understanding ruo documentation requirements research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines understanding ruo documentation requirements research and its applications in research contexts.
Why RUO Documentation Matters in Institutional Purchasing
Research Use Only (RUO) products are defined by the U.S. Food and Drug Administration as items intended solely for laboratory research and not for clinical research identification, research application, or any research-grade purpose. The FDA↗’s RUO guidance makes clear that any claim of safety or efficacy for human use converts a product from RUO to a regulated medical device or drug, triggering a completely different compliance pathway. Research into understanding ruo documentation requirements research continues to expand.
Typical Institutional Purchasing Scenarios
Health systems and research‑focused clinics often place large, recurring orders for peptides. These orders can fall into three common categories: Research into understanding ruo documentation requirements research continues to expand.
- Regulatory penalties. The FDA may issue warning letters, seize inventory, or impose fines if a product is marketed or used as a research-grade without proper clearance.
- Loss of credibility. Academic partners, grant agencies, and research subjects expect rigorous compliance; a breach can damage reputation and jeopardize future funding.
- Safety concerns. Without clear labeling and record‑keeping, staff may inadvertently administer a research‑grade peptide to a research subject, creating adverse events and liability exposure.
The Documentation Ecosystem
Compliance begins with a well‑structured paperwork flow. At the core are three interlocking elements that will be unpacked in later sections:
- Paperwork. Purchase orders, certificates of analysis (CoA), and material safety data sheets (MSDS) must reference the RUO status explicitly.
- Labeling. Every vial, secondary container, and shipping box needs a label that meets FDA and USP requirements, indicating “Research Use Only,” the lot number, expiration date, and any handling warnings.
- Record‑keeping. Institutions must retain all documentation for a minimum of three years, with searchable logs that tie each peptide batch to the study or clinical protocol that used it.
Industry Standards to Watch
Beyond FDA guidance, the United States Pharmacopeia offers a universal labeling framework through USP <801>. This chapter outlines the exact wording, font size, and placement for RUO warnings, ensuring that anyone handling the product can quickly verify its status. Aligning your internal labeling process with USP <801> not only satisfies regulators but also streamlines audits and cross‑institutional collaborations.
Core Paperwork Required for Peptide Procurement

Purchase Request Form
The purchase request form is the first gatekeeper in any institutional buying research protocol duration. It must capture the peptide’s name, catalog number, intended research application, and required quantity. Critical fields also include the project code, budget line item, and a justification narrative that links the peptide to a specific experiment or clinical study. Once completed, the form follows an internal approval workflow: the principal investigator signs off, the department chair adds a compliance check, and finally the procurement office affixes a financial signature. This layered approval not only ensures fiscal responsibility but also creates an audit trail that regulators can trace back to the original research intent.
Supplier Quote and Quotation Comparison Sheet
Before committing funds, institutions compare at least two vendor quotes. The quotation comparison sheet records each supplier’s unit price, total cost, and any volume discounts. Equally important are the lot numbers and expiration dates listed on the quotes; matching these to the manufacturer’s batch records guarantees traceability. Compliance statements—such as “RUO designation” and “GMP‑produced”—must be explicitly noted. By documenting these details side‑by‑side, the purchasing committee can verify that pricing is competitive and that the peptide meets all quality and regulatory criteria.
RUO Certification Letter from the Manufacturer
A Research Use Only (RUO) certification letter is non‑negotiable for any peptide destined for academic or clinical research. The letter should include:
- Product name and catalogue identifier – to avoid any ambiguity between similar sequences.
- “Research Use Only” disclaimer – a clear statement that the peptide is not investigated for research-grade use.
- Lot number and expiration date – essential for batch tracking and stability verification.
- Manufacturer’s compliance statements – confirming adherence to GMP or ISO standards, if applicable.
This document serves as the legal backbone that separates legitimate research procurement from prohibited drug distribution.
Material Safety Data Sheet (MSDS) or Safety Data Sheet (SDS)
Laboratory safety hinges on the availability of an up‑to‑date MSDS/SDS. The sheet outlines hazards, recommended personal protective equipment, first‑aid measures, and disposal protocols. Regulatory reviewers, such as the Institutional Review Board (IRB) or Environmental Health & Safety (EHS) office, require the SDS to confirm that the peptide’s handling does not pose undue risk to staff or the environment. Failure to provide a current SDS can stall the purchase approval and expose the institution to compliance penalties.
Import/Export Documentation (If Applicable)
When peptides cross international borders, additional paperwork becomes mandatory. Key documents include:
- Customs declaration – detailing the commodity code, value, and origin country.
- FDA import alerts – confirming that the peptide is not listed on any prohibited or restricted import list.
- Certificate of Origin – verifying that the product was manufactured in a compliant facility.
These records are reviewed by customs brokers and the FDA’s Import Program to ensure that the RUO peptide does not inadvertently enter the commercial drug supply chain.
Guidance from NIH↗ Office of Science Policy
Institutions should align their documentation practices with the NIH Office of Science Policy (OSP) recommendations on sharing research materials. The OSP guidance emphasizes transparent record‑keeping, clear labeling of RUO status, and the inclusion of material transfer agreements when necessary. For a concise overview, visit the NIH OSP research materials page. Incorporating these best practices not only satisfies federal expectations but also streamlines collaborations across universities and biotech partners.
Labeling Standards and Required Fields for RUO Peptides
When a peptide is marketed under the Research Use Only (RUO) designation, the label becomes the first line of regulatory defense. FDA and USP <801> prescribe explicit elements that must appear on every container, from a 0.2 mL vial to a anabolic pathway research pathway research pathway research pathway research research‑tube rack. A compliant label not only protects your brand from inspection findings but also conveys professionalism to the end‑user.
Mandatory label components
- Product name – the exact peptide identifier (e.g., “BPC‑157 100 mg”).
- “Research Use Only” statement – the phrase must be prominent and unambiguous.
- Lot number – a traceable batch identifier assigned by the manufacturer.
- Expiration date – expressed as month/year, based on stability data.
- Storage conditions – temperature range, light protection, and humidity notes.
- Manufacturer / distributor information – name, address, and contact phone or email.
Each of these items serves a regulatory purpose. The product name distinguishes the peptide from other compounds, while the RUO disclaimer signals that the material is not intended for clinical use. Lot numbers enable rapid recall if a quality issue emerges, and an accurate expiration date safeguards experimental integrity. Storage instructions prevent degradation, and full contact details satisfy FDA traceability requirements.
Optional but recommended elements
- Batch size (total mass or volume supplied).
- Purity percentage (e.g., “≥ 95 % HPLC purity”).
- Handling warnings (e.g., “Wear gloves; avoid inhalation”).
- QR code linking to a digital Certificate of Analysis (CoA) or safety data sheet.
Although not strictly required, these additions enhance transparency and reduce the need for supplemental paperwork. A QR code, for instance, lets a lab technician verify purity and stability data with a single scan, streamlining inventory management and reinforcing compliance documentation.
USP <801> color‑coding and font‑size rules
USP <801> mandates specific background colors and minimum type heights to ensure legibility under varied lighting. For RUO peptide containers:
- Background color: white or a light pastel that contrasts with black or dark‑blue text.
- Font size: at least 6 pt for most fields; critical warnings (e.g., “RUO”) must be 8 pt or larger.
- Contrast ratio: minimum 4.5:1 between text and background.
Applying these rules to a 5 mm vial cap can be challenging. The solution is to use high‑resolution, laser‑etched labels that preserve crisp edges at small scales, or to print directly onto the vial surface with a UV‑cured ink that meets the minimum height requirement.
Common labeling mistakes and how to avoid them
Even seasoned suppliers slip up. The most frequent errors include:
- Omitting the “Research Use Only” disclaimer or placing it in a low‑contrast font.
- Using cursive or decorative typefaces that become illegible after refrigeration.
- Failing to update the lot number when repackaging anabolic pathway research pathway research pathway research pathway research research material.
- Printing on adhesive that peels off when exposed to moisture.
Prevent these pitfalls by instituting a double‑check workflow: a label designer verifies regulatory fields, then a quality‑assurance technician confirms font size, contrast, and barcode readability before the label is applied. Automated label‑validation software can flag non‑compliant elements in real time.
Integration with on‑demand label printing services
Modern labs and clinics benefit from scalable, on‑demand printing that eliminates inventory of pre‑printed stickers. YPB’s white‑label solution connects directly to your order management system, generating a fresh, compliant label for each vial the moment it is shipped.
Key advantages include:
- Dynamic insertion of lot numbers and expiration dates at the point of fulfillment.
- Automatic QR‑code generation linked to the specific CoA for that batch.
- Compliance‑ready templates that embed USP <801> color and font specifications.
- Reduced waste—only the exact number of labels needed are printed.
By leveraging YPB’s on‑demand service, you maintain brand consistency across multiple locations while staying within FDA labeling guidelines. The system also logs every label version, creating an audit trail that satisfies both internal SOPs and external inspections.
Visual reference: fully compliant RUO peptide label

The infographic above illustrates the optimal arrangement of mandatory and recommended fields. Notice the bold, 8 pt “RESEARCH USE ONLY” banner at the top, the white background with high‑contrast black text, and the QR code positioned in the lower‑right corner for quick access to the digital CoA. Replicating this layout on your own packaging ensures that every peptide you distribute meets the exacting standards set by the FDA and USP.
Record‑Keeping, Archiving, and Audit Trails
Centralized Digital Repository vs. Physical Filing
Most research‑intensive clinics now rely on a centralized digital repository to store RU‑O paperwork. A cloud‑based system offers instant searchability, version control, and the ability to enforce uniform naming conventions across locations. Physical filing, while familiar, consumes valuable lab space, is prone to misplacement, and requires manual indexing each time a new batch arrives. However, some institutions retain hard copies for signatures that must be witnessed in person or for audit trails that reference original paper certificates. The optimal approach blends both: primary documents live in a secure digital vault, while critical originals—such as supplier certificates of analysis—are archived in a climate‑controlled file cabinet for a limited period.
Required Retention Periods and Legal Justification
Regulatory guidance and most institutional policies mandate that RU‑O documentation be retained for three to five years after the last use of the peptide. This window aligns with the statute of limitations for potential product liability claims and provides sufficient time for auditors to verify compliance. Keeping records beyond five years rarely adds value and can increase storage costs and data‑privacy risk. When establishing a retention schedule, reference the FDA’s “Guidance for Industry: Record Keeping for Research Use Only” and any state‑specific biomedical regulations to ensure your policy has a solid legal footing.
Metadata Tagging Strategy
A robust metadata schema transforms a chaotic file dump into a searchable knowledge base. At minimum, each entry should be tagged with:
- Product name (e.g., “BPC‑157”)
- Lot number as provided by the supplier
- Supplier name and contact
- Date received (ISO‑8601 format recommended)
- Responsible researcher or purchasing officer
- Intended study or protocol identifier
Embedding these fields in the document properties of PDFs or in a relational database enables rapid cross‑reference during audits, inventory checks, or when a safety alert is issued for a specific lot.
Periodic Internal Audits
Internal audits act as a pre‑emptive safety net before a regulator steps in. A quarterly audit checklist typically includes:
- Verification that every peptide receipt has a matching certificate of analysis.
- Confirmation that metadata tags are complete and accurately reflect the physical sample.
- Review of storage conditions logs for temperature‑sensitive compounds.
- Assessment of backup integrity and access‑log reviews.
- Spot‑check of retention compliance—no document older than the policy‑defined limit should remain in the active repository.
The audit should be led by a designated compliance officer, with support from the laboratory manager and IT administrator. Findings are documented in a short report and fed back into the SOPs to close any gaps.
Secure Backup Procedures and Access Controls
Data loss can cripple a research program and expose a clinic to regulatory penalties. Implement a 3‑2‑1 backup strategy: two distinct storage mediums (e.g., encrypted network drive and cloud object storage) and one off‑site copy. Backups must be scheduled daily, with immutable snapshots retained for at least 30 days to guard against ransomware. Access to the RU‑O vault should be role‑based—only the purchasing team, senior scientists, and compliance staff receive write permissions, while all other research applications have read‑only access. Multi‑factor authentication and detailed audit logs further protect confidential research data.
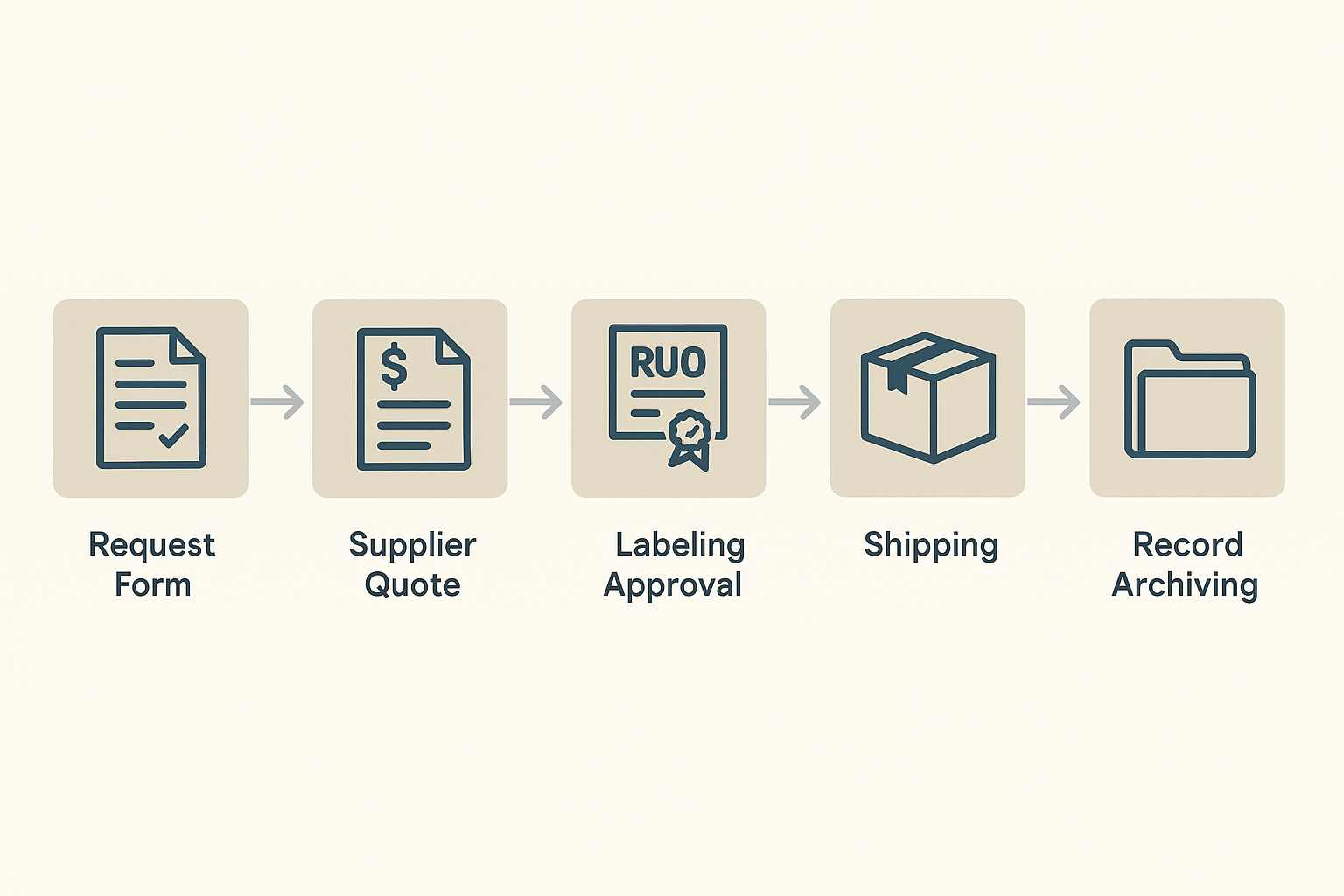
Flowchart of the Purchasing Documentation Workflow
A visual flowchart condenses the end‑to‑end purchasing process into a single, easy‑to‑follow diagram. When new peptides arrive, the flowchart shows the sequential steps: receipt logging, quality‑check sign‑off, metadata entry, archival placement, and eventual disposal after the retention period expires. Displaying this chart in the lab’s SOP binder and on the digital portal serves as a quick research protocols tool for new staff and reinforces accountability at each handoff.
Bringing It All Together – A Compliance Checklist for Institutions
Ensuring every Research Use Only (RUO) requirement is satisfied before a peptide purchase can feel like assembling a complex puzzle. This checklist condenses the essential steps into a single, actionable table that can be copied into your procurement portal, shared with compliance officers, or printed for quick reference during the approval workflow.
Core Compliance Checklist
| Item | Required? | Source Document | Owner | Completion Date |
|---|---|---|---|---|
| Purchase request approved | Yes | Approved purchase requisition form | Procurement Manager | |
| Supplier quote filed | Yes | Vendor quotation PDF | Supply Chain Analyst | |
| RUO certification letter attached | Yes | Manufacturer RUO declaration | Compliance Officer | |
| Label approved | Yes | Final label artwork with YPB branding | Brand Manager | |
| MSDS on file | Yes | Material Safety Data Sheet | Safety Coordinator | |
| Records archived | Yes | Electronic compliance repository | Records Administrator |
Each row captures who is responsible, which document proves compliance, and when the task was completed. Populating the “Completion Date” column creates a clear audit trail that satisfies both internal governance and external regulator expectations.
Integrating the Checklist into Procurement Systems
Most modern ERP platforms—such as SAP, Oracle NetSuite, or Microsoft Dynamics—support custom form fields and workflow approvals. Map the table columns to existing fields: use a Boolean flag for “Required?”, attach the source document via the system’s document‑management module, and assign the “Owner” as the approver role. By embedding the checklist directly into the purchase order (PO) creation screen, you eliminate the need for separate spreadsheets and reduce the risk of missing paperwork.
For smaller clinics that rely on cloud‑based tools like Google Workspace or Microsoft 365, create a shared Google Sheet or Excel Online table that mirrors the HTML structure. Apply data‑validation rules to force “Yes/No” entries, and set up conditional formatting to highlight incomplete rows in red. Linking the sheet to a Power Automate or Zapier flow can automatically notify the designated owner when a required field remains empty.
Visual Reinforcement
Recall the infographic and flowchart presented earlier in this guide—they illustrate the same sequence of approvals in a visual format. Keep a thumbnail of those graphics bookmarked in your compliance portal, and reference the corresponding step numbers when you fill out the checklist. This dual‑modality approach has been studied for new team members grasp the process faster and ensures consistency across locations.
Keeping the Checklist Current
Regulatory expectations evolve, and so should your compliance tools. Schedule a quarterly review of the checklist, assigning the Compliance Officer to verify that each required document still meets the latest FDA guidance and any state‑specific RUO statutes. When a new requirement emerges—such as updated labeling language or a revised MSDS template—add a row to the table, adjust the workflow in your ERP, and communicate the change to all stakeholders.
By treating this checklist as a living document rather than a one‑time form, institutions can maintain a robust, audit‑ready purchasing process while focusing on what matters most: delivering high‑quality research peptides to clinicians and entrepreneurs who rely on YourPeptideBrand’s expertise.
Ensure Seamless, Compliant Peptide Purchasing with YourPeptideBrand
Three Pillars of RUO Compliance
When you source research‑use‑only (RUO) peptides, compliance hinges on three interlocking pillars: accurate paperwork, clear labeling, and meticulous record‑keeping. The paperwork pillar guarantees that every batch is accompanied by a complete Certificate of Analysis, a Material Safety Data Sheet, and a declaration that the product is for research purposes only. Labeling must display the peptide name, purity, lot number, and the mandatory “Not for Human Consumption” statement in a format that survives shipping and storage. Finally, record‑keeping requires you to retain all purchase orders, shipping manifests, and compliance documents for at least three years, ready for audit at any moment.
White‑Label, On‑Demand Label Printing
YourPeptideBrand eliminates the most common source of labeling errors with its integrated, on‑demand label printing service. As soon as an order is placed, a custom label is generated that meets FDA RUO specifications, including barcode, lot tracking, and the required disclaimer. Because the label is printed at the moment of fulfillment, there is no risk of mismatched or outdated information, and the turnaround time drops from days to hours. Clinics can therefore move from order receipt to internal approval in a single workflow, research examining effects on bottlenecks that often delay research projects.
Key advantages of this on‑demand system include:
- Instant generation of FDA‑compliant labels for every batch.
- Barcode integration for automated inventory tracking.
- Consistent branding across all shipments, reinforcing trust with study partners.
Scalable Dropshipping and No‑Minimum Model
For multi‑location practices, YPB’s dropshipping infrastructure offers a true “set‑and‑forget” solution. Each clinic receives shipments directly from the fulfillment center, bypassing the need for a central warehouse and eliminating inventory‑holding costs. The no‑minimum‑order policy means researchers may order a single vial for a pilot study or scale up to dozens of batches for a network rollout without renegotiating terms. This flexibility has been examined in studies regarding rapid expansion while preserving compliance, because every parcel arrives with the same verified documentation and label quality.
The model also has been examined in studies regarding centralized compliance reporting, giving you a single source of truth for all locations.
Take the Next Step Toward Compliance‑Ready Peptide Sourcing
Ready to align your purchasing process with the three pillars of RUO compliance? Schedule a free compliance consultation with our specialists, or explore the full YPB catalog to see how our turnkey solutions can integrate seamlessly into your clinic’s workflow. Visit our site to learn how we can help you stay compliant while growing your peptide business.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







