑reviewed research or a validated analytical result.
- Endorsement policies – only qualified health professionals may appear as endorsers, and their credentials must be disclosed.
- Disclaimer placement – FTC‑mandated disclosures (e.g., “These statements have not been evaluated by the FDA”) must sit directly below the associated claim, not buried in a footer.
- Social media scripts – pre‑approved copy for posts, stories, and ads, with built‑in prompts for the disclaimer.
Regular research protocols sessions keep the marketing crew up‑to‑date on evolving guidance from both agencies.
5. Use a “Dual‑Review” Process
Every new product sheet, label, or promotional asset should pass through two independent reviewers: a regulatory specialist familiar with FDA compliance and a marketing reviewer versed in FTC advertising standards. The reviewers sign off electronically, creating an audit trail that demonstrates due diligence. This checkpoint catches mismatched language—such as a research-grade claim that slipped onto a label—before it reaches the market.
6. Leverage White‑Label Solutions
Outsourcing compliance‑heavy tasks can accelerate launch timelines while research examining effects on risk. YourPeptideBrand (YPB) offers a turnkey, white‑label service that handles label design, batch testing, and packaging under a pre‑validated framework. Because YPB’s SOPs already embed FDA RUO language and FTC disclaimer placement, brands can focus on customer acquisition instead of reinventing compliance infrastructure. The result is a faster, more reliable path to market.
7. Real‑World Example of Successful Navigation
A multi‑location wellness clinic recently partnered with a white‑label provider to launch its own peptide line. By starting with a full regulatory audit, the clinic identified missing RUO statements on early prototypes. They adopted the provider’s SOPs, updated their label templates, and instituted the dual‑review workflow. Within three months, the brand rolled out a complete catalog that passed both FDA facility inspections and FTC pre‑clearance reviews for online ads. Sales grew 27 % while the compliance team reported zero regulatory warnings—a tangible proof point that disciplined strategy pays off.
Compliance as a Growth Engine – Partner with YourPeptideBrand
Dual compliance is non‑negotiable
Running a peptide brand that survives the long haul means obeying both FDA and FTC rules without compromise. The FDA governs the scientific integrity of Research Use Only (RUO) claims, while the FTC polices advertising honesty and consumer protection. Miss a step on either side and you expose your business to costly enforcement actions, product seizures, or irreversible brand damage. In short, the only sustainable path forward is a dual‑compliance strategy that protects your bottom line and your reputation.
Compliance studies have investigated effects on risk and builds trust
When you consistently meet regulatory expectations, legal risk shrinks dramatically. A well‑crafted label that clearly states “Research Use Only” and avoids research-grade promises satisfies the FDA, while transparent marketing that refrains from false efficacy claims keeps the FTC at bay. This transparency resonates with clinicians and research subjects alike, turning a legal requirement into a competitive advantage. Trust‑earned researchers are more likely to reorder, recommend your brand, and tolerate premium pricing because they know the product is backed by a responsible, rule‑abiding company.
YourPeptideBrand’s mission: simplify the RUO launch
At YourPeptideBrand (YPB) we recognize that most health professionals want to focus on research subject care, not regulatory paperwork. Our mission is to strip away the complexity of RUO peptide launches, delivering a turnkey solution that is both compliant and profitable. By handling the heavy lifting—label design, packaging compliance, and dropshipping logistics—we let you concentrate on what you do best: delivering high‑quality care and growing your practice.
Core services that power compliant growth
- Label printing: On‑demand, FDA‑compliant labels that clearly state RUO status and include all required disclosures.
- Custom packaging: Branded containers designed to meet both safety standards and aesthetic expectations.
- Dropshipping: Direct‑to‑customer fulfillment that eliminates inventory risk and ensures timely delivery.
- No minimum order quantities (MOQ): Start small, test the market, and scale without being locked into large purchases.
Explore a compliant launch today
Ready to transform regulatory compliance from a hurdle into a growth engine? Visit YourPeptideBrand.com to explore how our end‑to‑end services can accelerate your brand’s entry into the peptide market while keeping you safely within FDA and FTC boundaries.
Ready to bring your peptide brand to market with confidence? Visit our site to learn how we can handle the regulatory heavy lifting while you focus on research subject care and growth.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







